
Brazil Pharmaceuticals Market Size, Share, Trends and Forecast by Type, Nature, and Region, 2026-2034
Brazil Pharmaceuticals Market Summary:
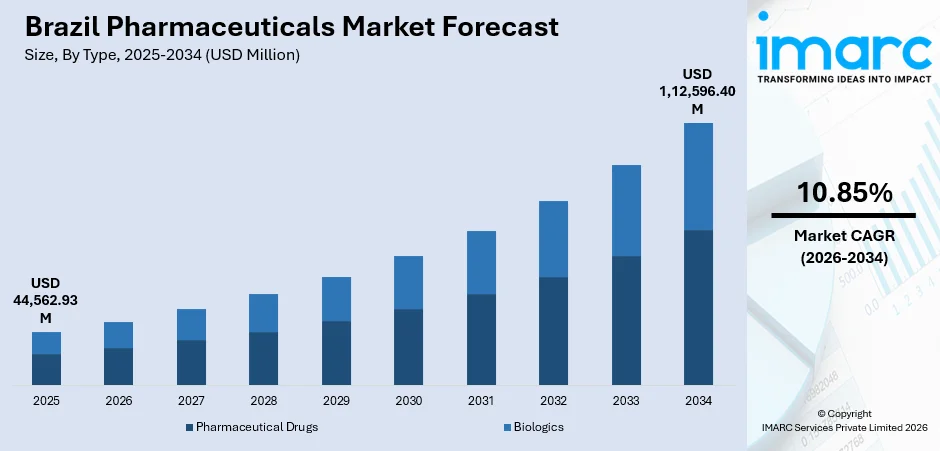
The Brazil pharmaceuticals market size was valued at USD 44,562.93 Million in 2025 and is projected to reach USD 1,12,596.40 Million by 2034, growing at a compound annual growth rate of 10.85% from 2026-2034.
Brazil’s pharmaceuticals market is expanding steadily, supported by rising healthcare expenditure, increasing prevalence of chronic diseases, growing access to public and private healthcare services, and strong demand for generic and specialty medicines, while ongoing investments in domestic manufacturing and regulatory advancements continue to strengthen the country’s position as a leading pharmaceutical hub in Latin America.
Key Takeaways and Insights:
- By Type: Pharmaceutical drugs led the market with an 81.4% share in 2025, fueled by rising prescription demand, the growth of hospital networks, and increased manufacturing of both branded and generic medicines across the country.
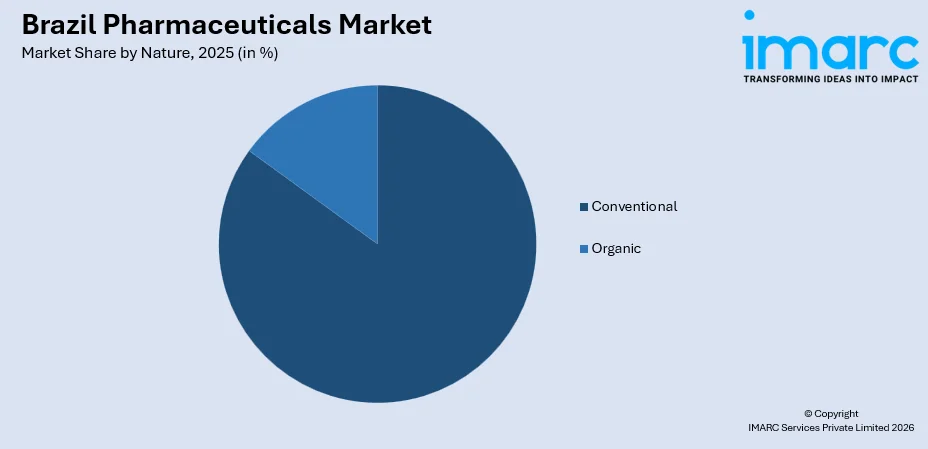
- By Nature: Conventional leads the market with a share of 85% in 2025, due to widespread availability, established manufacturing capacity, cost-effectiveness, and strong physician preference in clinical practice.
- By Region: Southeast represents the largest segment with a market share of 54.3% in 2025, owing to concentrated healthcare infrastructure, higher income levels, dense urban population, and major pharmaceutical industry presence.
- Key Players: Key players in Brazil’s pharmaceuticals market focus on expanding product portfolios, enhancing distribution networks, investing in research and development, and promoting the growth of generic drugs to strengthen market presence and meet the country’s changing healthcare demands.
To get more information on this market Request Sample
Brazil’s pharmaceuticals market is developing steadily as the country strengthens healthcare delivery and expands access to essential medicines. Growth is supported by rising demand for treatments in oncology, diabetes, and respiratory disorders, along with increasing adoption of biosimilars and high-value specialty therapies. According to the International Diabetes Federation (IDF), Brazil ranks fifth globally for diabetes prevalence. The country’s National Health Survey (PNS) estimates diabetes affects 7.9% of the urban population and 6.3% of residents in rural areas. Public health programs and government procurement continue to play a central role in ensuring the availability across regions. The market also benefits from improving clinical research activity and partnerships between multinational firms and domestic manufacturers. Regulatory agencies are advancing approval processes while maintaining strict quality standards, supporting greater innovation. Furthermore, the expansion of private healthcare coverage and retail pharmacy chains is further augmenting distribution efficiency.
Brazil Pharmaceuticals Market Trends:
Expansion of Pharmaceutical E-Commerce Channels
Brazilian pharmaceutical companies are using online platforms to sell medications and complete prescription orders. Retail pharmacy chains and digital health companies are investing in e-commerce models to improve convenience, reach remote consumers, and support home delivery services. The current trend is driving more people to use digital prescriptions together with complete customer care systems. The online distribution system has developed into a main distribution channel for urban residents who need pharmaceutical products.
Increasing Investment in Local Vaccine and Biotech Production
Brazil is strengthening domestic pharmaceutical capacity through rising investment in vaccine manufacturing and biotechnology-based production. Accordingly, Brazil’s pharmaceutical industry invested BRL 3.4 Billion in vaccines for high-incidence diseases, including dengue, RSV, and pneumococcal infections, through public-private partnerships, technology transfers, and expanded production, aiming to strengthen domestic supply and improve public vaccination coverage. Likewise, public-private collaborations are supporting the development of local facilities to reduce reliance on imports and improve supply resilience. This trend is especially significant for immunization programs and advanced biologic medicines. Furthermore, growing biotech investment is positioning Brazil as an emerging regional producer of complex therapeutic products.
Growth of Personalized and Precision Medicine Adoption
The Brazil pharmaceuticals market is experiencing a gradual shift toward personalized medicine, supported by advances in genomics and targeted therapy development. For example, in April 2025, CellAction received a Franco-Brazilian chair to advance CAR-T cell therapy for rare lymphomas, fostering bilateral scientific collaboration, accelerating clinical trials by 2026, and promoting translational research, international exchange of expertise, and equitable patient access to innovative treatments. Furthermore, hospitals and specialty clinics are adopting precision-based treatment approaches, particularly in cancer and rare disease management. Pharmaceutical companies are also expanding access programs and launching therapies tailored to individual patient profiles. This trend reflects increasing demand for innovative treatments and improved healthcare outcomes.
Market Outlook 2026-2034:
Brazil’s pharmaceuticals market outlook remains positive as healthcare demand continues to rise alongside population growth and expanding medical coverage. The IBGE’s Population Estimates report shows that Brazil’s population reached 213.4 million in 2025, an increase of 0.39% from the previous year. Future development is expected to be supported by a stronger focus on preventive care, increasing penetration of specialty pharmacies, and wider adoption of advanced drug formulations. The market is also likely to benefit from continued investments in regulatory transparency and manufacturing competitiveness. Apart from this, expansion into underserved regions and rising therapeutic demand for mental health and autoimmune treatments will further contribute to sustained growth prospects. The market generated a revenue of USD 44,562.93 Million in 2025 and is projected to reach a revenue of USD 1,12,596.40 Million by 2034, growing at a compound annual growth rate of 10.85% from 2026-2034.
Brazil Pharmaceuticals Market Report Segmentation:
|
Segment Category |
Leading Segment |
Market Share |
|
Type |
Pharmaceutical Drugs |
81.4% |
|
Nature |
Conventional |
85% |
|
Region |
Southeast |
54.3% |
Type Insights:
- Pharmaceutical Drugs
- Cardiovascular Drugs
- Dermatology Drugs
- Gastrointestinal Drugs
- Genito-Urinary Drugs
- Hematology Drugs
- Anti-Infective Drugs
- Metabolic Disorder Drugs
- Musculoskeletal Disorder Drugs
- Central Nervous System Drugs
- Oncology Drugs
- Ophthalmology Drugs
- Respiratory Diseases Drugs
- Biologics
- Monoclonal Antibodies (MAbs)
- Therapeutic Proteins
- Vaccines
Pharmaceutical drugs dominates with a market share of 81.4% of the total Brazil pharmaceuticals market in 2025.
The segment’s leadership is supported by strong demand for prescription medicines across public and private healthcare systems. Widespread treatment requirements for chronic diseases, along with extensive availability of branded and generic drugs, continue to reinforce the segment’s dominant position within the national pharmaceutical industry. An industry report highlights that Brazil’s aging population is driving a rise in non-communicable diseases (NCDs). Hypertension, diabetes, and cancer now cause more than 70% of deaths, highlighting a major transformation in the country’s overall health landscape.
The segment remains central amid the continued expansion of hospital care, retail pharmacy distribution, and government-supported access programs. Pharmaceutical drugs are widely used across multiple therapeutic areas, driving consistent consumption levels. Moreover, ongoing investment by manufacturers in product development and regulatory compliance further strengthens the importance of conventional drug formulations in Brazil’s overall healthcare landscape.
Nature Insights:
Access the comprehensive market breakdown Request Sample
- Organic
- Conventional
Conventional dominates with a market share of 85% of the total Brazil pharmaceuticals market in 2025.
The dominance reflects the widespread reliance on established chemical-based drug formulations that remain the primary choice across hospitals, clinics, and retail pharmacies. Conventional medicines are extensively prescribed for acute and chronic conditions, supported by strong domestic manufacturing capacity and broad availability within Brazil’s healthcare distribution networks.
The segment continues to lead due to cost efficiency, proven therapeutic effectiveness, and high physician confidence in traditional treatment protocols. Conventional drugs also benefit from well-developed regulatory frameworks and standardized production processes. Their accessibility across public health programs and private healthcare providers ensures sustained demand, reinforcing their central role in Brazil’s overall pharmaceutical consumption.
Region Insights:
- Southeast
- South
- Northeast
- North
- Central-West
Southeast exhibits a clear dominance with a 54.3% share of the total Brazil pharmaceuticals market in 2025.
The region benefits from its concentrated population, advanced healthcare infrastructure, and the presence of major hospitals, research centers, and pharmaceutical manufacturing hubs. As such, in October 2025, Roquette opened its Pharmaceutical Innovation Center in São Paulo, featuring advanced labs and training spaces. The facility strengthens Latin America as a collaborative hub, accelerating drug development, fostering partnerships, and promoting STEM opportunities for young women. Similarly, high consumer purchasing power also supports greater medicine consumption than in other regions.
Regional leadership is further reinforced by dense retail pharmacy networks and greater access to specialized treatments. The Southeast also attracts significant investment from multinational and domestic pharmaceutical companies due to its strong distribution channels and proximity to key medical institutions. Besides this, continued urban expansion and healthcare modernization are expected to sustain the region’s dominance in Brazil’s pharmaceutical market.
Market Dynamics:
Growth Drivers:
Why is the Brazil Pharmaceuticals Market Growing?
Expansion of Generic Drug Export Potential
Brazil’s pharmaceutical sector is increasingly focusing on strengthening its role as an exporter of generic medicines to other Latin American markets. Domestic manufacturers are improving production efficiency and meeting international regulatory requirements to access new trade opportunities. As such, in April 2025 Novo Nordisk invested USD 1.09 Billion to expand its Montes Claros plant in Brazil, boosting injectable drug production, including GLP-1s like Ozempic, creating 600 jobs, and incorporating sustainable energy and water management systems. This trend is supported by regional demand for affordable therapies and Brazil’s growing industrial base. Expansion of export-oriented strategies is improving competitiveness and encouraging investment in higher-capacity manufacturing facilities.
Rising Adoption of Digital Clinical Trials
The Brazil pharmaceuticals market is witnessing a gradual shift toward digitalization in clinical research processes. Companies are adopting remote patient monitoring, electronic data capture, and decentralized trial models to accelerate drug development timelines. Digital clinical trials improve patient recruitment across diverse regions and reduce operational barriers. Accordingly, Artha CRO in Brazil digitized all clinical operations with Datacapt, achieving 50% staffing reduction, real-time sponsor transparency, and scalable, GCP-compliant processes, enabling rapid deployment, operational efficiency, and strengthened client trust across 100% of studies. This trend is also enhancing research transparency and supporting the faster introduction of innovative therapies within the Brazilian healthcare system.
Increasing Role of Pharmaceutical Licensing Partnerships
Brazil is experiencing growth in licensing agreements between multinational pharmaceutical firms and local companies. These partnerships enable faster entry of advanced therapies into the domestic market while supporting technology transfer and local production. Licensing arrangements are particularly relevant for specialty drugs and complex treatments. This trend is helping domestic players expand portfolios and strengthening Brazil’s position as an attractive market for international pharmaceutical collaboration.
Market Restraints:
What Challenges the Brazil Pharmaceuticals Market is Facing?
High Dependence on Imported Active Ingredients
Brazil’s pharmaceutical industry faces challenges due to significant reliance on imported active pharmaceutical ingredients (APIs) and raw materials. Supply chain disruptions, currency fluctuations, and global pricing volatility can affect production stability and increase manufacturing costs. This dependence limits self-sufficiency and creates vulnerability for domestic drug availability. Strengthening local API production remains a critical hurdle for improving long-term resilience in the sector.
Pricing Pressure and Public Procurement Constraints
The Brazil pharmaceuticals market experiences strong pricing pressure, particularly due to government procurement policies and regulated drug pricing mechanisms. Public healthcare programs demand cost-effective supply, limiting profit margins for manufacturers. Competitive tender processes also create barriers for smaller companies seeking market access. These constraints can reduce incentives for high-value innovation and challenge the sustainable expansion of specialty and advanced therapeutic segments.
Regulatory Delays and Approval Timelines
Pharmaceutical companies in Brazil often encounter lengthy regulatory approval processes that can delay product launches and limit rapid access to new treatments. Compliance requirements for safety, efficacy, and quality standards are stringent, increasing administrative burden. Regulatory delays also discourage faster adoption of innovative therapies, particularly in biologics and rare disease treatments. Streamlining approval pathways remains essential for improving market responsiveness and competitiveness.
Competitive Landscape:
Brazil’s pharmaceuticals market is highly competitive, with strong participation from domestic manufacturers and multinational corporations. Local firms maintain a significant presence through extensive generic drug portfolios and well-established distribution networks, while global companies compete through patented therapies, biologics, and specialty medicines. Market competition is influenced by regulated pricing structures and public procurement dominance, which pressure margins and encourage efficiency. Companies increasingly invest in research and development (R&D) partnerships, licensing deals, and local production capabilities to strengthen positioning. The competitive environment also reflects rising demand for advanced treatments, driving product differentiation and innovation across therapeutic categories.
Recent Developments:
- In November 2025, Celltrion launched Omlyclo, the first omalizumab biosimilar in Brazil and major European countries, securing 70% of the Dutch market. Approved across multiple regions since 2024–2025, Omlyclo faces competition from other biosimilars under development by Aurobindo, Teva, Glenmark, and Kashiv.
- In November 2025, Zydus and Torrent announced its plan to enter the growing semaglutide market for diabetes and obesity. Torrent has filed with Brazil’s ANVISA, targeting post-March 2026 patent expiry, while Zydus plans launches in India, later expanding to Brazil and Canada. Both injectable and oral formulations are in development.
Brazil Pharmaceuticals Market Report Coverage:
|
Report Features |
Details |
|
Base Year of the Analysis |
2025 |
|
Historical Period |
2020-2025 |
|
Forecast Period |
2026-2034 |
|
Units |
USD Million |
|
Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
|
Types Covered |
|
|
Nature Covered |
Organic, Conventional |
|
Regions Covered |
Southeast, South, Northeast, North, Central-West |
|
Customization Scope |
10% Free Customization |
|
Post-Sale Analyst Support |
10-12 Weeks |
|
Delivery Format |
PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Frequently Asked Questions About the Brazil Pharmaceuticals Market Report
The Brazil pharmaceuticals market size was valued at USD 44,562.93 Million in 2025.
The Brazil pharmaceuticals market is expected to grow at a compound annual growth rate of 10.85% from 2026-2034 to reach USD 1,12,596.40 Million by 2034.
Pharmaceutical drugs dominated the market with 81.4% share in 2025. This dominance reflects strong demand for prescription and over-the-counter medications, driving revenue growth and shaping the country’s healthcare landscape.
Key factors driving the Brazil pharmaceuticals market include rising prevalence of chronic diseases, increasing healthcare access, government support for public health programs, expanding middle-class population, growing demand for innovative and generic drugs, and continuous investments in research, development, and advanced manufacturing capabilities.
Major challenges in the Brazil pharmaceuticals market include complex regulatory approval processes, high costs of research and development, limited healthcare infrastructure in rural areas, pricing pressures from government policies, competition from low-cost generics, and difficulties in supply chain management and distribution across the country’s vast territory.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












