
Blood Warmer Devices Market Size, Share, Trends and Forecast by Product, Type, End Use, and Region, 2025-2033
Blood Warmer Devices Market Size and Share:
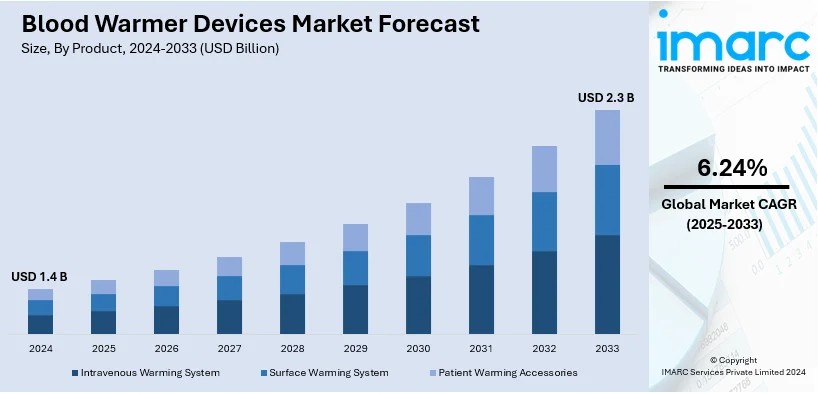
The global blood warmer devices market size was valued at USD 1.4 Billion in 2024. Looking forward, IMARC Group estimates the market to reach USD 2.3 Billion by 2033, exhibiting a CAGR of 6.24% from 2025-2033. North America currently dominates the market, holding a market share of over 40.7% in 2024. The blood warmer devices market is witnessing growth due to increasing surgical procedures, rising trauma cases, growing awareness about hypothermia management, expanding healthcare infrastructure, and ongoing advancements in portable and automated technologies.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2024 |
|
Forecast Years
|
2025-2033 |
|
Historical Years
|
2019-2024
|
| Market Size in 2024 | USD 1.4 Billion |
| Market Forecast in 2033 | USD 2.3 Billion |
| Market Growth Rate (2025-2033) | 6.24% |
This market is driven by the increased incidence of complications due to hypothermia during surgical procedures and trauma care. Such devices are necessary for keeping the patient's body at the right temperature when blood transfusion is involved, thus helping to minimize risks associated with cold blood infusion, including cardiac arrest or immune suppression. Advances in surgical procedures in the globe, particularly high-risk surgeries involving patients that are very old or are critically ill, have also increased the demand for improved blood warming solutions. New technologies being developed for portable and automated blood warmers are becoming widely adopted across hospitals, ambulances, and military medical units. This rise in healthcare provider awareness toward temperature management of transfusions and stringent regulatory policies on the basis of patient safety are supporting market growth. Furthermore, growth of healthcare infrastructure in emerging economies and investments in medical technology are showing steady growth for the market.
The United States has emerged as a key regional market for blood warmer devices. This is due to the increased incidence of surgical procedures and trauma cases that require quick blood transfusions. The blood warmer devices are essential in keeping the blood at optimal temperatures during transfusion, hence safe for the patient and prevent complications such as hypothermia. The increasing chronic conditions and cancer treatments, which involve frequent transfusions, support the demand. Advancements in device technology, such as easy-to-use and portable warmers, are also driving market demand in hospitals and emergency medical services. Regulatory approvals for such new devices, along with the increasing awareness about the cruciality of maintaining thermal management in transfusion protocols, is also driving the markets forward. The growing proportion of the elderly population, requiring surgical and critical care interventions, is also significantly growing demand for blood warmer devices in the United States.
Blood Warmer Devices Market Trends:
Increasing infusion therapies
On account of the rising number of coronavirus disease (COVID-19) cases, the need for infusion therapies is increasing. This represents one of the major factors escalating the demand for blood warmer devices to bring fluids to body temperature, encourage faster patient recovery, and lower the risk of transfusion shock. Moreover, the excessive alcohol and recreational drugs consumption and rising incidence of diabetes, arthritis, Parkinson's disease, and spinal cord injuries increase the risk of developing hypothermia. According to IDF Diabetes Atlas, people affected with diabetes (1,000s) in 2021 in China includes 140,869.6. This, along with the rising rates of surgical operations and procedures and the aging population, is contributing to the market growth. According to Eurostat, in 2019, there were 90.4 Million older people (aged 65 years or more) living in the EU-27. Additionally, these devices are required in exchange transfusion among infants with cold agglutinin disease. The emerging trend of private medical practice is driving the market further. Apart from this, governing agencies of several countries are expanding their budgets on defense health systems, which is projected to strengthen the market growth.
Rising number of surgical procedures
Growing surgical procedures and trauma cases is one of the prominent reasons in favor of the blood warmer devices market. Blood and fluid need to be maintained at an appropriate temperature during the time of transfusion. In an emergency or surgical scenario, hypothermia prevention becomes the most important aspect in a patient's care. Such requirements make blood warmers all the more indispensable. Moreover, in a trauma-related emergency, a faster blood transfusion may also be needed, thus heightening the demand for these reliable and portable blood warmers in hospitals and ambulances.
Increasing incidences of chronic diseases and cancer
The rising prevalence of chronic conditions, fueled by aging populations and lifestyle-related factors, is driving demand for advanced care solutions. As per MayoClinic, the estimated cost of chronic disease is predicted to reach USD 47 Trillion by the year 2030. Chronic diseases and cancer treatments often necessitate regular blood transfusions, making blood warmer devices an essential component in patient care. Conditions such as anemia, kidney disorders, and cancer-related therapies like chemotherapy frequently involve transfusions to replenish blood components or maintain critical fluid levels. During these procedures, warming blood and fluids is crucial to prevent complications like hypothermia, which can exacerbate patient discomfort and compromise recovery. Patients undergoing chemotherapy or dialysis are particularly vulnerable, as these treatments can weaken their immune systems and lower body temperatures.
Blood Warmer Devices Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global blood warmer devices market, along with forecast at the global, regional, and country levels from 2025-2033. The market has been categorized based on product, type, and end use.
Analysis by Product:
- Intravenous Warming System
- Surface Warming System
- Patient Warming Accessories
Intravenous warming systems stand as the largest component in 2024, holding around 47.1% of the market. Intravenous warming systems dominate the market due to their critical role in preventing hypothermia during fluid administration, particularly in emergency and surgical settings. Meanwhile, surface warming systems are increasingly utilized in post-operative and intensive care units, offering external temperature regulation to maintain normothermia. Additionally, the demand for patient warming accessories, including warming blankets and disposable covers, has been rising as they complement the primary devices and enhance operational efficiency in various healthcare scenarios. This segmentation highlights the diverse applications and growing adoption of blood warming technologies across medical environments.
Analysis by Type:
- Portable
- Non-Portable
Non-portable leads the market with around 55.5% of market share in 2024. Non-portable blood warmers are predominantly utilized in hospitals, clinics, and specialized care units, where stationary equipment ensures uninterrupted operations and integration with existing healthcare infrastructure. The choice between portable and non-portable devices is influenced by factors such as the care environment, patient volume, and technological advancements, contributing to the evolving dynamics of the blood warmer devices market.
Portable blood warmers are gaining traction due to their versatility, ease of use, and application in emergency and field settings, such as ambulances, military operations, and disaster management scenarios. Their compact design and ability to provide efficient temperature control make them ideal for mobile applications, where quick and effective blood warming is critical.
Analysis by End Use:
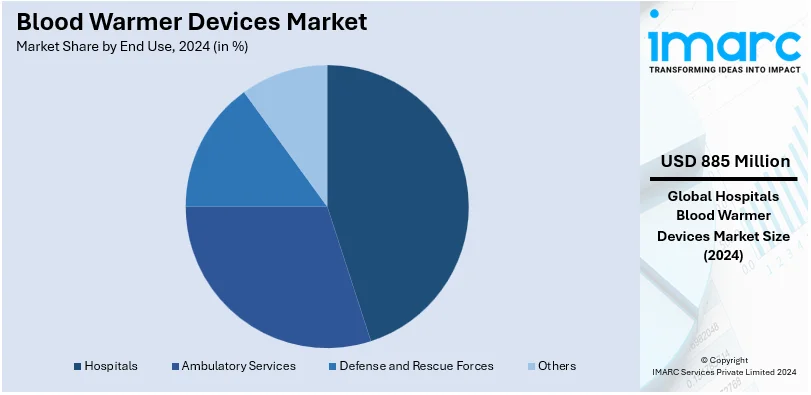
- Hospitals
- Ambulatory Services
- Defense and Rescue Forces
- Others
Hospitals leads the market with around 65.4% of market share in 2024. Hospitals represent the largest end-use segment, driven by the widespread use of blood warmers in surgeries, trauma care, and transfusion procedures to prevent hypothermia. The ambulatory services sector follows closely, where blood warmer devices are essential for emergency medical services, ensuring patient stability during transportation. Defense and rescue forces also constitute a significant segment, as these devices are crucial in military and disaster-response settings for stabilizing injured personnel in extreme environments. Moreover, the others segment encompasses niche applications such as veterinary care and outpatient clinics, contributing to the overall demand for blood warmers in diverse scenarios. These varied applications underscore the versatility and critical importance of blood warmer devices across multiple sectors.
Regional Analysis:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
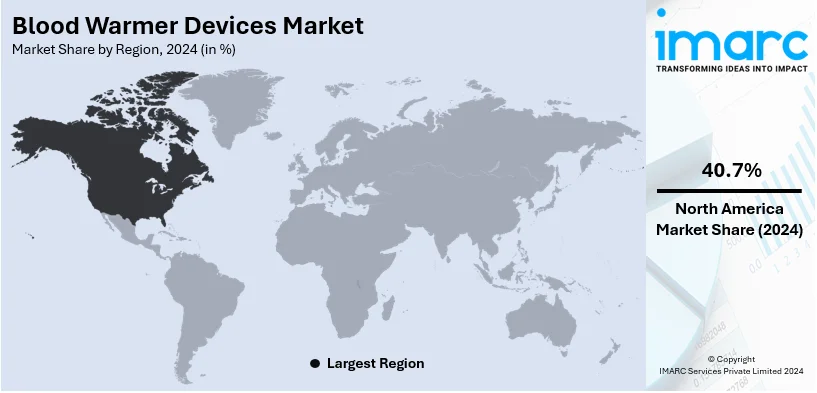
In 2024, North America accounted for the largest market share of over 40.7%. North America dominates the market due to advanced medical technologies and high adoption in emergency care and surgeries. North America has very high health care spending, ensuring that hospitals and clinics upgrade to state-of-the-art medical equipment. The well-set healthcare infrastructure in the region ensures the adoption of advanced blood warming devices as a necessity to be implemented in order to augment patient safety during surgeries and transfusions.
In the Asia-Pacific region, countries such as China, Japan, India, South Korea, Australia, and Indonesia show significant growth, driven by increasing healthcare investments, rising patient awareness, and a growing aging population. The European market, including Germany, France, the United Kingdom, Italy, Spain, Russia, and others, remains robust due to a well-established healthcare sector and widespread utilization of medical devices in clinical practices. In Latin America, countries like Brazil and Mexico, alongside others, are emerging as growth areas with improving healthcare infrastructure.
The Middle East and Africa region is steadily advancing, supported by governmental efforts to enhance healthcare systems and the adoption of medical devices in developing economies. This regional segmentation highlights the global demand and potential growth avenues for blood warmer devices.
Key Regional Takeaways:
United States Blood Warmer Devices Market Analysis
The United States accounts for the 90% of the blood warmer devices in North America. The blood warmer devices market in the United States is currently experiencing robust growth due to several specific drivers. Healthcare providers are increasingly adopting advanced blood warming technologies to address the rising number of surgical procedures, trauma cases, and chronic disease treatments requiring transfusions. According to the Centres for Disease Control and Prevention, in the United States in 2021, 1,777,566 new cancer cases were reported. Hospitals and clinics are actively investing in portable and energy-efficient blood warmers to enhance patient safety and prevent complications such as hypothermia during transfusion processes. Manufacturers are innovating continuously, focusing on developing devices with improved temperature control accuracy and rapid warming capabilities, which are gaining significant traction among healthcare professionals. Regulatory bodies are reinforcing strict compliance guidelines, encouraging the adoption of FDA-approved blood warmer devices with enhanced safety features. Concurrently, the demand for such devices is growing among military and emergency medical services due to their role in critical field operations. Technological advancements, including the integration of wireless connectivity for real-time monitoring, are also driving adoption. The increasing focus on value-based healthcare is pushing hospitals to incorporate these devices into their operational workflows to minimize transfusion-related complications and improve patient outcomes. Moreover, awareness campaigns by organizations are educating healthcare professionals about the critical benefits of blood warmers, further fuelling market growth. These factors collectively propel the blood warmer devices market in the United States.
Asia Pacific Blood Warmer Devices Market Analysis
The Asia Pacific blood warmer devices market is currently being driven by the increasing adoption of advanced healthcare technologies in developing economies, as healthcare providers are actively focusing on improving patient care and outcomes during critical surgical and emergency procedures. Hospitals and clinics across the region are upgrading their medical equipment to reduce risks associated with hypothermia, particularly during transfusions and major surgeries. Rising awareness about the benefits of maintaining normothermia is prompting medical institutions to incorporate blood warmer devices into standard transfusion protocols. Governments and private organizations are also funding initiatives to strengthen healthcare infrastructure, enabling greater adoption of these devices in rural and urban areas. Meanwhile, manufacturers are focusing on developing portable and user-friendly blood warmers to cater to the growing demand for compact devices in ambulatory and home care settings. Increasing cases of trauma and chronic conditions necessitating frequent blood transfusions, especially in countries like India and China, are further supporting the market. According to the Press Information Bureau (PIB), the prevalence of anaemia among six groups as per the National Family Health Survey (2019-21), is 57.0% in women (15-49 years), 31.1% in adolescent boys (15-19 yrs.), 59.1% in adolescent girls, 25.0% in men (15-49 years), 52.2% in pregnant women (15-49 years) and 67.1% in children (6-59 months).
Europe Blood Warmer Devices Market Analysis
The blood warmer devices market in Europe is witnessing growth due to the rising adoption of advanced healthcare technologies aimed at improving patient outcomes during transfusion and surgical procedures. Hospitals and healthcare facilities are increasingly implementing blood warming devices to mitigate risks associated with hypothermia during blood transfusions, a concern amplified by stringent European healthcare standards. The market is being driven by a growing emphasis on intraoperative patient warming techniques, which are becoming integral in enhancing surgical safety. Regulatory compliance across European countries is fostering the integration of CE-certified devices, ensuring reliability and performance, thus encouraging procurement by healthcare providers. Additionally, manufacturers are continuously innovating to offer portable and user-friendly devices that align with the increasing demand for home healthcare settings and emergency medical services. The growing prevalence of chronic conditions such as cancer and cardiovascular diseases is prompting a surge in surgical procedures, thereby driving the need for effective blood warming solutions. According to the government of UK, in 2018, there were 316,680 new cases of invasive cancer (excluding non-melanoma skin cancers) diagnosed in England. Moreover, advancements in battery-operated and disposable blood warmers are catering to the demand for infection control and operational efficiency in intensive care units and emergency departments. These factors collectively underscore the ongoing transformation in the European blood warmer devices market, catering to evolving healthcare needs.
Latin America Blood Warmer Devices Market Analysis
The blood warmer devices market in Latin America is witnessing significant growth, driven by region-specific factors that address immediate medical and healthcare needs. Hospitals and emergency care centres across the region are increasingly adopting advanced blood warming technologies to improve patient outcomes, particularly in critical care settings and trauma cases. Rising awareness about the importance of maintaining normothermia during blood transfusions and fluid administration is encouraging healthcare providers to invest in portable and efficient devices. Governments and private entities are expanding investments in healthcare infrastructure, particularly in underserved areas, to bridge gaps in critical care services. Simultaneously, the prevalence of chronic diseases and the increasing number of surgical procedures are boosting the demand for blood warmers in perioperative care. According to the National Institutes of Health, in Brazil, about one-fifth of children under five years of age are anaemic. Medical tourism in countries such as Brazil and Mexico are attracting patients from across the globe, necessitating the adoption of advanced medical technologies, including blood warming devices, to meet international standards. Furthermore, manufacturers are actively collaborating with local distributors and healthcare institutions to introduce cost-effective solutions tailored to the region's diverse economic landscape. The emphasis on enhancing patient safety protocols is also driving the integration of advanced features, such as real-time monitoring and temperature control, in blood warmer devices.
Middle East and Africa Blood Warmer Devices Market Analysis
The Middle East and Africa blood warmer devices market is witnessing growth driven by the increasing prevalence of trauma cases and surgical procedures, requiring effective temperature management solutions to prevent hypothermia in patients. Hospitals and clinics are actively integrating advanced blood warmer devices to enhance patient safety during transfusions, aligning with the region's focus on improving healthcare standards. Governments and healthcare authorities are currently investing in upgrading medical infrastructure, particularly in countries like Saudi Arabia, South Africa, and the UAE, which is boosting the adoption of innovative medical devices, including blood warmers. The market is also benefitting from ongoing collaborations between global manufacturers and regional distributors, ensuring the availability of cost-effective and portable solutions suitable for emergency care and transport settings. Simultaneously, rising awareness campaigns about the benefits of temperature-controlled transfusions are creating demand from smaller healthcare facilities. Medical tourism, especially in the Gulf Cooperation Council (GCC) countries, is driving the need for high-quality healthcare services, further fuelling the uptake of these devices. According to the Ministry of Economy, the medical tourism industry in the UAE generated AED 12.1 Billion (USD 3.29 Billion) in 2018. Additionally, non-governmental organizations (NGOs) and international health programs are supporting blood warming technology adoption in rural areas to improve emergency medical care. These factors are collectively fostering a robust and sustained growth trajectory for the blood warmer devices market in the region.
Competitive Landscape:
At present, companies are focusing on developing advanced blood warmer devices with enhanced features such as portability, rapid warming capabilities, and precise temperature control. For instance, the introduction of portable blood warmers has addressed the need for efficient warming solutions in emergency and military settings, thereby expanding the market reach. Moreover, to expand their product portfolios and enhance market presence, key players are engaging in mergers, acquisitions, and partnerships. These collaborations facilitate the sharing of technology and expertise, leading to the development of innovative products and access to new markets. Additionally, companies are expanding their operations into emerging markets with growing healthcare infrastructure to increase their customer base. By establishing distribution networks and local partnerships, they can effectively cater to the rising demand for blood warmer devices in these regions.
The report provides a comprehensive analysis of the competitive landscape in the blood warmer devices market with detailed profiles of all major companies, including:
- 3M Company
- Barkey GmbH & Co. KG
- Baxter International Inc.
- Belmont Medical Technologies
- Biegler GmbH
- EMIT Corporation
- Gentherm Incorporated
- Sarstedt AG & Co. KG
- Smith’s Medical
- The Surgical Company PTM
- Vyaire Medical Inc.
Latest News and Developments:
- June 2024: Danish company °MEQU has obtained 510(k) approval from the US Food and Drug Administration (FDA) for its portable blood and IV-fluid warming device, the °M Warmer System. Moreover, the °M Warmer System can assist military and civilian medical personnel in preventing hypothermia in patients experiencing hemorrhage by providing warmed transfusions and IV fluids at the point of injury, during transport, and in hospitals.
Blood Warmer Devices Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2024 |
| Historical Period | 2019-2024 |
| Forecast Period | 2025-2033 |
| Units | Billion USD |
| Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Products Covered | Intravenous Warming System, Surface Warming System, Patient Warming Accessories |
| Types Covered | Portable, Non-Portable |
| End Uses Covered | Hospitals, Ambulatory Services, Defense and Rescue Forces, Others |
| Region Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | 3M Company, Barkey GmbH & Co. KG, Baxter International Inc., Belmont Medical Technologies, Biegler GmbH, EMIT Corporation, Gentherm Incorporated, Sarstedt AG & Co. KG, Smith’s Medical, The Surgical Company PTM, Vyaire Medical Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the blood warmer devices market from 2019-2033.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global blood warmer devices market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's Five Forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the blood warmer devices industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Frequently Asked Questions About the Blood Warmer Devices Market Report
Blood warmer devices are medical instruments designed to warm blood or intravenous (IV) fluids before transfusion or administration to a patient. These devices ensure that the temperature of the fluid or blood matches the patient’s body temperature, typically around 37°C (98.6°F), to prevent complications like hypothermia or thermal shock during medical treatments.
The blood warmer devices market was valued at USD 1.4 Billion in 2024.
IMARC estimates the global blood warmer devices market to exhibit a CAGR of 6.24% during 2025-2033.
The blood warmer devices market is witnessing growth due to increasing surgical procedures, rising trauma cases, growing awareness about hypothermia management, expanding healthcare infrastructure, and ongoing advancements in portable and automated technologies.
In 2024, intravenous warming systems represented the largest segment due to their critical role in preventing hypothermia during fluid administration, particularly in emergency and surgical settings.
Non portable leads the market as they are predominantly utilized in hospitals, clinics, and specialized care units, where stationary equipment ensures uninterrupted operations and integration with existing healthcare infrastructure.
Hospitals are the leading segment, driven by the widespread use of blood warmers in surgeries, trauma care, and transfusion procedures.
On a regional level, the market has been classified into North America, Asia Pacific, Europe, Latin America, and Middle East and Africa, wherein North America currently dominates the global market.
Some of the major players in the global blood warmer devices market include 3M Company, Barkey GmbH & Co. KG, Baxter International Inc., Belmont Medical Technologies, Biegler GmbH, EMIT Corporation, Gentherm Incorporated, Sarstedt AG & Co. KG, Smith’s Medical, The Surgical Company PTM, Vyaire Medical Inc., etc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












