
Cardiac PoC Testing Devices Market by Product Type (Cardiac Markers Test, Analyzers), End User (Hospitals, Diagnostic Laboratories, and Others), and Region 2026-2034
Market Overview:
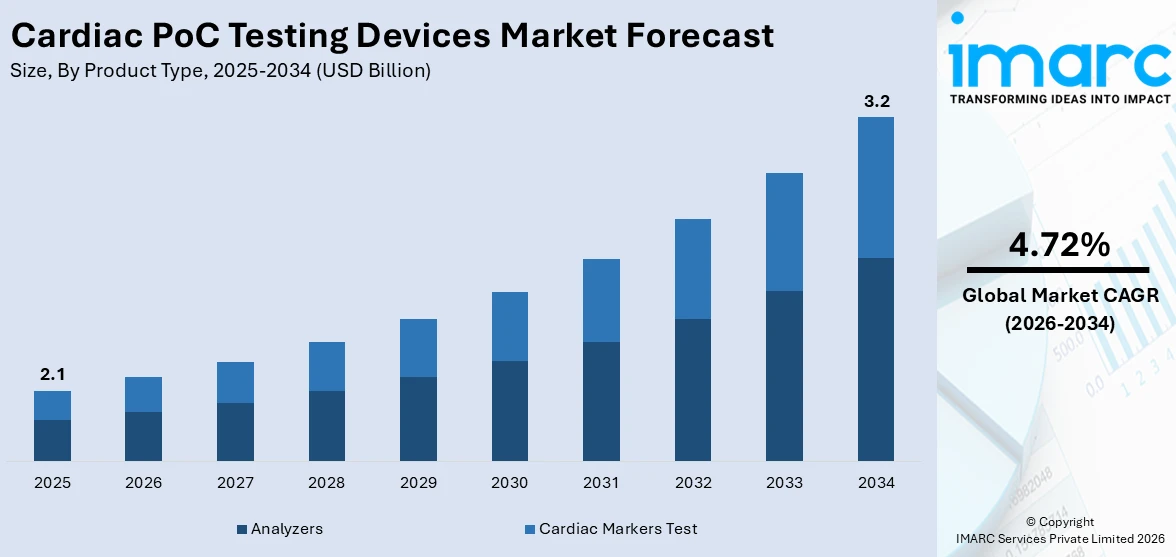
The global cardiac PoC testing devices market size reached USD 2.1 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 3.2 Billion by 2034, exhibiting a growth rate (CAGR) of 4.72% during 2026-2034. The growing prevalence of cardiovascular diseases due to excessive consumption of tobacco; rising need to detect brain natriuretic peptides, kinase, synthesizing troponin, and lactate dehydrogenase isoenzymes; and the thriving medical industry represent some of the key factors driving the market.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 2.1 Billion |
|
Market Forecast in 2034
|
USD 3.2 Billion |
| Market Growth Rate (2026-2034) | 4.72% |
Cardiac point-of-care (PoC) testing devices are innovative and portable medical instruments designed for the rapid and accurate diagnosis of cardiac conditions. They are compact, user-friendly, and offer real-time results, enabling physicians to make prompt and informed decisions about patient care. They provide healthcare professionals with the ability to conduct diagnostic tests at the bedside of patients, eliminate the need for laboratory-based testing, and reduce turnaround times for critical cardiac assessments. They enhance the efficiency of clinical workflows by delivering accurate and reliable results within minutes. They assist in performing a variety of diagnostic tests, including cardiac biomarker analysis, coagulation monitoring, cholesterol profiling, and electrolyte measurement. They aid in obtaining immediate test results that facilitate more efficient patient triage, reduce overcrowding in emergency departments, and optimize resource allocation. As they minimize costs associated with sample transportation, lab personnel, and infrastructure by reducing the reliance on centralized laboratories, the demand for cardiac PoC testing devices is rising worldwide.
To get more information on this market Request Sample
Cardiac PoC Testing Devices Market Trends:
At present, the rising prevalence of cardiovascular diseases due to the excessive consumption of tobacco and alcohol represents one of the crucial factors bolstering the growth of the market. Moreover, there is an increase in the utilization of cardiac PoC testing devices to detect myocardial infarction and coronary syndromes around the world. This, along with the thriving medical industry, is supporting the growth of the market. In addition, the growing employment of these devices to identify brain natriuretic peptides, kinase, synthesizing troponin, and lactate dehydrogenase isoenzymes that are generated during heart failure is positively influencing the market. Besides this, key players are focusing on launching smartphone-based tests for cardiovascular diseases. They are also developing technologically advanced cardiac biomarker analyzers, which is bolstering the growth of the market. Additionally, there is a rise in the demand for cardiac analyzers, as they have software to provide an internal standard to account for membrane variability that is intrinsic to tests and integrated LCD displays for user convenience and visual clarity. This, coupled with the ability of analyzers to quantify the concentration of biomarkers in whole blood, serum, and plasma, which helps in clinical diagnosis, is impelling the growth of the market. Furthermore, the growing technological advancements in PoC testing devices are offering a favorable market outlook.
Key Market Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global cardiac PoC testing devices market, along with forecasts at the global, regional, and country levels from 2026-2034. Our report has categorized the market based on product type and end user.
Product Type Insights:
- Cardiac Markers Test
- Cardiac Troponin (cTn) Test
- Myoglobin Test
- Creatine Kinase MB Isoenzyme (CK-MB) Test
- Combinational Test Kits
- Brain Natriuretic Peptide (BNP) Test
- Analyzers
The report has provided a detailed breakup and analysis of the cardiac PoC testing devices market based on the product type. This includes cardiac markers test [cardiac troponin (cTn) test, myoglobin test, creatine kinase MB isoenzyme (CK-MB) test, combinational test kits, and brain natriuretic peptide (BNP) test] and analyzers. According to the report, analyzers represented the largest segment.
End User Insights:
Access the comprehensive market breakdown Request Sample
- Hospitals
- Diagnostic Laboratories
- Others
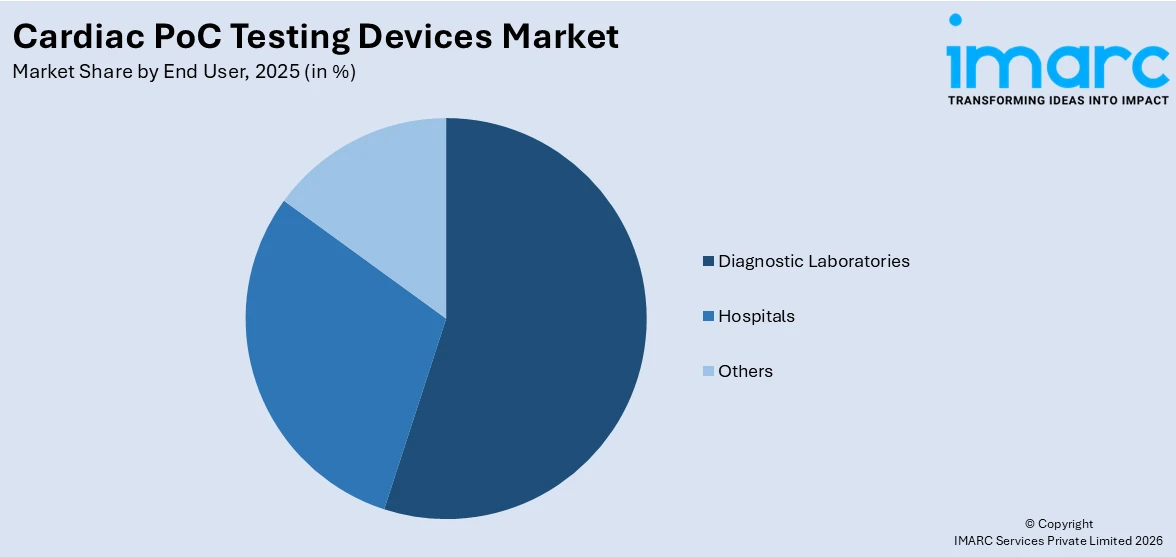
A detailed breakup and analysis of the cardiac PoC testing devices market based on the end user has also been provided in the report. This includes hospitals, diagnostic laboratories, and others. According to the report, diagnostic laboratories accounted for the largest market share.
Regional Insights:
- North America
- United States
- Canada
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America was the largest market for cardiac PoC testing devices. Some of the factors driving the North America cardiac PoC testing devices market included the growing cardiovascular burden among the population, increasing healthcare expenditure, rising geriatric population, etc.
Competitive Landscape:
The report has also provided a comprehensive analysis of the competitive landscape in the global cardiac PoC testing devices market. Detailed profiles of all major companies have been provided. Some of the companies covered include Abbott Laboratories, ACON Laboratories Inc., Alfa Scientific Designs Inc., Bio-Rad Laboratories Inc., Danaher Corporation, F. Hoffmann-La Roche AG, LifeSign LLC, Nano-Ditech Corporation, Nexus-Dx Inc., etc. Kindly note that this only represents a partial list of companies, and the complete list has been provided in the report.
Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical and Forecast Trends, Industry Catalysts and Challenges, Segment-Wise Historical and Predictive Market Assessment:
|
| Product Types Covered |
|
| End Users Covered | Hospitals, Diagnostic Laboratories, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Abbott Laboratories, ACON Laboratories Inc., Alfa Scientific Designs Inc., Bio-Rad Laboratories Inc., Danaher Corporation, F. Hoffmann-La Roche AG, LifeSign LLC, Nano-Ditech Corporation, Nexus-Dx Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report:
- How has the global cardiac PoC testing devices market performed so far, and how will it perform in the coming years?
- What are the drivers, restraints, and opportunities in the global cardiac PoC testing devices market?
- What is the impact of each driver, restraint, and opportunity on the global cardiac PoC testing devices market?
- What are the key regional markets?
- Which countries represent the most attractive cardiac PoC testing devices market?
- What is the breakup of the market based on the product type?
- Which is the most attractive product type in the cardiac PoC testing devices market?
- What is the breakup of the market based on the end user?
- Which is the most attractive end user in the cardiac PoC testing devices market?
- What is the competitive structure of the global cardiac PoC testing devices market?
- Who are the key players/companies in the global cardiac PoC testing devices market?
Key Benefits for Stakeholders:
- IMARC’s report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the cardiac PoC testing devices market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global cardiac PoC testing devices market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assist stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the cardiac PoC testing devices industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












