
Blood Transfusion Diagnostics Market Report by Product (Reagents and Kits, Instruments, and Others), Application (Blood Grouping, Disease Screening), End Use (Hospitals, Blood Banks, Diagnostics Laboratories, and Others), and Region 2026-2034
Market Overview:
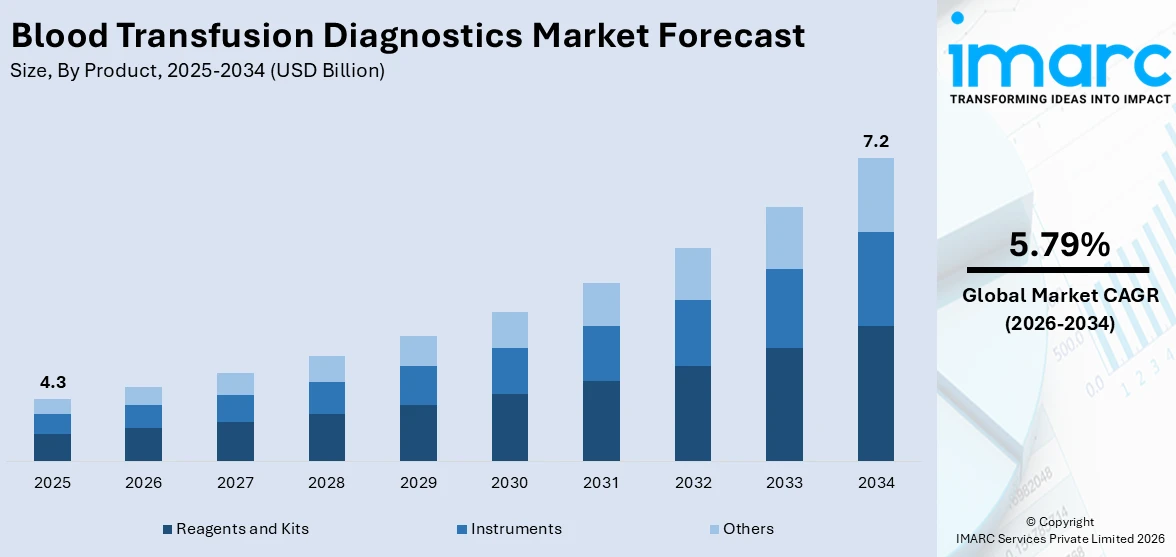
The global blood transfusion diagnostics market size reached USD 4.3 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 7.2 Billion by 2034, exhibiting a growth rate (CAGR) of 5.79% during 2026-2034. The increasing number of voluntary blood donations, the growing need for more robust, accurate, and rapid diagnostic solutions among the masses, and the rising organ transplantation procedures are some of the major factors propelling the market.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025 |
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
| Market Size in 2025 | USD 4.3 Billion |
| Market Forecast in 2034 | USD 7.2 Billion |
| Market Growth Rate 2026-2034 | 5.79% |
Blood transfusion diagnostics are crucial laboratory procedures used to screen and match blood types between donors and recipients to ensure a safe and successful transfusion. The testing includes several processes, such as identifying the four main blood types, determining positive or negative Rh factors, and testing donor and recipient blood compatibility. Diagnostics also involve screening for blood-borne pathogens including HIV, hepatitis B and C, syphilis, and other infectious agents, to prevent their transmission. Additionally, antibody screening is performed to identify any unusual antibodies in the recipient's blood that might react adversely to the transfused blood. These diagnostics are of paramount importance in managing the safety and efficacy of blood transfusions, reducing potential complications, and enabling personalized medicine by considering individual immunological and genetic differences in patients.
To get more information on this market Request Sample
The global increase in voluntary blood donations has been a crucial factor driving the blood transfusion diagnostics market. As societies become more aware of the importance of blood donations in saving lives, the number of blood donations has increased. This necessitates the need for more robust, accurate, and rapid diagnostic solutions to ensure the safety and compatibility of donated blood, thereby influencing the market. Additionally, the rising rate of organ transplantation procedures, which often require blood transfusions, leads to an increased demand for blood transfusion diagnostics. Also, emerging infectious diseases and newly identified pathogens are propelling the demand for more sophisticated blood transfusion diagnostics. Pathogens, such as SARS-CoV-2, which caused the COVID-19 pandemic, require additional screening of donated blood to ensure safety. This trend drives innovation and market growth as diagnostic manufacturers continually evolve to meet the demands of changing health landscapes.
Blood Transfusion Diagnostics Market Trends/Drivers:
Increasing Prevalence of Blood-Related Disorders and Diseases
The increasing prevalence of blood-related disorders, including hemophilia, anemia, blood cancer, and sickle cell disease, is one of the major drivers for the blood transfusion diagnostics industry. The rise of infectious diseases, including HIV, hepatitis B and C, Zika, and West Nile virus also necessitates rigorous blood screening before transfusion to prevent transmission. Furthermore, conditions that might require blood transfusion, such as severe trauma, surgical procedures, and childbirth complications, are consistently present in the global health landscape. As the global population grows and ages, the prevalence of these conditions is expected to increase, thus propelling the demand for safe and effective blood transfusion diagnostics.
Government Regulations and Blood Safety Initiatives
Government regulations and initiatives aimed at ensuring blood safety are significantly impacting the blood transfusion diagnostics industry. Countries across the globe are imposing stringent regulations to guarantee the safety and efficacy of blood transfusion procedures. This includes mandatory screening of donated blood for infectious agents and tight control over the overall process of collection, storage, and transfusion. These regulations drive the need for high-quality diagnostic solutions that can meet regulatory standards. In addition, many governments, NGOs, and international bodies are conducting awareness campaigns about the importance of safe blood donation and transfusion. These initiatives are driving the demand for advanced diagnostic tools, thereby contributing to the growth.
Technological Advancements and Innovation
Continual advancements in technology and innovation in diagnostics systems has seen significant developments, including more sophisticated automation processes, next-generation sequencing, and molecular genotyping techniques that are increasingly accurate, fast, and less invasive. Advanced technologies are employed to enhance diagnostic accuracy, minimize errors, reduce transfusion-transmitted infections, and enhance the compatibility between the donor and recipient. For instance, the advent of polymerase chain reaction (PCR) techniques and gel card agglutination methods have considerably improved the blood screening process. The evolution of digital health, AI, machine learning, and blockchain technologies are also propelling the integration of automated data analysis in blood transfusion diagnostics, improving accuracy, efficiency, and traceability.
Blood Transfusion Diagnostics Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global blood transfusion diagnostics market report, along with forecasts at the global, regional, and country levels from 2026-2034. Our report has categorized the market based on product, application, and end use.
Breakup by Product:
- Reagents and Kits
- Instruments
- Others
Reagents and kits dominates the market
The report has provided a detailed breakup and analysis of the blood transfusion diagnostics market based on the product. This includes reagents and kits, instruments, and others. According to the report, reagents and kits represented the largest segment.
The blood transfusion diagnostics industry is driven by the increasing number of blood transfusions performed across the globe. With a growing population and advancements in medical procedures, the demand for blood products and the need for accurate and efficient diagnostics have escalated. Additionally, the rising prevalence of blood disorders and infectious diseases, such as HIV, hepatitis, and malaria, has further propelled the demand for reagents and kits as these conditions require thorough screening and testing of donated blood. Furthermore, technological advancements in the field of transfusion medicine, including the development of innovative reagents and kits that offer improved sensitivity, specificity, and rapid turnaround time, have also played a crucial role in driving the market forward.
On the contrary, instruments play a vital role in the blood transfusion diagnostics industry, and their demand is driven by the rising volume of blood transfusions across the globe. This has necessitated the need for efficient and automated instruments to handle the growing workload. These instruments streamline the workflow, reduce human error, and enhance productivity in blood banks and transfusion centers. Additionally, the rising awareness and implementation of stringent regulatory guidelines regarding blood safety have stimulated the demand for advanced instruments. These instruments enable accurate and reliable testing, screening, and compatibility matching of blood products, ensuring the safety of transfusions and minimizing the risk of adverse reactions.
Breakup by Application:
Access the comprehensive market breakdown Request Sample
- Blood Grouping
- Disease Screening
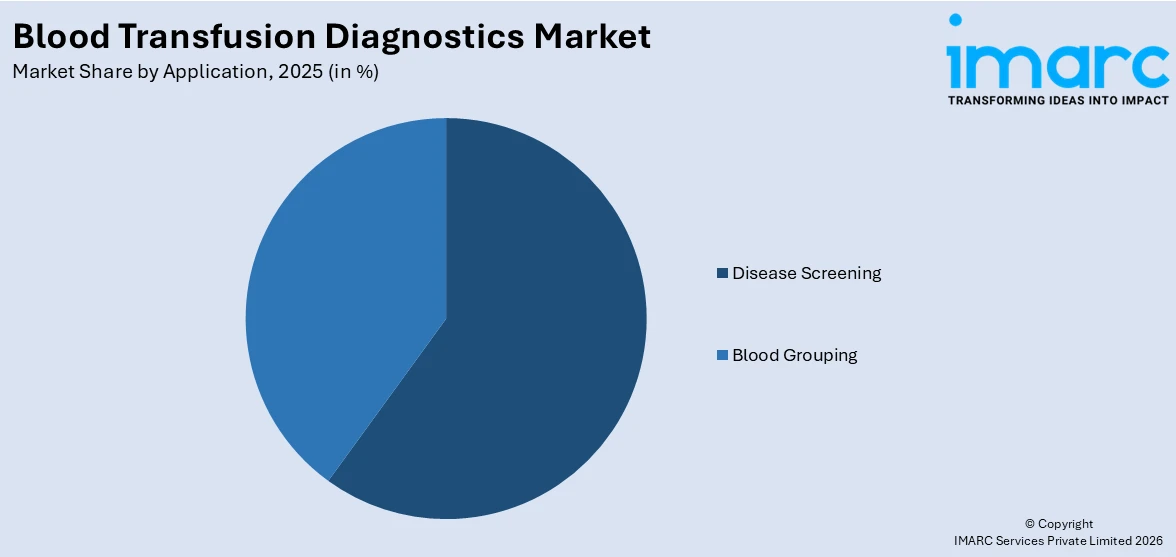
Disease screening hold the largest share in the market
A detailed breakup and analysis of the blood transfusion diagnostics market based on the application has also been provided in the report. This includes blood grouping and disease screening. According to the report, disease screening accounted for the largest market share.
the blood transfusion diagnostics industry is witnessing a growing demand for disease screening applications due to a heightened emphasis on patient safety and the prevention of transfusion-transmitted infections. As a result, regulatory bodies and healthcare organizations are increasingly focusing on implementing stringent screening protocols to detect diseases such as HIV, hepatitis B and C, syphilis, and others. In addition, advancements in technology have led to the development of more efficient and accurate screening methods, such as nucleic acid amplification techniques and serological assays. These innovative tools enable rapid and sensitive detection of infectious agents, enhancing the overall safety of blood transfusion procedures.
On the other hand, the blood grouping application in the blood transfusion diagnostics industry is driven by the growing demand for blood transfusions. The rapid population growth, aging demographics, and advancements in medical procedures have fueled the need for accurate and reliable blood grouping tests. Blood grouping plays a critical role in determining blood compatibility between donors and recipients, ensuring safe and successful transfusions. Along with this, the rising prevalence of blood disorders, such as anemia, hemophilia, and sickle cell disease, has further propelled the demand for blood grouping tests. These tests help identify specific blood types, including A, B, AB, and O, as well as Rh factors, enabling healthcare professionals to select compatible blood products for patients in need. Additionally, the implementation of strict regulatory guidelines and quality standards regarding blood safety has significantly contributed to the market drivers for blood grouping applications.
Breakup by End Use:
- Hospitals
- Blood Banks
- Diagnostics Laboratories
- Others
Blood banks dominates the market
The report has provided a detailed breakup and analysis of the blood transfusion diagnostics market based on the end use. This includes hospitals, blood banks, diagnostics laboratories, and others. According to the report, blood bank represented the largest segment.
The blood transfusion diagnostics industry is experiencing significant market drivers for blood banks' end use. The growing demand for blood and blood products in various medical procedures, including surgeries, trauma cases, and chronic disease management, is driving the need for well-equipped blood banks. Blood banks play a crucial role in ensuring an adequate and safe supply of blood by collecting, storing, and distributing blood products. Additionally, advancements in technology have enabled more efficient and accurate blood typing and compatibility testing, ensuring appropriate matching between donors and recipients. This has led to increased confidence in the transfusion process and improved patient outcomes.
On the other hand, hospitals are the primary recipients of blood transfusion products, as they require a steady supply to meet the needs of patients undergoing surgeries, trauma cases, and various medical treatments. The demand for blood transfusions is growing due to factors such as a growing aging population, the prevalence of chronic diseases, and advancements in medical procedures that require blood support. In addition, hospitals prioritize patient safety, and accurate blood typing and cross-matching are crucial to prevent transfusion reactions and ensure successful outcomes. This drives the adoption of advanced blood transfusion diagnostics technologies that offer reliable and rapid results.
Breakup by Region:
- North America
- United States
- Canada
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Others
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America exhibits a clear dominance, accounting for the largest the blood transfusion diagnostics market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (Germany, France, the United Kingdom, Italy, Spain, and others); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa.
The blood transfusion diagnostics industry in North America is driven by a well-established healthcare infrastructure, with a high demand for blood transfusion products across various medical procedures and treatments. The increasing prevalence of chronic diseases, aging population, and advancements in medical practices contribute to the growing need for blood transfusions in North America. Additionally, stringent regulatory frameworks and guidelines set by organizations ensure the safety and quality of blood transfusion diagnostics. Compliance with these regulations drives the adoption of advanced screening technologies and comprehensive testing protocols, creating a favorable market environment for blood transfusion diagnostics companies.
On the contrary, Asia Pacific is estimated to expand further in this domain due to the region's large population and growing healthcare expenditure. In confluence with this, the rising awareness about the importance of blood donation and the implementation of initiatives to encourage voluntary donation drive the need for efficient blood transfusion diagnostics. Moreover, the rapid economic development and urbanization in countries, including China and India are leading to improved healthcare infrastructure.
Competitive Landscape:
The global blood transfusion diagnostics market is experiencing significant growth due to the escalating investments in research and development (R&D) activities to develop innovative technologies and products. They focus on improving screening methods, enhancing accuracy and sensitivity, and reducing turnaround time for test results. Along with this, companies continually strive to advance their diagnostic technologies. This includes the development of automated systems, high-throughput platforms, and point-of-care testing devices that offer rapid and reliable results. Integration with digital solutions and data management systems is also a focus area to streamline workflows and enhance efficiency. In addition, Blood transfusion diagnostics companies maintain rigorous quality control measures and adhere to regulatory guidelines to ensure the safety and efficacy of their products. They invest in robust quality management systems and undergo regular inspections and certifications to meet industry standards and maintain compliance with regulatory authorities. Moreover, Companies partner with blood banks, hospitals, and diagnostic laboratories to conduct clinical trials, validate their technologies, and expand their market presence.
The report has provided a comprehensive analysis of the competitive landscape in the global blood transfusion diagnostics market. Detailed profiles of all major companies have also been provided. Some of the key players in the market include:
- Abbott Laboratories
- BioMérieux
- Bio-Rad Laboratories Inc.
- Danaher Corporation
- Diagast
- Grifols S.A
- Merck KGaA
- Ortho Clinical Diagnostics
- Quotient Limited.
- Werfen S.A.
Blood Transfusion Diagnostics Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical and Forecast Trends, Industry Catalysts and Challenges, Segment-Wise Historical and Predictive Market Assessment:
|
| Products Covered | Reagents and Kits, Instruments, Others |
| Applications Covered | Blood Grouping, Disease Screening |
| End Uses Covered | Hospitals, Blood Banks, Diagnostics Laboratories, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Abbott Laboratories, BioMérieux, Bio-Rad Laboratories Inc., Danaher Corporation, Diagast, Grifols S.A, Merck KGaA, Ortho Clinical Diagnostics, Quotient Limited., Werfen S.A., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the blood transfusion diagnostics market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global blood transfusion diagnostics market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assist stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the blood transfusion diagnostics industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Frequently Asked Questions About the Blood Transfusion Diagnostics Market Report
The global blood transfusion diagnostics market reached a value of USD 4.3 Billion in 2025.
The market is projected to grow at a CAGR of 5.79% during 2026-2034, reaching USD 7.2 Billion by 2034.
Key growth drivers include voluntary blood donation increase, organ transplantation procedure rise, infectious disease screening mandates, blood safety standards, and emerging pathogen detection requirements.
The report covers segmentation by product, application, end use, and region. Each segment includes detailed market size and forecast analysis.
Major players in the global blood transfusion diagnostics market include Abbott Laboratories, BioMérieux, Bio-Rad Laboratories Inc., Danaher Corporation, Diagast, Grifols S.A., Merck KGaA, Ortho Clinical Diagnostics, Quotient Limited, Werfen S.A., etc.
Key trends include PCR-based nucleic acid screening, gel card agglutination adoption, AI-driven diagnostic automation, digital health integration, and molecular genotyping advancement.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












