
Artificial Pancreas Device Systems (APDS) Market Report by Device Type (Threshold Suspend Device Systems, Control-to-Range Systems, Control-to-Target Systems), Treatment Type (Bi-hormonal, Insulin Only, Hybrid), End-User (Hospitals, Medical Centres, and Others), and Region 2026-2034
Market Overview:
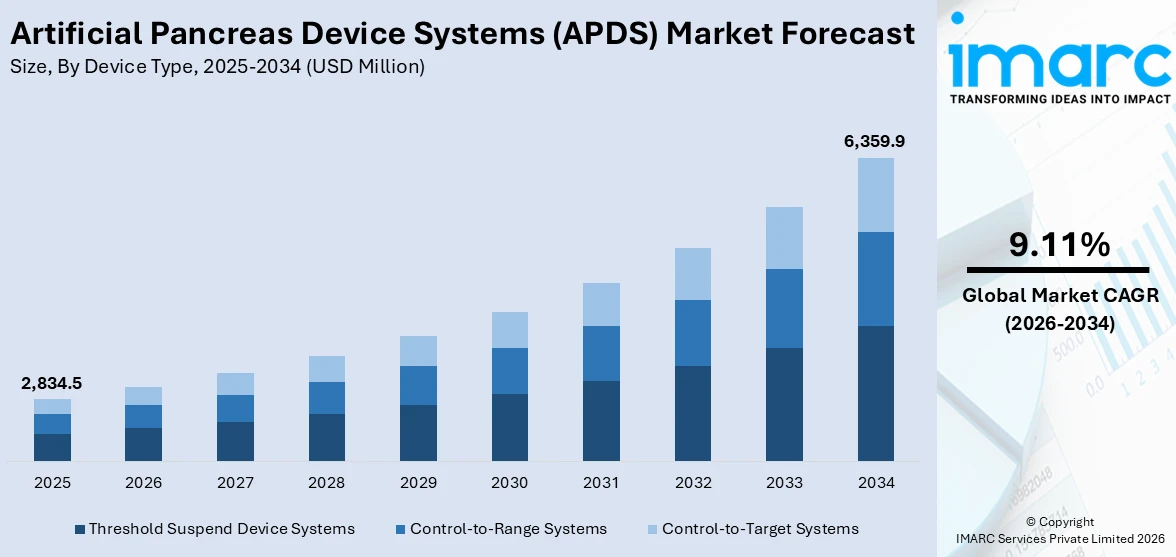
The global artificial pancreas device systems (APDS) market size reached USD 2,834.5 Million in 2025. Looking forward, IMARC Group expects the market to reach USD 6,359.9 Million by 2034, exhibiting a growth rate (CAGR) of 9.11% during 2026-2034.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
| Market Size in 2025 | USD 2,834.5 Million |
| Market Forecast in 2034 | USD 6,359.9 Million |
| Market Growth Rate (2026-2034) | 9.11% |
APDS refers to a closed-loop or automated insulin delivery system that manages blood glucose levels by regulating the production of insulin. It consists of a continuous glucose monitor (CGM), a subcutaneous insulin infusion pump and a controller that links the monitor with the pump. It runs a complex algorithm that determines the adequate levels of insulin and time of delivery based on the evaluation of real-time glucose readings. It is primarily used to minimize the risks of extreme fluctuations of glucose levels in the body.
To get more information on this market Request Sample
The increasing prevalence of pancreatic cancer, pancreatitis and other chronic ailments is one of the key factors driving the growth of the market. Furthermore, the growing occurrence of diabetes and obesity, due to age-related factors, physical inactivity and unhealthy dietary patterns, is also contributing to the widespread adoption of APDS across the globe. The rising demand for efficient diabetes monitoring and minimally invasive (MI) drug delivery systems is also providing a boost to the market growth. APDS automatically monitors the glucose levels and administers doses of insulin throughout the day in controlled amounts. Various technological innovations, including the development of software-based wireless systems that are integrated with automated controls, are further contributing to the market growth. Moreover, regulatory bodies and governments of various nations are significantly investing in research and development (R&D) for the improvement of healthcare infrastructure across the globe. For instance, the Food and Drug Association (US FDA) in the United States is approving an increasing number of new and innovative products, which is creating a positive outlook for the market in the country.
Key Market Segmentation:
IMARC Group provides an analysis of the key trends in each sub-segment of the global artificial pancreas device systems (APDS) market report, along with forecasts at the global, regional and country level from 2026-2034. Our report has categorized the market based on device type, treatment type and end-user.
Breakup by Device Type:
- Threshold Suspend Device Systems
- Control-to-Range Systems
- Control-to-Target Systems
Breakup by Treatment Type:

Access the comprehensive market breakdown Request Sample
- Bi-hormonal
- Insulin Only
- Hybrid
Breakup by End-User:
- Hospitals
- Medical Centres
- Others
Breakup by Region:
- North America
- United States
- Canada
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
Competitive Landscape:
The report provides a comprehensive analysis of the competitive landscape in the global artificial pancreas device systems (APDS) market with detailed profiles of all major companies, including:
- Beta Bionics, Inc.
- Bigfoot Biomedical Inc.
- Diabeloop
- Insulet Corporation
- Medtronic Plc
- Tandem Diabetes Care, Inc.
Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Million USD |
| Segment Coverage | Device Type, Treatment Type, End-User, Region |
| Region Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Beta Bionics, Inc., Bigfoot Biomedical Inc., Diabeloop, Insulet Corporation, Medtronic Plc, Tandem Diabetes Care, Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report:
- How has the global artificial pancreas device systems market performed so far and how will it perform in the coming years?
- What are the key regional markets?
- What has been the impact of COVID-19 on the global artificial pancreas device systems market?
- What is the breakup of the market based on the device type?
- What is the breakup of the market based on the treatment type?
- What is the breakup of the market based on the end-user?
- What are the various stages in the value chain of the industry?
- What are the key driving factors and challenges in the industry?
- What is the structure of the global artificial pancreas device systems market and who are the key players?
- What is the degree of competition in the industry?
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












