
Choroidal Neovascularization Market Report by Product Type (Intravenous, Intravitreal), Diagnosis (Fluorescein Angiography (FA), Indocyanine Green (ICG) Angiography, Spectral Domain Optical Coherence Tomography (OCT), and Others), Treatment Type (Angiogenesis Inhibitors Injection, Photodynamic Therapy, Laser photocoagulation, Low-dose Radiation Therapy, Surgery), Application (Extreme Myopia, Malignant Myopic Degeneration, Age-Related Developments), and Region 2026-2034
Market Overview:
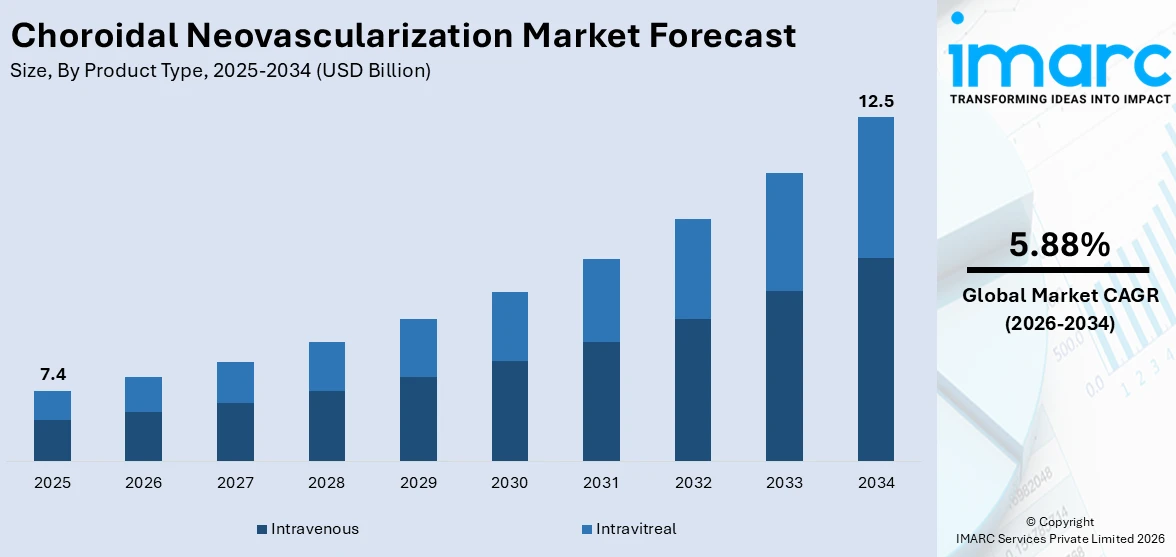
The global choroidal neovascularization market size reached USD 7.4 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 12.5 Billion by 2034, exhibiting a growth rate (CAGR) of 5.88% during 2026-2034.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 7.4 Billion |
|
Market Forecast in 2034
|
USD 12.5 Billion |
| Market Growth Rate 2026-2034 | 5.88% |
Choroidal neovascularization (CNV) refers to the creation of new blood vessels in the choroid layer of the eyes affecting the Bruch's membrane and retinal pigment epithelium (RPE). Its symptoms include lipid exudation, subretinal blood and fluid, and distortion, waviness and black spots in the central vision. At present, a combination of procedures, such as fluorescein angiography (FA), indocyanine green (ICG) angiography and optical coherence tomography (OCT) scanning, are utilized for diagnosing CNV. As per the results, doctors then prescribe anti-vascular endothelial growth factor (VEGF) drugs, thermal laser treatment and photodynamic therapy (PDT), to prevent the growth of the blood vessels.
To get more information on this market Request Sample
The market is experiencing growth on account of the rising prevalence of age-related macular degeneration (ARMD), ocular histoplasmosis, pathologic myopia, angioid streaks, choroidal tears and inflammatory diseases of the choroid and retina. Moreover, the increasing geriatric population, which is more vulnerable to serious health conditions, is acting as another key factor bolstering the market growth. Furthermore, the growing adoption of combination drugs, along with the introduction of long-lasting anti-VEGF drugs, is propelling the demand for CNV treatments worldwide. Other factors, such as improving healthcare infrastructure and the increasing investments in research and development (R&D) activities, are also contributing to the growth of the market.
Key Market Segmentation:
IMARC Group provides an analysis of the key trends in each sub-segment of the global choroidal neovascularization market report, along with forecasts at the global, regional and country level from 2026-2034. Our report has categorized the market based on product type, type of diagnosis, treatment type and application.
Breakup by Product Type:
- Intravenous
- Intravitreal
Breakup by Type of Diagnosis:
- Fluorescein Angiography (FA)
- Indocyanine Green (ICG) Angiography
- Spectral Domain Optical Coherence Tomography (OCT)
- Others
Breakup by Treatment Type:
- Angiogenesis Inhibitors Injection
- Photodynamic Therapy
- Laser photocoagulation
- Low-dose Radiation Therapy
- Surgery
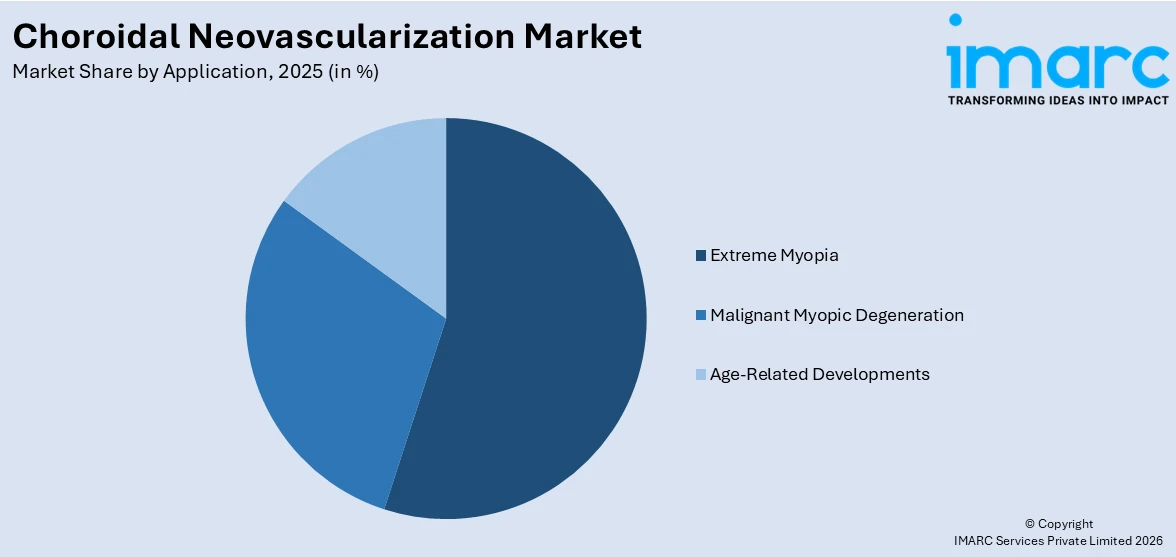
Breakup by Application:
Access the comprehensive market breakdown Request Sample
- Extreme Myopia
- Malignant Myopic Degeneration
- Age-Related Developments
Breakup by Region:
- North America
- United States
- Canada
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
Competitive Landscape:
The report has also analysed the competitive landscape of the market with some of the key players being Bausch Health Companies Inc., Bayer AG, Gilead Sciences, Inc., F. Hoffmann-La Roche AG, MaaT Pharma, Novartis AG, Pfizer Inc., Quark Pharmaceuticals Inc. (SBI ALApharma Co. Limited), Regeneron Pharmaceuticals Inc. and Sanwa Kagaku Kenkyusho Co. Ltd. (Suzuken Co. Ltd.).
Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Segment Coverage | Product Type, Type of Diagnosis, Treatment Type, Application, Region |
| Region Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Bausch Health Companies Inc., Bayer AG, Gilead Sciences, Inc., F. Hoffmann-La Roche AG, MaaT Pharma, Novartis AG, Pfizer Inc., Quark Pharmaceuticals Inc. (SBI ALApharma Co. Limited), Regeneron Pharmaceuticals Inc. and Sanwa Kagaku Kenkyusho Co. Ltd. (Suzuken Co. Ltd.) |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report:
- How has the global choroidal neovascularization market performed so far and how will it perform in the coming years?
- What has been the impact of COVID-19 on the global choroidal neovascularization market?
- What are the key regional markets?
- What is the breakup of the market based on the product type?
- What is the breakup of the market based on the type of diagnosis?
- What is the breakup of the market based on the treatment type?
- What is the breakup of the market based on the application?
- What are the various stages in the value chain of the industry?
- What are the key driving factors and challenges in the industry?
- What is the structure of the global choroidal neovascularization market and who are the key players?
- What is the degree of competition in the industry?
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












