
Australia Cancer Biomarkers Market Size, Share, Trends and Forecast by Profiling Technology, Biomolecule, Cancer Type, Application, End User, and Region, 2026-2034
Australia Cancer Biomarkers Market Summary:
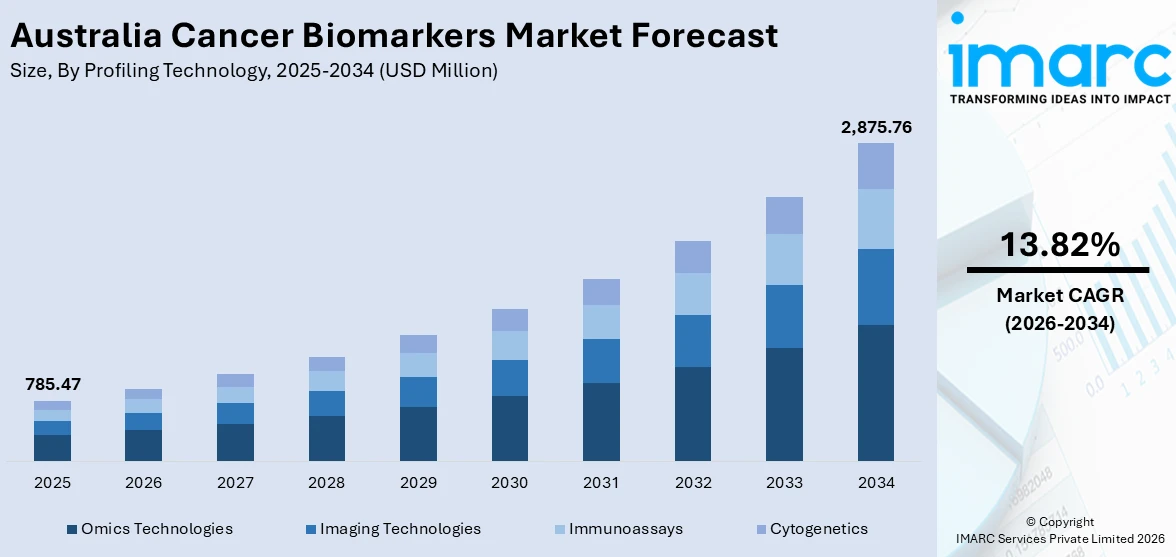
The Australia cancer biomarkers market size was valued at USD 785.47 Million in 2025 and is projected to reach USD 2,875.76 Million by 2034, growing at a compound annual growth rate of 13.82% during 2026-2034.
The market is propelled by the rising prevalence of cancer across Australia, the growing demand for precision diagnostics, and accelerating government investment in biomarker research. Advancing omics technologies, expanding applications of liquid biopsy, and increasing adoption of personalized oncology approaches continue to generate significant momentum. Collaborative initiatives between academic institutions, research foundations, and diagnostic companies further contribute to the Australia cancer biomarkers market share.
Key Takeaways and Insights:
- By Profiling Technology: Omics technologies represent the largest segment with a market share of 45.5% in 2025, underpinned by the widespread adoption of next-generation sequencing, genomic, proteomic, and metabolomic platforms that enable comprehensive multi-dimensional tumor characterization across diverse cancer types.
- By Biomolecule: Protein biomarkers dominate the market with a share of 67.0% in 2025, driven by their established clinical utility in diagnostics, broad accessibility, faster turnaround times, and lower analytical costs relative to genetic testing approaches.
- By Cancer Type: Breast cancer leads the market with a share of 24.8% in 2025, owing to its high incidence among Australian women and robust investment in companion diagnostics and targeted therapy biomarkers for this disease.
- By Application: Diagnostics represent the largest segment with a market share of 48.0% in 2025, reflecting the critical role of biomarkers in early cancer detection, patient stratification, and clinical monitoring of treatment response across Australian healthcare settings.
- By End User: Diagnostic laboratories dominate the market with a share of 42.5% in 2025, supported by advanced analytical infrastructure, specialized scientific expertise, and high-throughput testing capabilities that position them as the primary hub for cancer biomarker analysis.
- By Region: Australia Capital Territory & New South Wales lead the market with a share of 35.5% in 2025, due to the concentration of leading academic medical centers, nationally significant research institutions, and a highly developed diagnostic laboratory network.
- Key Players: The Australia cancer biomarkers market is moderately competitive, with global diagnostic companies, specialized biotech firms, and academic spin-offs collaborating across the oncology value chain to develop and commercialize advanced biomarker testing solutions.
To get more information on this market Request Sample
The cancer biomarkers market in Australia is growing due to the rising cancer burden and the increasing shift toward precision oncology within the national healthcare system. The growing number of cancer diagnoses is driving the need for advanced diagnostic approaches that support early disease identification and more accurate treatment planning. According to government data, around 169,759 new cancer cases are expected to be diagnosed in Australia in 2025, including 94,956 cases among males and 74,803 among females. Cancer risk remains substantial across the population, with approximately 43% of individuals projected to be diagnosed with cancer by the age of 85. This substantial disease burden is catalyzing the demand for diagnostic tools that can identify tumors at earlier stages and guide individualized therapy strategies. As a result, healthcare providers are increasingly adopting biomarker-based diagnostics within hospitals, research centers, and specialized laboratories. The expanding integration of molecular testing technologies into oncology care is strengthening the role of biomarkers in improving diagnosis, treatment selection, and long-term patient management across Australia.
Australia Cancer Biomarkers Market Trends:
Increasing Research Focus on Early-Onset Cancer Biomarkers
The growing concern regarding the increasing incidence of cancer among younger populations is encouraging research institutions to prioritize the discovery of biomarkers that enable early detection and improved disease monitoring. Biomarkers are increasingly studied for their ability to identify tumor activity at earlier stages and assess relapse risks, which can significantly improve treatment planning and patient outcomes. This research emphasis is expanding the clinical applications of biomarker-based diagnostics and strengthening their role in personalized oncology care. In 2025, researchers led by Professor Michael Samuel at the University of South Australia initiated a three-year research project investigating the rise of bowel cancer among individuals under 50, focusing on identifying tumor-derived biomarkers that can help predict relapse and guide personalized treatment strategies.
Advancements in Immune-Based Biomarkers Supporting Treatment Personalization
The rising adoption of immunotherapy in oncology is increasing interest in immune-based biomarkers that help predict patient responses to specific treatments. These biomarkers evaluate immune system interactions within tumors and allow clinicians to identify which patients may benefit from particular therapies. Such developments improve treatment planning by reducing ineffective therapies and guiding patients toward more appropriate clinical options. Research into immune biomarkers is therefore becoming a key driver supporting the expansion of cancer biomarker applications. In 2025, researchers at Monash University developed the BCG-R5 immune-based biomarker panel designed to identify bladder cancer patients who are less likely to respond to standard BCG immunotherapy, enabling earlier selection of alternative treatment strategies.
Standardization of Molecular Testing Enhancing Biomarker-Guided Treatment
Efforts to standardize molecular testing procedures are supporting the broader integration of cancer biomarkers into clinical practice. Standardized guidelines help ensure consistent testing methodologies, accurate interpretation of biomarker results, and improved coordination among healthcare providers. This improves diagnostic reliability and allows clinicians to select targeted therapies with greater confidence. Standardization also supports the integration of biomarker testing into national cancer screening and treatment programs. In 2025, the Royal College of Pathologists of Australasia, in collaboration with the Thoracic Oncology Group of Australasia, released the country’s first best practice recommendations for molecular testing in lung cancer, outlining multiple guidelines to standardize biomarker testing and improve treatment selection for patients.
Market Outlook 2026-2034:
The Australia cancer biomarkers market is projected to demonstrate strong revenue growth due to increasing cancer prevalence, rising demand for early disease detection, and expanding adoption of precision medicine. The market generated a revenue of USD 785.47 Million in 2025 and is projected to reach a revenue of USD 2,875.76 Million by 2034, growing at a compound annual growth rate of 13.82% from 2026-2034. As liquid biopsy, omics technologies, and AI-powered platforms become more accessible and clinically validated, market participants across diagnostics, pharmaceuticals, and healthcare delivery will benefit from expanding opportunities throughout the forecast horizon.
Australia Cancer Biomarkers Market Report Segmentation:
| Segment Category | Leading Segment | Market Share |
|---|---|---|
|
Profiling Technology |
Omics Technologies |
45.5% |
|
Biomolecule |
Protein Biomarkers |
67.0% |
|
Cancer Type |
Breast Cancer |
24.8% |
|
Application |
Diagnostics |
48.0% |
|
End User |
Diagnostic Laboratories |
42.5% |
|
Region |
Australia Capital Territory & New South Wales |
35.5% |
Profiling Technology Insights:
- Omics Technologies
- Imaging Technologies
- Immunoassays
- Cytogenetics
Omics technologies dominate with a market share of 45.5% of the total Australia cancer biomarkers market in 2025.
Omics technologies lead the market due to their ability to analyze complex biological data at the genomic, proteomic, and metabolomic levels. These technologies enable researchers and clinicians to identify specific molecular alterations associated with different cancer types. By examining large-scale biological datasets, omics platforms help uncover disease mechanisms and support the development of precise diagnostic and therapeutic strategies. The growing adoption of personalized medicine and targeted cancer therapies is increasing reliance on omics-based profiling tools for accurate disease characterization and improved treatment planning.
The dominance of the segment is because of continuous advancements in high-throughput sequencing, bioinformatics, and molecular analysis technologies. These innovations allow healthcare professionals and researchers to process large volumes of genetic and molecular information with greater speed and accuracy. Omics technologies play a crucial role in biomarker discovery, enabling the identification of novel molecular indicators that improve cancer detection and prognosis. Additionally, increasing research collaborations, expanding clinical studies, and greater investment in precision oncology initiatives are strengthening the adoption of omics-based profiling technologies across Australia.
Biomolecule Insights:
- Genetic Biomarkers
- Protein Biomarkers
- Glyco-Biomarkers
Protein biomarkers lead with a market share of 67.0% of the total Australia cancer biomarkers market in 2025.
Protein biomarkers account for the majority of the market share because of their extensive use in cancer detection, monitoring, and treatment evaluation. These biomarkers are widely utilized in clinical diagnostics because proteins released by tumor cells or affected tissues can be measured in blood or other biological samples. Their ability to provide reliable indications of disease presence and progression makes them essential in routine cancer screening and diagnostic procedures. Healthcare providers frequently rely on protein-based tests due to their established clinical validation and compatibility with widely used laboratory techniques.
The segment’s dominance is also supported by continuous advancements in proteomics research and diagnostic technologies. Improved analytical methods have enhanced the sensitivity and accuracy of protein biomarker detection, enabling earlier identification of cancer-related biological changes. Protein biomarkers are commonly used to track treatment response and monitor disease recurrence, making them valuable tools in long-term cancer management. In addition, ongoing research initiatives and the expansion of laboratory testing capabilities are encouraging the broader adoption of protein biomarker-based diagnostics across healthcare facilities in Australia.
Cancer Type Insights:
- Breast Cancer
- Lung Cancer
- Colorectal Cancer
- Prostate Cancer
- Stomach
- Others
Breast cancer exhibits a clear dominance with a 24.8% share of the total Australia cancer biomarkers market in 2025.
Breast cancer dominates the market driven by the high demand for advanced diagnostic and monitoring tools used in early detection and treatment planning. Biomarkers play a critical role in identifying specific molecular characteristics associated with breast cancer, allowing healthcare professionals to classify tumor types and determine appropriate therapies. The increasing use of biomarker testing helps clinicians detect disease at earlier stages and guide personalized treatment strategies. The growing awareness about regular cancer screening and the importance of early diagnosis is further encouraging the adoption of biomarker-based tests for breast cancer management.
The segment’s dominance is also reinforced by significant advancements in molecular diagnostics and targeted therapies used in breast cancer treatment. Biomarker tests assist in identifying hormone receptor status and other genetic indicators that influence treatment response. This enables oncologists to select more effective therapies tailored to individual patients. In addition, ongoing research efforts, improved laboratory technologies, and expanding clinical testing capabilities are strengthening the role of biomarker-based diagnostics in breast cancer care, contributing to greater adoption of these tests across healthcare facilities in Australia.
Application Insights:
Access the comprehensive market breakdown Request Sample
- Diagnostics
- Prognostics
- Risk Assessment
- Drug Discovery and Development
- Others
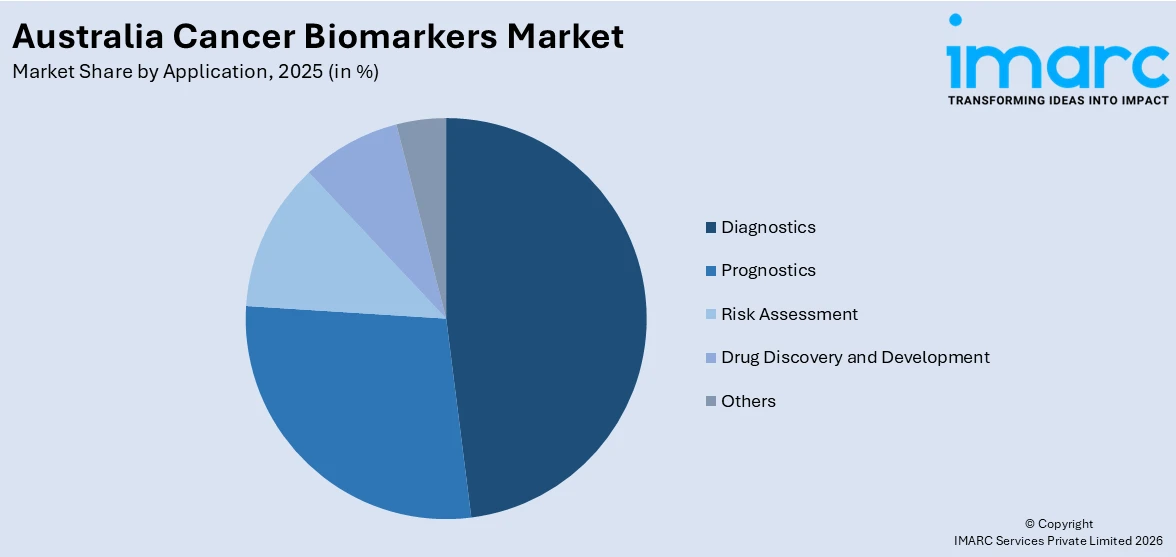
Diagnostics dominates with a market share of 48.0% of the total Australia cancer biomarkers market in 2025.
Diagnostics represent the largest segment attributed to the growing need for early cancer detection and accurate disease identification. Biomarker-based diagnostic tests enable healthcare professionals to detect cancer at earlier stages by identifying specific molecular or genetic indicators in blood, tissue, or other biological samples. Early diagnosis significantly improves treatment planning and patient outcomes, which is increasing the use of biomarker testing across hospitals and diagnostic laboratories. The rising focus on precision medicine is further encouraging the adoption of advanced diagnostic technologies that support targeted cancer detection and personalized treatment approaches.
The segment’s dominance is also driven by the growing integration of molecular diagnostics and genomic testing in routine clinical practice. Healthcare providers are increasingly relying on biomarker-based diagnostic tools to identify cancer subtypes, monitor disease progression, and guide therapy selection. Continuous advancements in laboratory technologies, including next-generation sequencing and immunoassay techniques, are improving the accuracy and efficiency of cancer diagnostics. Additionally, the rising number of cancer screening initiatives and increased awareness about early detection are further strengthening the demand for biomarker-based diagnostic applications across Australia.
End User Insights:
- Hospitals
- Academic and Research Institutions
- Ambulatory Surgical Centers
- Diagnostic Laboratories
- Others
Diagnostic laboratories lead with a market share of 42.5% of the total Australia cancer biomarkers market in 2025.
Diagnostic laboratories hold the biggest market share owing to their central role in conducting specialized testing required for cancer detection, monitoring, and treatment planning. These laboratories are equipped with advanced molecular diagnostic technologies capable of analyzing genetic, proteomic, and biochemical markers associated with various cancer types. Healthcare providers rely heavily on diagnostic laboratories for accurate biomarker analysis that supports early diagnosis and personalized treatment decisions. The increasing adoption of precision medicine and targeted therapies is further strengthening the demand for comprehensive biomarker testing services offered by diagnostic laboratories.
The dominance of diagnostic laboratories is also supported by their ability to process large volumes of clinical samples efficiently while maintaining high testing accuracy and standardized procedures. Many laboratories operate with sophisticated automated systems and advanced sequencing technologies that enhance testing speed and reliability. Additionally, hospitals and oncology centers often collaborate with specialized diagnostic laboratories to access advanced biomarker testing capabilities that may not be available in-house. The growing number of cancer screening programs and rising demand for early disease detection are further increasing the utilization of diagnostic laboratories across Australia.
Regional Insights:
- Australia Capital Territory & New South Wales
- Victoria & Tasmania
- Queensland
- Northern Territory & Southern Australia
- Western Australia
Australia Capital Territory & New South Wales exhibits a clear dominance with a 35.5% share of the total Australia cancer biomarkers market in 2025.
Australia Capital Territory & New South Wales lead the market due to the strong concentration of advanced healthcare infrastructure, major research institutions, and specialized cancer treatment centers. These regions host several leading hospitals, diagnostic laboratories, and medical research organizations that actively develop and adopt biomarker-based diagnostic technologies. The growing focus on precision medicine and early cancer detection is further increasing the demand for biomarker testing across clinical settings. In addition, strong collaboration between academic institutions, biotechnology companies, and healthcare providers is supporting continuous research and innovation in oncology diagnostics.
The region’s leadership is also influenced by significant investments in medical research, expanding clinical trial activities, and increasing adoption of genomic testing in cancer management. Healthcare providers in Australia Capital Territory and New South Wales are actively integrating biomarker-based screening and diagnostic approaches to improve treatment outcomes and enable personalized therapies. The presence of a large patient population and well-established healthcare networks also contribute to higher testing volumes. Furthermore, government-supported healthcare programs and cancer research initiatives are encouraging the adoption of advanced diagnostic technologies, strengthening the market growth across these regions.
Market Dynamics:
Growth Drivers:
Why is the Australia Cancer Biomarkers Market Growing?
Expansion of National Cancer Data Systems Supporting Biomarker Development
The establishment of integrated national cancer data systems is strengthening biomarker research and clinical implementation. Unified data frameworks allow healthcare providers, research institutions, and policy organizations to collect standardized clinical information that can be used to identify disease patterns and evaluate biomarker performance across large populations. Improved data availability supports biomarker validation studies, accelerates research collaboration, and enhances evidence-based decision making in oncology. In 2025, the Australian government introduced the National Cancer Data Framework, which aimed to create a coordinated national system for collecting and using cancer-related data across health services, cancer registries, and screening programs to improve research, clinical care, and policy planning.
Government Funding Supporting Biomarker Research and Biobank Development
The growing government investment in oncology research is accelerating the development of cancer biomarkers across Australia. Public funding programs are supporting universities and research institutions in conducting large-scale studies focused on discovering and validating biomarkers that can improve cancer detection and monitoring. These initiatives also help maintain biobanks and clinical data resources that are essential for biomarker validation and translational research. Increased financial support encourages collaboration between healthcare providers, laboratories, and academic institutions, strengthening the national research ecosystem. In June 2025, Edith Cowan University’s Cancer Blood Biomarkers Study received $205,000 from the Western Australia Government’s Future Health Research and Innovation Fund to advance research led by Professor Elin Gray on blood-based biomarkers for cancers including melanoma, lung, and ovarian cancer.
Rise of Biomarker-Guided Clinical Trials in Precision Oncology
The increasing use of biomarker-guided clinical trials is bolstering the growth of the cancer biomarkers market in Australia by promoting the development of targeted treatment strategies based on molecular characteristics rather than cancer type alone. This research approach enables clinicians to identify patient groups that may respond to specific therapies by analyzing genetic or protein-based biomarkers associated with tumor behavior. Biomarker-driven trials are expanding treatment possibilities for cancers that show resistance to conventional therapies and are strengthening the role of molecular diagnostics in oncology research. In 2025, researchers led by the Australian National University initiated a clinical trial evaluating the experimental drug PMR-116 using a biomarker-based “basket” trial design, targeting cancers driven by the MYC protein across multiple cancer types including prostate, breast, ovarian, and certain blood cancers.
Market Restraints:
What Challenges the Australia Cancer Biomarkers Market is Facing?
High Cost of Advanced Biomarker Testing Limiting Broad Adoption
The high cost associated with sophisticated biomarker platforms such as next-generation sequencing, comprehensive proteomics, and advanced liquid biopsy assays presents a significant barrier to widespread adoption across Australia's public healthcare system. Many advanced cancer biomarker tests remain outside the scope of government reimbursement frameworks, requiring patients or institutions to fund testing privately. This cost burden disproportionately affects regional and rural healthcare facilities where budgetary constraints are more acute, limiting equitable access to precision oncology diagnostics.
Regulatory and Reimbursement Challenges Slowing Commercialization of Novel Biomarkers
The pathway to regulatory approval and reimbursement listing for novel cancer biomarkers in Australia is complex and time-intensive. New diagnostic biomarkers must demonstrate clinical utility, analytical validity, and cost-effectiveness to satisfy the requirements of Australia's Therapeutic Goods Administration and Medical Services Advisory Committee. These demanding regulatory hurdles and extended timelines between biomarker discovery and market entry constrain the pace of commercial adoption, particularly for emerging technologies such as multi-cancer early detection platforms and novel liquid biopsy assays with broader screening applications.
Shortage of Specialized Oncology Diagnostics Workforce in Regional Areas
The effective deployment of advanced cancer biomarker testing across Australia is constrained by a shortage of specialized scientific and medical personnel capable of performing and interpreting complex molecular diagnostic assays. This challenge is particularly pronounced in regional and remote areas where access to accredited diagnostic laboratories and oncology specialists is limited. The geographic concentration of expertise and advanced testing infrastructure in major metropolitan centers creates persistent inequities in patient access to biomarker-guided cancer care across the country.
Competitive Landscape:
The Australia cancer biomarkers market exhibits moderate competitive intensity, characterized by the presence of established multinational diagnostic companies alongside innovative domestic biotechnology firms and academic spin-offs. Global leaders leverage advanced sequencing platforms, immunohistochemistry solutions, and companion diagnostic portfolios to serve Australia's oncology ecosystem, while domestic entities contribute through translational proteomics, liquid biopsy development, and specialized molecular testing services. Competition is increasingly shaped by the integration of AI-powered analytics, multi-omics capabilities, and strategic partnerships between research institutions and commercial diagnostic providers seeking to accelerate delivery of next-generation precision oncology solutions.
Recent Developments:
- September 2025: Proteomics International Laboratories announced promising results for its PromarkerEso blood test designed to detect esophageal adenocarcinoma at early stages. Presented at the ISDE World Congress in Brisbane, the test demonstrated high accuracy, with sensitivity of up to 91% for stage two cancers and 81% for stage one. The minimally invasive diagnostic tool combined biomarker analysis with patient risk factors to support earlier detection and improve survival outcomes.
- July 2025: Researchers at UNSW Sydney introduced an AI-powered blood test for breast cancer that detects tumor biomarkers through a non-invasive method. Developed with biotech company BCAL Diagnostics, the test was already being used in clinics and screening centers in Sydney and Melbourne. The technology analyzed molecular signals in blood to identify cancer earlier and may also support monitoring of treatment response and disease progression.
Australia Cancer Biomarkers Market Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Million USD |
| Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Profiling Technologies Covered | Omics Technologies, Imaging Technologies, Immunoassays, Cytogenetics |
| Biomolecules Covered | Genetic Biomarkers, Protein Biomarkers, Glyco-Biomarkers |
| Cancer Types Covered | Breast Cancer, Lung Cancer, Colorectal Cancer, Prostate Cancer, Stomach, Others |
| Applications Covered | Diagnostics, Prognostics, Risk Assessment, Drug Discovery and Development, Others |
| End Users Covered | Hospitals, Academic and Research Institutions, Ambulatory Surgical Centers, Diagnostic Laboratories, Others |
| Regions Covered | Australia Capital Territory & New South Wales, Victoria & Tasmania, Queensland, Northern Territory & Southern Australia, Western Australia |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report
The Australia cancer biomarkers market size was valued at USD 785.47 Million in 2025.
The Australia cancer biomarkers market is expected to grow at a compound annual growth rate of 13.82% during 2026-2034 to reach USD 2,875.76 Million by 2034.
Omics Technologies dominated the market with approximately 45.5% revenue share in 2025, driven by widespread adoption of next-generation sequencing and multi-omics platform integration across Australian cancer diagnostics.
Key factors driving the Australia cancer biomarkers market include increasing research on early-onset cancers and the need for improved early detection tools. In 2025, the University of South Australia launched a three-year study led by Professor Michael Samuel to identify biomarkers predicting relapse in bowel cancer patients under 50.
Major challenges include high costs of advanced biomarker testing limiting broad adoption, complex regulatory and reimbursement pathways slowing commercialization, and a shortage of specialized oncology diagnostics expertise in regional and rural areas across Australia.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












