
Duchenne Muscular Dystrophy Drugs Market Size, Share, Trends and Forecast by Product Type, Therapeutic Approach, End User, and Region, 2026-2034
Duchenne Muscular Dystrophy Drugs Market Size and Share:
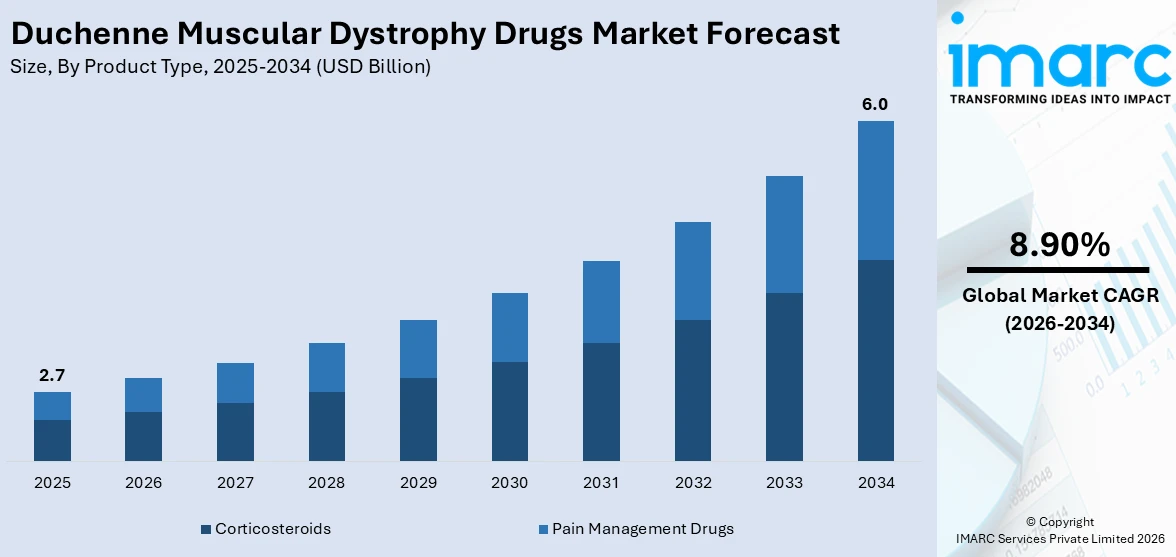
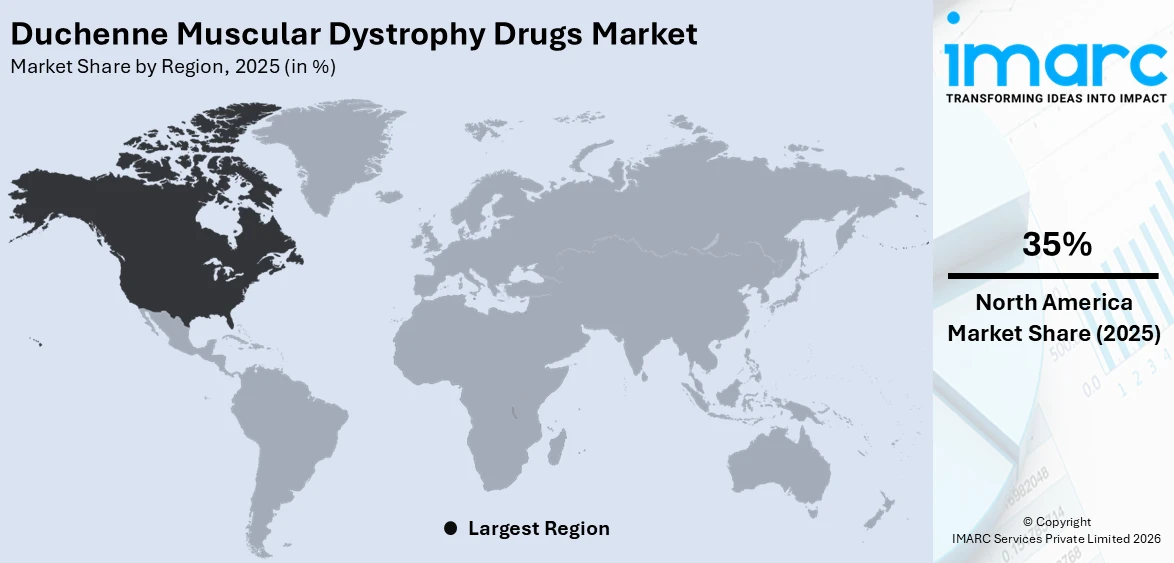
The global duchenne muscular dystrophy drugs market size was valued at USD 2.7 Billion in 2025. Looking forward, IMARC Group estimates the market to reach USD 6.0 Billion by 2034, exhibiting a CAGR of 8.90% from 2026-2034. North America currently dominates the market, holding a market share of 35% in 2025. The region benefits from a well-established healthcare infrastructure, strong regulatory frameworks supporting orphan drug designations, high levels of investment in rare disease research, and widespread insurance coverage that facilitates patient access to advanced therapies, all contributing to the duchenne muscular dystrophy drugs market share.
The global duchenne muscular dystrophy drugs market is primarily driven by the rising prevalence of Duchenne muscular dystrophy and growing awareness among healthcare providers and caregivers regarding early diagnosis and intervention. Advances in molecular biology, genetic engineering, and precision medicine are enabling the development of innovative therapeutic modalities, including gene therapies and antisense oligonucleotides, which are broadening the treatment landscape. Furthermore, increased government funding for rare disease research, coupled with favorable regulatory pathways such as orphan drug designations and accelerated approvals, is encouraging pharmaceutical companies to invest heavily in DMD drug development. The expanding clinical trial pipeline, supported by collaborations between academic institutions, biotechnology firms, and patient advocacy groups, is accelerating the translation of novel compounds from preclinical stages to commercial availability, thereby propelling the duchenne muscular dystrophy drugs market growth across all major regions.
To get more information on this market Request Sample
The United States has emerged as a major region in the duchenne muscular dystrophy drugs market owing to many factors. The country possesses a robust pharmaceutical research ecosystem, with leading academic medical centers and biotechnology companies dedicating substantial resources to neuromuscular disease therapies. Supportive federal policies, including the Orphan Drug Act and expedited review pathways, have incentivized drug development for rare conditions like DMD. For instance, the FDA expanded the approval of ELEVIDYS (delandistrogene moxeparvovec) in 2024, making it available as a gene therapy for ambulatory and non-ambulatory individuals aged four years and older with confirmed DMD gene mutations. Additionally, comprehensive newborn screening programs and advanced diagnostic capabilities are enabling earlier identification of affected patients, which supports timely therapeutic intervention and drives the market growth across the nation.
Duchenne Muscular Dystrophy Drugs Market Trends:
Advancements in Gene Therapy Approaches
The rapid evolution of gene therapy technologies is significantly reshaping the therapeutic landscape for Duchenne muscular dystrophy. Researchers are leveraging adeno-associated virus vectors to deliver functional micro-dystrophin genes directly into muscle tissue, aiming to restore partial dystrophin production and slow disease progression. These single-dose intravenous infusion therapies represent a paradigm shift from chronic symptomatic management to potentially durable disease-modifying interventions. Additionally, the development of CRISPR-based gene editing platforms is opening new avenues for precise correction of dystrophin gene mutations, with several candidates advancing through preclinical evaluation. For instance, in June 2025, the FDA granted Rare Pediatric Disease Designation to PBGENE-DMD, an in vivo gene editing therapy by Precision BioSciences developed using the ARCUS platform, which targets exons 45 to 55 of the dystrophin gene and holds the potential to benefit up to 60 percent of DMD patients. These breakthroughs are attracting substantial investment and regulatory support globally.
Rising Adoption of Novel Corticosteroids
The introduction of next-generation corticosteroids with improved safety profiles is transforming the standard of care for DMD patients and supporting the duchenne muscular dystrophy drugs market forecast. Traditional corticosteroids such as prednisone and deflazacort have long been the cornerstone of DMD management, providing meaningful benefits in preserving muscle function and delaying loss of ambulation. However, their prolonged use is associated with significant adverse effects including growth stunting, bone density loss, and behavioral changes. Novel dissociative corticosteroids have been developed to retain anti-inflammatory efficacy while minimizing these side effects through selective receptor modulation. For instance, in November 2025, Santhera Pharmaceuticals reported positive five-year data from its GUARDIAN study of vamorolone (Agamree), demonstrating durable efficacy comparable to standard corticosteroids with significantly lower rates of vertebral fractures and improved growth trajectories in DMD patients. These clinical advantages are increasing prescribers' confidence and patient adoption worldwide.
Expanding Nonsteroidal Treatment Options
The approval and development of nonsteroidal therapies are diversifying the DMD treatment arsenal and addressing the duchenne muscular dystrophy drugs market trends. Histone deacetylase inhibitors represent a novel mechanistic approach that targets the downstream pathological processes caused by dystrophin deficiency, including chronic inflammation, impaired muscle regeneration, and progressive fat infiltration. These agents work by modulating gene expression to restore essential cellular repair mechanisms in dystrophic muscle tissue, offering benefits independent of the underlying genetic mutation type. The availability of treatments effective across all genetic variants of DMD is particularly significant, as it expands the eligible patient population beyond mutation-specific therapies. For instance, in April 2025, the European Medicines Agency's CHMP adopted a positive opinion recommending conditional marketing authorisation for Duvyzat (givinostat) by Italfarmaco for DMD patients aged six years and older who are ambulatory. This development follows the FDA approval of givinostat in March 2024, positioning it as a complementary treatment option alongside corticosteroid regimens.
Duchenne Muscular Dystrophy Drugs Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global duchenne muscular dystrophy drugs market, along with forecast at the global, regional, and country levels from 2026-2034. The market has been categorized based on product type, therapeutic approach, and end user.
Analysis by Product Type:
- Corticosteroids
- Prednisolone
- Prednisone
- Deflazacort
- Pain Management Drugs
Corticosteroids holds 65% of the market share. Corticosteroids constitute the most widely prescribed class of drugs for managing DMD, owing to their well-established efficacy in slowing disease progression and preserving motor function. These medications work by reducing inflammation and modulating immune responses within dystrophic muscle tissue, thereby delaying the onset of critical milestones such as loss of ambulation and respiratory decline. Prednisone, prednisolone, and deflazacort remain the primary treatment options within this category, each offering distinct pharmacological profiles suited to individual patient needs. The extensive clinical experience and strong body of evidence supporting corticosteroid therapy have solidified their role as a cornerstone of Duchenne muscular dystrophy (DMD) management worldwide. Ongoing advancements in steroid formulations aim to preserve anti-inflammatory efficacy while minimizing the side effects commonly associated with traditional corticosteroids, further strengthening their importance in long-term DMD treatment strategies.
Analysis by Therapeutic Approach:
- Mutation Suppression
- Exon Skipping
- Steroid Therapy
Exon Skipping leads the market with a share of 40%. Exon skipping therapies represent a targeted molecular approach that uses antisense oligonucleotides to modify pre-messenger RNA splicing, enabling the production of a truncated but partially functional dystrophin protein. This therapeutic strategy addresses specific exon mutations in the dystrophin gene, making it applicable to defined patient subpopulations based on their genetic profiles. The FDA has approved multiple exon skipping therapies targeting different exon mutations, including eteplirsen for exon 51, golodirsen for exon 53, and casimersen for exon 45 skipping. These approvals have collectively expanded treatment eligibility to a significant proportion of the DMD patient population. For instance, in July 2025, the FDA granted Breakthrough Therapy designation to delpacibart zotadirsen by Avidity Biosciences, a novel Antibody Oligonucleotide Conjugate designed for DMD patients with mutations amenable to exon 44 skipping, marking the first such conjugate in development for DMD.
Analysis by End User:
Access the comprehensive market breakdown Request Sample
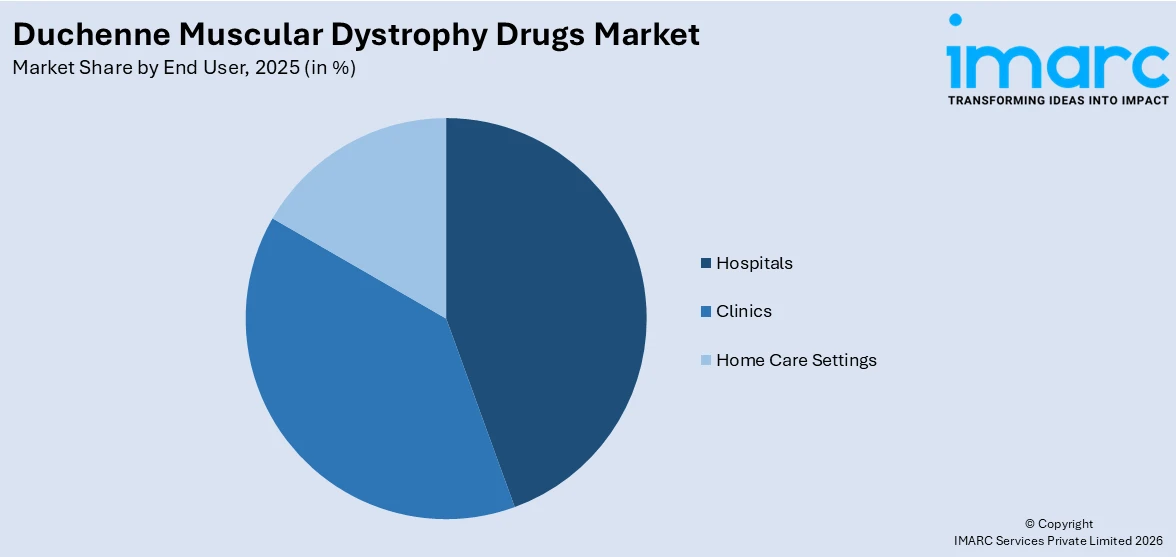
- Hospitals
- Clinics
- Home Care Settings
Hospitals dominate the market, with a share of 48%. Hospitals serve as the primary setting for DMD drug administration and patient management due to the complex nature of the disease and the specialized infrastructure required for advanced therapies. Gene therapies, particularly intravenous infusion-based treatments, necessitate hospital-based delivery with comprehensive monitoring capabilities to manage potential immune-mediated adverse events and ensure patient safety. Furthermore, hospitals house multidisciplinary care teams comprising neurologists, pulmonologists, cardiologists, and rehabilitation specialists who coordinate comprehensive treatment plans for DMD patients across all disease stages. The growing number of designated neuromuscular centers of excellence within hospital networks is strengthening the hospital segment's position, thereby creating a positive duchenne muscular dystrophy drugs market outlook. For instance, in May 2025, Sarepta Therapeutics reported that its ENDEAVOR study participants aged two years at treatment showed mean micro-dystrophin expression of 93.87 percent of normal levels in hospital-administered ELEVIDYS infusions.
Regional Analysis:
To get more information on the regional analysis of this market Request Sample
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America, accounting for 35% of the share, enjoys the leading position in the market. The region's dominance is attributed to its advanced healthcare infrastructure, strong pharmaceutical research and development ecosystem, and favorable regulatory environment that expedites the approval of novel DMD therapies through orphan drug designations and accelerated review pathways. Comprehensive insurance coverage systems and robust reimbursement frameworks in the United States and Canada ensure widespread patient access to high-cost treatments, including gene therapies and specialty pharmaceuticals. The presence of leading biotechnology companies and research institutions dedicated to neuromuscular disease innovation further strengthens the regional market. For instance, Sarepta Therapeutics reported net product revenue of USD 370 million for the third quarter of 2025, driven by strong commercial uptake of its DMD portfolio including ELEVIDYS gene therapy and three approved exon skipping treatments across North America.
Key Regional Takeaways:
United States Duchenne Muscular Dystrophy Drugs Market Analysis
The United States represents the largest individual market for DMD therapies globally, underpinned by a sophisticated healthcare system, extensive clinical trial infrastructure, and high rates of disease awareness and early diagnosis. The regulatory landscape in the United States is particularly conducive to rare disease drug development, with the FDA offering multiple incentive mechanisms including orphan drug exclusivity, priority review vouchers, and breakthrough therapy designations that accelerate the path from clinical development to market authorization. The country's well-developed private insurance market and government healthcare programs provide substantial coverage for specialty pharmaceuticals, enabling patient access to therapies that carry annual costs ranging from several hundred thousand to over three million dollars per treatment. For instance, approximately 15,000 individuals in the United States are currently living with DMD, creating a concentrated patient population that drives sustained therapeutic demand and innovation investment. The robust presence of leading DMD-focused companies, extensive neuromuscular care networks, and ongoing expansion of newborn screening programs further reinforce the United States as the primary driver of market expansion within the region.
Europe Duchenne Muscular Dystrophy Drugs Market Analysis
Europe represents a significant market for DMD therapeutics, supported by strong regulatory frameworks, well-funded public healthcare systems, and a growing emphasis on rare disease research across the continent. The European Medicines Agency is in the position of centralizing access to novel DMD treatments by conditional marketing authorities and orphan medicinal product designation to stimulate pharmaceutical development. The major markets include countries like Germany, the United Kingdom, France, and Italy, and each of them possesses specialized expertise in managing neuromuscular diseases and research in clinical research. Early DMD cases are being enhanced by national screening programs and the genetic diagnostic capacity of European states, which facilitates prompt initiation of treatment. The cooperative research climate in the continent, facilitated by Telethon and other national rare disease associations' initiatives, remains in progress to increase the therapeutic pipeline and clinical care of DMD patients.
Asia-Pacific Duchenne Muscular Dystrophy Drugs Market Analysis
Asia-Pacific is emerging as a rapidly growing market for DMD therapeutics, driven by improving healthcare infrastructure, increasing disease awareness, and expanding access to advanced treatment modalities across the region. Japan stands at the forefront of DMD innovation in the Asia-Pacific, with an efficient regulatory framework and strong government support for rare disease management. Countries including China, India, South Korea, and Australia are witnessing greater diagnostic capabilities and rising adoption of specialty pharmaceuticals for neuromuscular conditions. For instance, in September 2025, the Ministry of Health, Labour and Welfare in Japan granted Orphan Drug designation for DYNE-251 by Dyne Therapeutics, intended for individuals with DMD carrying mutations amenable to exon 51 skipping. Government-led initiatives to integrate genomic medicine into clinical practice and expand clinical trial participation across the region are expected to accelerate market development.
Latin America Duchenne Muscular Dystrophy Drugs Market Analysis
Latin America represents a growing market for DMD therapeutics as healthcare systems across the region progressively expand access to specialized treatments for rare neuromuscular diseases. Countries such as Brazil and Mexico are leading regional efforts to improve diagnostic infrastructure and therapeutic availability for DMD patients. Growing government focus on rare disease frameworks and the development of specialized reference centers for neuromuscular disorders are enhancing early diagnosis and comprehensive patient management. In Latin America, efforts to strengthen disease awareness, expand screening initiatives, and streamline referral pathways are helping reduce diagnostic delays and improve access to appropriate care for individuals affected by Duchenne muscular dystrophy.
Middle East and Africa Duchenne Muscular Dystrophy Drugs Market Analysis
The Middle East and Africa region presents emerging opportunities for DMD drug market development, driven by gradually improving healthcare infrastructure and rising awareness of genetic neuromuscular disorders. Several Gulf Cooperation Council nations are investing in advanced medical facilities and genetic diagnostic capabilities to address rare diseases including DMD. Strategic collaborations between regional healthcare providers and international research institutions are promoting knowledge exchange and strengthening clinical capabilities. These partnerships support improved diagnostic standards, training programs, and access to specialized care. Expanding awareness and treatment infrastructure is particularly important in underserved areas of the Middle East and Africa, where enhancing early detection and comprehensive management of Duchenne muscular dystrophy remains a growing priority.
Competitive Landscape:
The competitive landscape of the DMD drugs market is characterized by intense research activity and strategic collaborations among pharmaceutical and biotechnology companies focused on advancing novel therapeutic modalities. Key market participants are investing substantially in gene therapy platforms, exon skipping technologies, and nonsteroidal anti-inflammatory agents to address the significant unmet medical needs of DMD patients. Companies are pursuing diverse strategies including portfolio expansion through pipeline development, regulatory milestone achievements across multiple geographies, and commercial partnerships to enhance market penetration. The competitive environment is further shaped by the high cost of advanced therapies, evolving reimbursement frameworks, and ongoing safety monitoring requirements that influence market positioning. Strategic acquisitions, licensing agreements, and collaborative research programs with academic institutions and patient advocacy organizations continue to drive innovation and competitive differentiation across the market.
The report provides a comprehensive analysis of the competitive landscape in the duchenne muscular dystrophy drugs market with detailed profiles of all major companies, including:
- FibroGen Inc.
- Italfarmaco S.p.A.
- NS Pharma Inc. (Nippon Shinyaku Co. Ltd.)
- PTC Therapeutics Inc.
- Santhera Pharmaceuticals
- Sarepta Therapeutics Inc.
Latest News and Developments:
- In February 2026, Cumberland Pharmaceuticals Inc., a specialty drug developer dedicated to therapies for rare conditions, announced that the US Food and Drug Administration (FDA) had awarded Fast Track Designation to its investigational oral treatment. The therapy is designed to address a life-threatening cardiac complication associated with Duchenne muscular dystrophy (DMD), supporting its accelerated development and regulatory review pathway.
- In December 2025, Upsher-Smith Laboratories introduced a new corticosteroid therapy for patients in the United States aged five years and older who are living with Duchenne muscular dystrophy (DMD). Marketed under the brand name Kymbee, the product is an oral tablet formulation of deflazacort. Deflazacort is a commonly prescribed corticosteroid for DMD and is also sold under other brand names, including Emflaza.
Duchenne Muscular Dystrophy Drugs Market Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Product Types Covered |
|
| Therapeutic Approaches Covered | Mutation Suppression, Exon Skipping, Steroid Therapy |
| End Users Covered | Hospitals, Clinics, Home Care Settings |
| Regions Covered | North America, Asia-Pacific, Europe, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, China, Japan, India, South Korea, Australia, Indonesia, Germany, France, United Kingdom, Italy, Spain, Russia, Brazil, Mexico |
| Companies Covered | FibroGen Inc., Italfarmaco S.p.A., NS Pharma Inc. (Nippon Shinyaku Co. Ltd.), PTC Therapeutics Inc., Santhera Pharmaceuticals, Sarepta Therapeutics Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC's report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the duchenne muscular dystrophy drugs market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global duchenne muscular dystrophy drugs market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's Five Forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the Duchenne Muscular Dystrophy Drugs industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Frequently Asked Questions About the Duchenne Muscular Dystrophy Drugs Market Report
The duchenne muscular dystrophy drugs market was valued at USD 2.7 Billion in 2025.
The duchenne muscular dystrophy drugs market is projected to exhibit a CAGR of 8.90% during 2026-2034, reaching a value of USD 6.0 Billion by 2034.
The duchenne muscular dystrophy drugs market is driven by the rising prevalence of DMD, advances in gene therapy and exon skipping technologies, favorable regulatory pathways including orphan drug designations, increased government and private funding for rare disease research, expanding newborn screening programs, and growing adoption of novel corticosteroid alternatives with improved safety profiles.
North America currently dominates the duchenne muscular dystrophy drugs market, accounting for a share of 35%. The region's leadership is supported by advanced healthcare infrastructure, strong regulatory incentives for rare disease therapies, robust pharmaceutical research ecosystems, and comprehensive insurance coverage facilitating patient access to innovative treatments.
Some of the major players in the duchenne muscular dystrophy drugs market include FibroGen Inc., Italfarmaco S.p.A., NS Pharma Inc. (Nippon Shinyaku Co. Ltd.), PTC Therapeutics Inc., Santhera Pharmaceuticals, Sarepta Therapeutics Inc., etc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












