
Brazil Aesthetic Devices Market Size, Share, Trends and Forecast by Type of Device, Application, End User, and Region, 2026-2034
Brazil Aesthetic Devices Market Size & Forecast 2026-2034
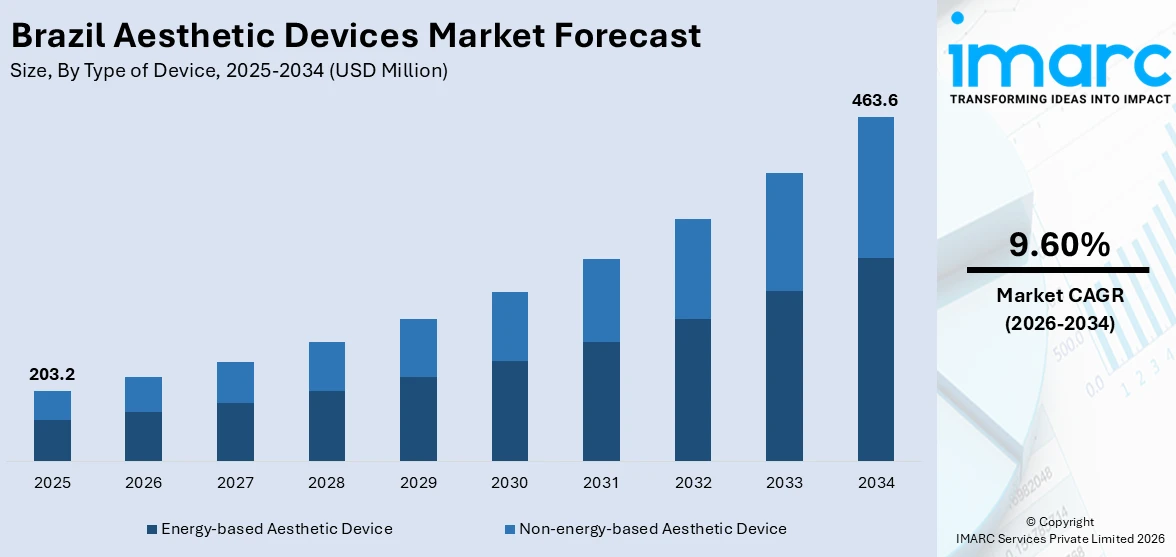
The Brazil aesthetic devices market size was valued at USD 203.24 Million in 2025, and is projected to reach USD 481.35 Million by 2034, growing at a compound annual growth rate of 9.70% from 2026-2034, driven by a confluence of rising consumer demand for minimally invasive cosmetic procedures, accelerating medical tourism activity, and continuous technological innovation in both energy-based and non-energy-based device categories. The non-energy-based segment encompassing botulinum toxin, dermal fillers, and aesthetic threads, chemical peels, microdermabrasion, and implants is propelling Brazil's aesthetic devices market share, supported by the country's vibrant beauty culture, expanding middle class, and a robust regulatory framework overseen by the Agência Nacional de Vigilância Sanitária (ANVISA).
To get more information on this market Request Sample
Brazil Aesthetic Devices Industry Analysis—Key Insights
- Non-energy-based aesthetic devices command 56.0% of the type of device share in 2025, the segment's dominance reflects the enduring consumer preference for botulinum toxin and dermal fillers, products that deliver rapid, repeatable results with minimal downtime, making them the structural backbone of Brazil's aesthetic treatment landscape.
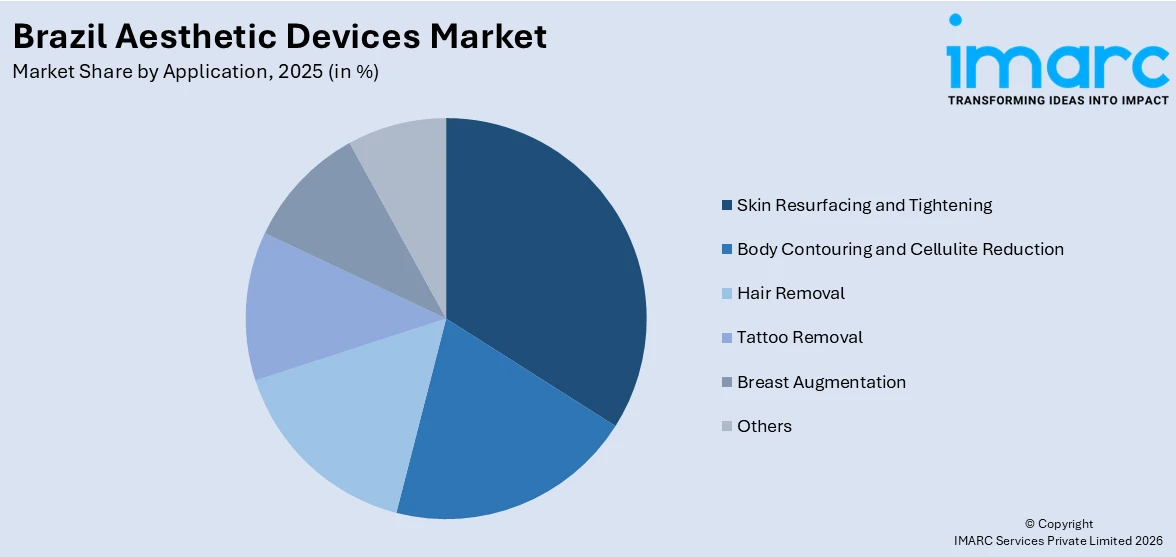
- Hair Removal leads application at 25.0% in 2025, Brazil's diverse skin tone demographics, warm climate, and deep-rooted grooming culture sustain exceptionally high demand for laser and IPL-based hair removal across clinics and aesthetic centers nationwide.
- Clinics dominate the end user segment at 48.0% in 2025, specialized aesthetic clinics offer a combination of personalized treatment protocols, competitive pricing, and practitioner expertise that positions them ahead of hospitals and home-use settings in capturing device utilization.
- Southeast leads regionally with 52.0% market share in 2025, the region's concentration of high-income urban populations in São Paulo and Rio de Janeiro, combined with a dense network of premium clinics, medical schools, and international brand partnerships, consolidates its position as Brazil's dominant aesthetic device market.
Brazil Aesthetic Devices Market Trends and Dynamics 2026
Market Trends:
Accelerating Adoption of Non-Energy-Based Injectables Across Demographics
The non-energy-based aesthetic devices segment, particularly botulinum toxin and hyaluronic acid-based dermal fillers, is experiencing heightened adoption across Brazil's broader demographic landscape. Growing awareness campaigns by dermatology associations, social media normalization of injectable aesthetics, and the affordability of procedures relative to surgical alternatives are collectively expanding the patient base. In September 2025, Hugel Inc. deepened its presence in Brazil by signing a new distribution agreement with Derma Dream for its botulinum toxin product, Letybo, enabling wider geographic reach across Brazil's urban and secondary-city markets.
Medical Tourism Driving Premium Device Deployment in Southeast Brazil
Brazil's growing status as a global medical tourism destination is directly stimulating investment in advanced aesthetic devices at clinics in São Paulo, Rio de Janeiro, and other Southeast cities. International patients benefit from treatment costs that save 50–80% comparable U.S. or European prices, incentivizing premium procedure volumes. The Brazilian Ministry of Tourism reported in September 2023 that medical tourism contributed approximately USD 1.5 billion to the economy in the preceding year, with a substantial portion attributed to cosmetic and aesthetic treatments, reinforcing the link between tourism growth and high-end device procurement.
Technological Innovation in AI-Enabled Energy-Based Platforms
Energy-based aesthetic devices in Brazil are undergoing rapid technological evolution, with manufacturers integrating artificial intelligence-enabled controls, smart sensors, and hybrid multi-modality platforms that combine radiofrequency, laser, and ultrasound functionalities in single workstations. In October 2025, CLASSYS signed an agreement to acquire 77.5% equity in JL Health, a leading South American distributor of energy-based aesthetic equipment, directly strengthening its Brazilian device deployment infrastructure to capture this growth opportunity.
- Male Consumer Growth: Men are increasingly seeking aesthetic procedures, including body contouring, hair removal, and anti-wrinkle treatments, broadening the traditional consumer demographic and creating new protocol demands for clinic operators.
- Home-Use Device Expansion: ANVISA's risk-based regulatory framework is enabling faster market entry for lower-risk consumer devices, spurring a new category of home-use aesthetic tools that complement professional clinic-based treatments.
Growth Drivers:
Rising Popularity of Minimally Invasive Cosmetic Procedures
The widespread shift away from surgical interventions toward non-invasive and minimally invasive procedures is the foundational growth engine of Brazil's aesthetic devices market. Brazilian consumers increasingly prefer treatments that deliver measurable improvements with minimal downtime, lower complication risk, and competitive pricing relative to surgical alternatives. This trend spans demographic groups from younger consumers seeking preventive anti-aging measures to older adults addressing wrinkles, volume loss, and skin laxity, ensuring broad and sustained demand across the device portfolio. Furthermore, the expanding availability of consumer financing for aesthetic treatments has lowered cost barriers, driving higher treatment frequency per patient and increasing per-clinic device utilization rates.
Expansion of Aesthetic Medical Tourism and International Patient Volumes
Brazil's combination of world-class aesthetic practitioners, ANVISA-regulated high-quality devices, and treatment costs lower than North American and European prices positions the country as a premier global destination for medical aesthetics tourism. The influx of international patients, primarily from the United States, Europe, and neighboring Latin American nations, generates consistent high-value device utilization, incentivizes clinic investment in premium multi-modality platforms, and drives professional training demand. Clinics in Southeast Brazil, particularly in São Paulo and Rio de Janeiro, are actively building multilingual service capabilities and international-standard facility accreditation to capture this opportunity.
Aging Population Creating Sustained Demand for Anti-Aging Devices
Brazil's rapid demographic aging is creating a structurally growing customer base for anti-aging aesthetic devices. According to the Brazilian Institute of Geography and Statistics (IBGE), individuals aged 65 and over constituted 15.6% of the population in 2023, a figure projected to reach 37.8% by 2070, representing one of the world's most pronounced aging trajectories. Older consumers are increasingly seeking minimally invasive options, including dermal fillers, botulinum toxin, radiofrequency skin tightening, and laser resurfacing, to address wrinkles, volume loss, and skin laxity with natural-looking outcomes. The growing alignment of aesthetic device technology with the specific skin concerns of the Fitzpatrick IV–VI skin types prevalent across Brazil's diverse population further strengthens device adoption.
Market Restraints:
Stringent Regulatory and Compliance Barriers Under ANVISA
ANVISA's rigorous approval and post-market surveillance requirements impose significant compliance costs and extended timelines on device manufacturers seeking to enter or expand within the Brazilian market. ANVISA notes that 60% of aesthetic complaints relate to safety, prompting mandatory adverse-event logging, surprise inspections, and heightened documentation standards. These requirements raise market entry barriers for smaller international players and increase operational complexity for domestic distributors, potentially slowing the pace of new technology adoption within certain device categories.
High Capital Cost of Advanced Multi-Modality Aesthetic Platforms
Premium multi-application workstations combining laser, radiofrequency, and ultrasound capabilities can exceed USD 250,000 per unit, creating substantial financial barriers for single-physician practices and smaller independent clinics. This capital burden limits device adoption in non-metropolitan markets and constrains technology refresh cycles. Insurers' increasing of malpractice premiums of clinic overhead, driven by an obligation-of-result legal standard in Brazil's civil code, further compresses clinic margins and reduces capacity for high-cost equipment investments.
Shortage of Skilled Aesthetic Practitioners in Secondary Markets
Brazil faces a structural imbalance in the geographic distribution of trained aesthetic practitioners, with the majority concentrated in Southeast urban centers. Secondary and tertiary cities, particularly in the North and Central-West regions, lack sufficient accredited practitioners capable of operating advanced energy-based platforms, limiting device deployment and clinical utilization rates. The absence of standardized national certification programs for aesthetic technicians further constrains market expansion into underserved regions despite rising consumer demand.
Brazil Aesthetic Devices Market Report Segmentation:
| Segment Category | Leading Segment | Market Share | Year |
|---|---|---|---|
| Type of Device | Non-energy-based Aesthetic Device | 56.0% | 2025 |
| Application | Hair Removal | 25.0% | 2025 |
| End User | Clinics | 48.0% | 2025 |
| Region | Southeast | 52.0% | 2025 |
Type of Device Insights
Non-energy-based Aesthetic Device – 56.0% Market Share (2025) | Leading Type of Device
Non-energy-based devices dominate the Brazilian aesthetic devices market, anchored by the extraordinary commercial resilience of botulinum toxin and dermal fillers. Botulinum toxin treatments are widely accessible and comparatively affordable in Brazil relative to other markets, enabling adoption across diverse income brackets. According to the International Society of Aesthetic Plastic Surgery, globally total number of botulinum toxin procedures reached approximately 7.8 million in 2024, and demand has continued to expand since. Hyaluronic acid-based dermal fillers represent another pillar of this segment, with ExoCoBio receiving GMP Certification for HA fillers from ANVISA in March 2023, enabling South American exports and product registration activities. Chemical peels and microdermabrasion treatments supplement clinic revenue with high-margin, low-capital procedures that require minimal practitioner specialization, while implants particularly breast implants benefit from Brazil's status as the second-largest breast augmentation market globally.
|
Segment Breakdown Non-energy-based Aesthetic Device- (Botulinum Toxin, Dermal Fillers and Aesthetic Threads, Chemical Peels, Microdermabrasion, Implants, and Others) (56.0%) · Energy-based Aesthetic Device- (Laser-based Aesthetic Device, Radiofrequency (RF)-based Aesthetic Device, Light-based Aesthetic Device, and Ultrasound Aesthetic Device) |
Application Insights
Access the comprehensive market breakdown Request Sample
Hair Removal – 25.0% Market Share (2025) | Leading Application
Hair removal is the leading application in Brazil's aesthetic devices market by revenue share, driven by a unique convergence of climatic, demographic, and cultural factors. Brazil's predominantly tropical climate creates year-round demand for hair removal treatments, eliminating the seasonal demand fluctuations seen in northern hemisphere markets. The country's highly diverse skin-tone population, with a significant proportion of Fitzpatrick IV–VI patients, has historically created challenges for traditional hair removal technologies, but newer ND: YAG laser platforms with faster repetition rates now reduce treatment chair time, improving clinic throughput and patient satisfaction. Both male and female consumers contribute to strong volume growth, with male hair removal increasingly accepted across Brazilian grooming culture. Laser-based hair removal generates high consumable revenues and repeat treatment cycle packages for clinic operators.
|
Segment Breakdown · Hair Removal (25.0%) · Skin Resurfacing and Tightening · Body Contouring and Cellulite Reduction · Tattoo Removal · Breast Augmentation · Others |
End User Insights
Clinics – 48.0% Market Share (2025) | Leading End User
Aesthetic clinics represent the primary revenue engine within the end-user segmentation, operating as the principal point of care for the vast majority of non-surgical aesthetic procedures in Brazil. Their structural advantage over hospital-based aesthetic units lies in focused operational models dedicated procedure rooms, specialized staff, flexible appointment schedules, and pricing structures calibrated for out-of-pocket cosmetic consumers. According to the Brazilian Institute of Geography and Statistics (IBGE), Brazil’s Unified Healthcare System (SUS) serves as the exclusive healthcare provider for nearly 72% of the population, covering around 164 million people, providing a dense infrastructure for clinic-based device adoption.
|
Segment Breakdown Clinics (48.0%) · Hospitals · Home Settings |
Regional Insights
Southeast – 52.0% Market Share (2025) | Leading Region
The Southeast, comprising São Paulo, Rio de Janeiro, Minas Gerais, and Espírito Santo, constitutes Brazil's aesthetic device market heartland. Rio de Janeiro's beach culture and year-round outdoor lifestyle create sustained demand for body contouring, hair removal, and skin care procedures. The region attracts the highest concentration of international brand investments, medical tourism patients, and practitioner training centers, reinforcing its dominant market positioning through the forecast period.
Regional Breakdown
|
Segment Breakdown Southeast (52.0%) · South · Northeast · North · Central-West |
South:
The South region encompasses Rio Grande do Sul, Santa Catarina, and Paraná, characterized by a well-educated, middle-to-upper-income consumer base with a strong cultural affinity for aesthetic wellness. The region benefits from a robust private healthcare infrastructure, growing medical tourism from neighboring Argentina and Uruguay, and rising clinic density in cities such as Curitiba and Florianópolis. Device adoption is particularly active in energy-based platforms and injectable categories, with practitioners increasingly trained to international standards.
Northeast:
The Northeast is an emerging growth region for Brazil's aesthetic devices market, supported by rising disposable incomes, urban expansion in cities such as Fortaleza, Salvador, and Recife, and growing consumer awareness of non-invasive aesthetic procedures. Historically underserved relative to Southeast Brazil, the region is now attracting investment from national clinic chains and international device distributors seeking geographic diversification, with infrastructure development progressively closing the gap with more mature markets.
North:
The North region represents a smaller but growing segment, where increasing urbanization and expanding primary healthcare access are gradually creating conditions for aesthetic device market entry. Device adoption is currently concentrated in capital cities such as Belém and Manaus, where higher-income urban populations and improving clinic infrastructure are enabling early-stage growth in non-energy-based injectables and hair removal applications.
Central-West:
The Central-West, encompassing Brasília (Federal District), Mato Grosso, and Goiás, benefits from Brasília's large public sector workforce and above-average household income profile, driving consistent demand for aesthetic procedures. The region's capital city operates a growing number of specialized aesthetic clinics, with practitioners adopting both injectable and energy-based platforms. Agribusiness wealth in Mato Grosso and Goiás is also creating an affluent rural-urban consumer segment with rising aesthetic treatment intent.
Market Outlook 2026-2034
What is the future outlook of the Brazil aesthetic devices market?
The Brazil aesthetic devices market is projected to sustain a compound annual growth rate of 9.70%, expanding from USD 203.24 Million in 2025 to USD 481.35 Million by 2034. Sustained growth will be supported by the accelerating non-energy-based injectables segment, particularly next-generation botulinum toxin formulations and biostimulatory dermal fillers, alongside the rapid adoption of AI-integrated energy-based platforms that deliver personalized multi-modality treatments. Brazil's expanding medical tourism sector, maturing aesthetic clinic infrastructure outside Southeast urban centers, and the growing participation of male consumers across all age cohorts will collectively broaden the market's addressable base through the forecast horizon, reinforcing Brazil's position as Latin America's dominant aesthetic devices market.
Brazil Aesthetic Devices Market Leading Key Players:
The Brazil aesthetic devices market features a competitive landscape of global multinational corporations and regional specialists competing through regulatory approvals, portfolio innovation, practitioner training investments, and strategic distribution partnerships. Key players are leveraging Brazil's medical tourism growth and expanding clinic infrastructure to strengthen device placement and consumable revenue streams.
| Company | Leading Brands | Highlights |
|---|---|---|
| Galderma S.A. | Restylane, Sculptra, Dysport | Expanded biostimulatory injectable portfolio for collagen regeneration in Brazil; strong training program network |
| Cynosure LLC | ICON, SculpSure, TempSure | Unveiled hybrid RF-laser platform for non-invasive body contouring |
| CLASSYS Inc. | Shurink, Ultraformer III | Acquired 77.5% equity in JL Health in October 2025 to strengthen direct distribution of energy-based devices in Brazil |
Key players include Galderma S.A., AbbVie Inc. (Allergan Aesthetics), Merz Pharma GmbH & Co. KGaA, Lumenis Be Ltd., Sisram Medical Ltd. (Alma Lasers), Candela Corporation, Cynosure LLC, CLASSYS Inc., Venus Concept, and Cutera Inc., among others.
Latest Development & News:
- October 2025: CLASSYS, a medical aesthetics specialist, signed an agreement to acquire 77.5% equity in JL Health, a prominent South American distributor specializing in energy-based aesthetic medical equipment. The acquisition, executed through CLASSYS's wholly owned Brazilian subsidiary and funded entirely through cash on hand, strengthens the company's direct market presence in Latin America and supports its strategy to scale operations in high-growth aesthetic device markets.
- November 2024: ViOL expanded its presence in Brazil through a new partnership with ENTERA, showcasing the Sylfirm X radiofrequency microneedling platform at Conecta Experience 2024 and hosting the Sylfirm X Global Symposium. The initiative enhanced reach among Brazilian dermatology and plastic surgery professionals while promoting advanced Korean aesthetic technology within the Brazilian practitioner community.
- September 2025: Hugel Inc. deepened its commercial presence in Brazil by entering a new distribution agreement with Derma Dream for its botulinum toxin product, Letybo. Derma Dream's infrastructure including academic institutional ties, practitioner training centers, and logistics capabilities positions Hugel to capitalize on Brazil's growing demand for injectable aesthetic products across urban and regional markets.
Brazil Aesthetic Devices Market Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Million USD |
| Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Type of Devices Covered |
|
| Applications Covered | Skin Resurfacing and Tightening, Body Contouring and Cellulite Reduction, Hair Removal, Tattoo Removal, Breast Augmentation, Others |
| End Users Covered | Hospitals, Clinics, Home Settings |
| Regions Covered | Southeast, South, Northeast, North, Central-West |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the Brazil aesthetic devices market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the Brazil aesthetic devices market.
- Porter's Five Forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the Brazil aesthetic devices industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Key Questions Answered in This Report
The Brazil aesthetic devices market was valued at USD 203.24 Million in 2025.
The Brazil aesthetic devices market is projected to reach USD 481.35 Million by 2034.
Non-energy-based aesthetic device dominates the market with a 56.0% share, driven by the high repeat frequency of botulinum toxin and dermal filler procedures, the broad accessibility of chemical peels and microdermabrasion, and strong consumer demand for implants, including breast augmentation.
Hair removal commands the market with a 25.0% application share, sustained by Brazil's year-round warm climate, diverse skin-tone demographics, and culturally entrenched grooming practices that generate consistently high treatment volumes across clinic and spa settings.
Clinics lead the end-user segment with a 48.0% share, reflecting their specialized operational model, competitive pricing structures, and practitioner expertise that collectively make them the primary channel for aesthetic device utilization in Brazil.
The Southeast region leads with a 52.0% share, driven by São Paulo and Rio de Janeiro's concentration of high-income consumers, dense clinic infrastructure, international brand investments, and strong medical tourism activity.
Key players include Galderma S.A., AbbVie Inc. (Allergan Aesthetics), Merz Pharma GmbH & Co. KGaA, Lumenis Be Ltd., Sisram Medical Ltd. (Alma Lasers), Candela Corporation, Cynosure LLC, CLASSYS Inc., Venus Concept, and Cutera Inc., among others.
Key trends include accelerating adoption of non-energy-based injectables across broader demographics, medical tourism driving premium device deployment in Southeast Brazil, integration of AI-enabled controls in energy-based platforms, growing male participation in aesthetic treatments, and rapid expansion of home-use device accessibility under ANVISA's risk-based regulatory framework.
Growth is driven by the rising popularity of minimally invasive cosmetic procedures, the expansion of aesthetic medical tourism supported by competitive pricing, an aging population seeking anti-aging device-based treatments, social media normalization of aesthetic procedures, and the expansion of specialized aesthetic clinic infrastructure into secondary Brazilian cities.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












