
Indian Diagnostic & Therapeutic Antibodies Market Report & Forecast: 2026-2034
Market Overview:
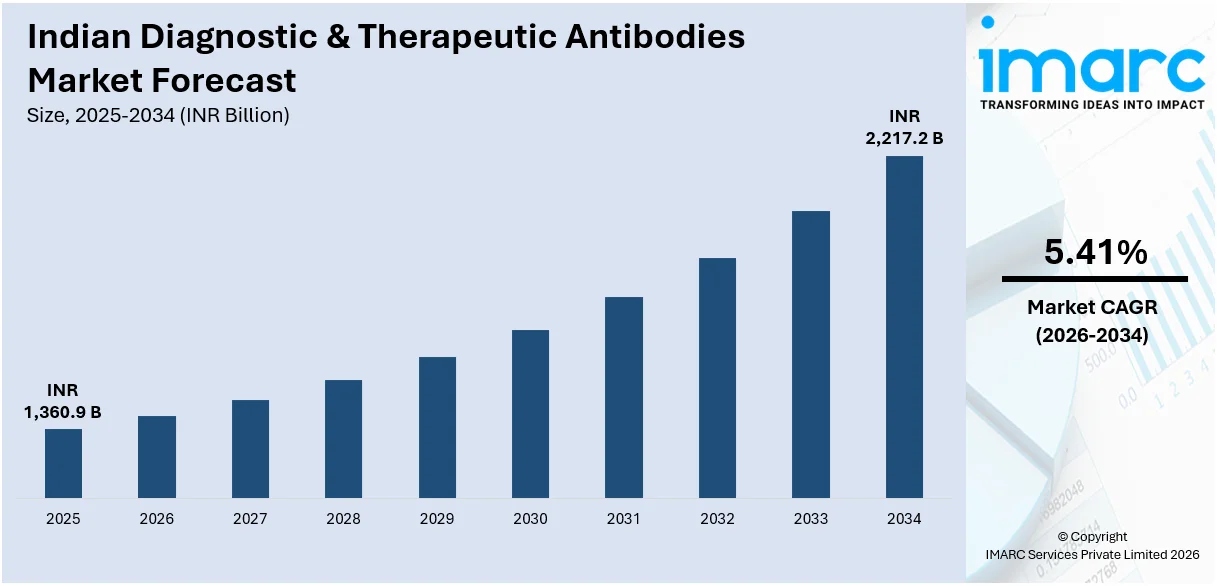
The Indian diagnostic & therapeutic antibodies market size reached INR 1,360.9 Billion in 2025. Looking forward, IMARC Group expects the market to reach INR 2,217.2 Billion by 2034, exhibiting a growth rate (CAGR) of 5.41% during 2026-2034.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
INR 1,360.9 Billion |
|
Market Forecast in 2034
|
INR 2,217.2 Billion |
| Market Growth Rate 2026-2034 | 5.41% |
Advancements in antibody technology has revolutionized both the diagnostic and therapeutic sciences. As of today, monoclonal antibodies can be produced virtually against any antigen and they have transformed the diagnostic industry in view of their specificity towards specific antigens and almost unlimited production capabilities. In the field of therapeutics, they have changed the treatment possibilities and significantly improved the life expectancies of patients who were earlier having very high unmet needs.
To get more information on this market Request Sample
IMARC Group, one of the world’s leading research and advisory firms, in its latest report “Indian Diagnostic & Therapeutic Antibodies Market Report & Forecast: 2026-2034” finds that the Indian market for both therapeutic and diagnostic antibodies is expected to grow exponentially in the coming years. Findings from the report suggest that more than 60% of the total antibodies market is currently dominated by diagnostic antibodies. This segment, however, is more mature, has lower entry barriers and is extremely price sensitive. The market for therapeutic antibodies in India, on the other hand, is still in its infancy and more than half of the globally launched therapeutic monoclonal antibodies are currently not even available here. In the coming years, however, we expect this market to explode. Driven by a continuous increase in healthcare expenditures, increasing affordability, and a number of new product launches, we expect the market for therapeutic antibodies to exhibit strong growth during 2026-2034.
IMARC’s new report entitled “Indian Diagnostic & Therapeutic Antibodies Market Report & Forecast: 2026-2034” provides an analytical and statistical insight into the Indian Antibodies market. The study, that has been undertaken using desk-based as well as qualitative primary research, provides and draws upon analysis of the following aspects of the Indian antibodies market.
Key Aspects Analyzed:
Diagnostic Antibodies
Understanding the strengths, weaknesses, opportunities and challenges in the Indian diagnostic antibodies market
Understanding the technological considerations and regulatory factors in the Indian diagnostic antibodies market
- Evaluating the various types of antibody based diagnostic tests conducted in India and analyzing their strengths and weaknesses
- Understanding the key regulations for registering, manufacturing and importing diagnostics in India
Comprehensive situation analysis of the Indian diagnostic antibodies market
Segments covered: HIV, Hepatitis B, Hepatitis C, Tuberculosis, Dengue, Syphilis and Oncology
Focus of the analysis in each segment:
- Number of tests conducted each year
- Breakup by type of tests conducted
- Current and future market sales
- Average cost of diagnostic tests
- Key players operating in the market
Therapeutic Antibodies
Understanding the strengths, weaknesses, opportunities and challenges in the Indian therapeutic antibodies market
Understanding the technological considerations and regulatory factors in the Indian therapeutic antibodies market
- Evaluation and identification of monoclonal antibodies based on the sources from which they are derived and on the targets against which they act.
- Understanding the key regulations for registering, manufacturing and importing therapeutic antibodies in India.
Evaluating the patent details of all globally marketed monoclonal antibodies in India, US and Europe
- Identifying the patentee name and patent numbers of all in-market monoclonal antibodies in India, US and Europe
- Identifying the monoclonal antibodies that are patent protected in India, US and Europe and those that are not
- Estimating the patent expiries of various monoclonal antibodies in India, US and Europe.
Comprehensive situation analysis of the Indian therapeutic antibodies market
- Analyzing current and future sales of monoclonal antibodies in India
- Identifying all globally launched monoclonal antibodies and understanding their performance and launch status in India
- Analyzing the global and Indian monoclonal antibodies market by Indication
- Evaluation of the top selling monoclonal antibodies
- Key biosimilar antibody development programmes in India and abroad
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












