
Capnography Devices Market Report by Product (Capnometers, Accessories), Product Type (Handheld, Conventional), Technology (Main-stream, Side-stream, Micro-stream), Application (Trauma and Emergency Care, Cardiac Care, Respiratory Monitoring, and Others), End-User (Hospitals and Clinics, Ambulatory Surgery Centers (ASCs), and Others), and Region 2026-2034
Market Overview:
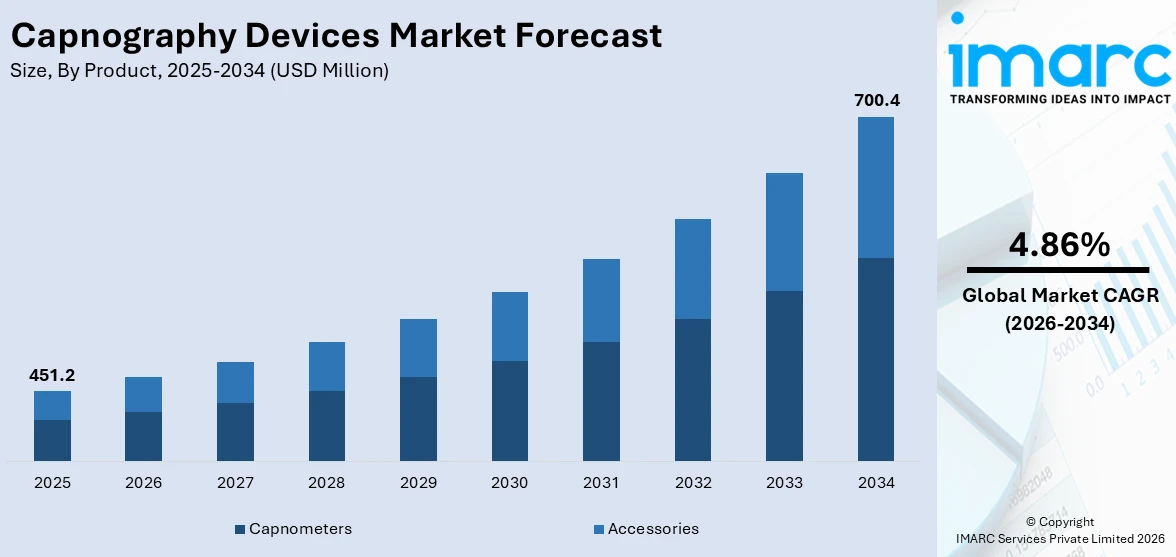
The global capnography devices market size reached USD 451.2 Million in 2025. Looking forward, IMARC Group expects the market to reach USD 700.4 Million by 2034, exhibiting a growth rate (CAGR) of 4.86% during 2026-2034.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 451.2 Million |
|
Market Forecast in 2034
|
USD 700.4 Million |
| Market Growth Rate (2026-2034) | 4.86% |
Capnography devices refer to various medical equipment that are used for measuring the amount of carbon dioxide (CO2) present in the patient’s bloodstream. They measure the inhaled and exhaled concentration of CO2 in the body. The devices are used for patients undergoing anesthesia to ensure that they are receiving ample oxygen throughout the surgical procedure. These devices are commonly available in handheld, standalone and multi-parameter variants based on main-, side- and micro-stream technologies. They also aid in diagnosing the cause of respiratory distress, providing real-time feedback on treatment effects and detecting shocks.
To get more information on this market Request Sample
The increasing prevalence of respiratory and chronic obstructive pulmonary diseases (COPD), such as pulmonary embolism and asthma, is one of the key factors driving the growth of the market. These devices are also being used for monitoring patients under intensive care who are suffering from the coronavirus disease (COVID-19). Furthermore, the rising geriatric population, which is more susceptible to lung disorders, is also providing driving the market growth. Capnography devices are widely being used for operative and post-operative care of patients and are commonly used in operation, recovery and critical rooms, wards and ambulances. Various product innovations, such as the development of technologically advanced devices that offer ease-of-connection, convenient sterilization and enable accurate monitoring of non-intubated substances, are acting as another growth-inducing factor. The market is further driven by the launch of variants that are integrated with photodetectors, molecular correlation spectroscopy and miniaturized multi-gas monitors that are highly accurate and have an adjustable flow rate. Other factors, including significant improvements in the healthcare infrastructure, along with the development of portable and point-of-care (POC) capnography devices, are projected to create a positive outlook for the market.
Key Market Segmentation:
IMARC Group provides an analysis of the key trends in each sub-segment of the global capnography devices market report, along with forecasts at the global, regional and country level from 2026-2034. Our report has categorized the market based on product, product type, technology, application and end-user.
Breakup by Product:
- Capnometers
- Accessories
Breakup by Product Type:
- Handheld
- Conventional
Breakup by Technology:
- Main-stream
- Side-stream
- Micro-stream
Breakup by Application:
Access the comprehensive market breakdown Request Sample
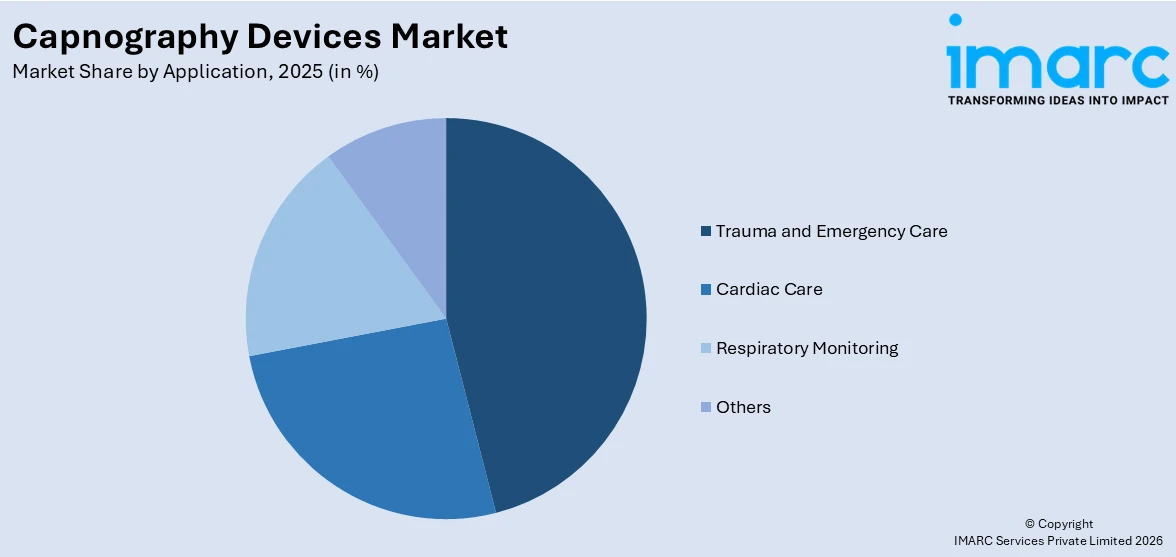
- Trauma and Emergency Care
- Cardiac Care
- Respiratory Monitoring
- Others
Breakup by End-User:
- Hospitals and Clinics
- Ambulatory Surgery Centers (ASCs)
- Others
Breakup by Region:
- North America
- United States
- Canada
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
Competitive Landscape:
The report has also analysed the competitive landscape of the market with some of the key players being Becton, Dickinson and Company (BD), Burtons Medical Equipment Ltd., Opto Cardiac Care Limited (Criticare Technologies Inc.), FUTEK Advanced Sensor Technology, Inc., DiaMedica Therapeutics, Inc. (DMAC), Drägerwerk AG, EDAN Instruments Inc., Hill-Rom Services Inc., Infinium Medical, Inc., Koninklijke Philips N.V., Masimo Corporation, Medtronic Inc., Nihon Kohden Corporation, Nonin Medical Inc., Smiths Medical MD, Inc. and Zoe Medical Incorporated. etc.
Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Million USD |
| Segment Coverage | Product, Product Type, Technology, Application, End-User, Region |
| Region Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Becton, Dickinson and Company (BD), Burtons Medical Equipment Ltd., Opto Cardiac Care Limited (Criticare Technologies Inc.), FUTEK Advanced Sensor Technology, Inc., DiaMedica Therapeutics, Inc. (DMAC), Drägerwerk AG, EDAN Instruments Inc., Hill-Rom Services Inc., Infinium Medical, Inc., Koninklijke Philips N.V., Masimo Corporation, Medtronic Inc., Nihon Kohden Corporation, Nonin Medical Inc., Smiths Medical MD, Inc. and Zoe Medical Incorporated |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Frequently Asked Questions About the Capnography Devices Market Report
The global capnography devices market was valued at USD 451.2 Million in 2025.
We expect the global capnography devices market to exhibit a CAGR of 4.86% during 2026-2034.
The rising utilization of capnography devices in diagnosing the cause of respiratory distress, providing real-time feedback on treatment effects, and detecting shocks, is primarily driving the global capnography devices market.
The sudden outbreak of the COVID-19 pandemic has led to the increasing adoption of capnography devices for monitoring critically infected COVID-19 patients.
Based on the product, the global capnography devices market can be segmented into capnometers and accessories. Currently, capnometers hold the majority of the total market share.
Based on the product type, the global capnography devices market has been divided into handheld and conventional, where handheld capnography devices currently exhibit a clear dominance in the market.
Based on the technology, the global capnography devices market can be categorized into main-stream, side-stream, and micro-stream. Currently, side-stream technology accounts for the majority of the global market share.
Based on the application, the global capnography devices market has been segregated into trauma and emergency care, cardiac care, respiratory monitoring, and others. Among these, trauma and emergency care currently holds the largest market share.
Based on the end-user, the global capnography devices market can be bifurcated into hospitals and clinics, Ambulatory Surgery Centers (ASCs), and others. Currently, hospitals and clinics exhibit a clear dominance in the market.
On a regional level, the market has been classified into North America, Asia Pacific, Europe, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global capnography devices market include Becton, Dickinson and Company (BD), Burtons Medical Equipment Ltd., Opto Cardiac Care Limited (Criticare Technologies Inc.), FUTEK Advanced Sensor Technology, Inc., DiaMedica Therapeutics, Inc. (DMAC), Drägerwerk AG, EDAN Instruments Inc., Hill-Rom Services Inc., Infinium Medical, Inc., Koninklijke Philips N.V., Masimo Corporation, Medtronic Inc., Nihon Kohden Corporation, Nonin Medical Inc., Smiths Medical MD, Inc. and Zoe Medical Incorporated. etc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












