
Elastomer Infusion Pump Market Size, Share, Trends and Forecast by Product, Application, End User, and Region, 2026-2034
Elastomer Infusion Pump Market Size and Share:
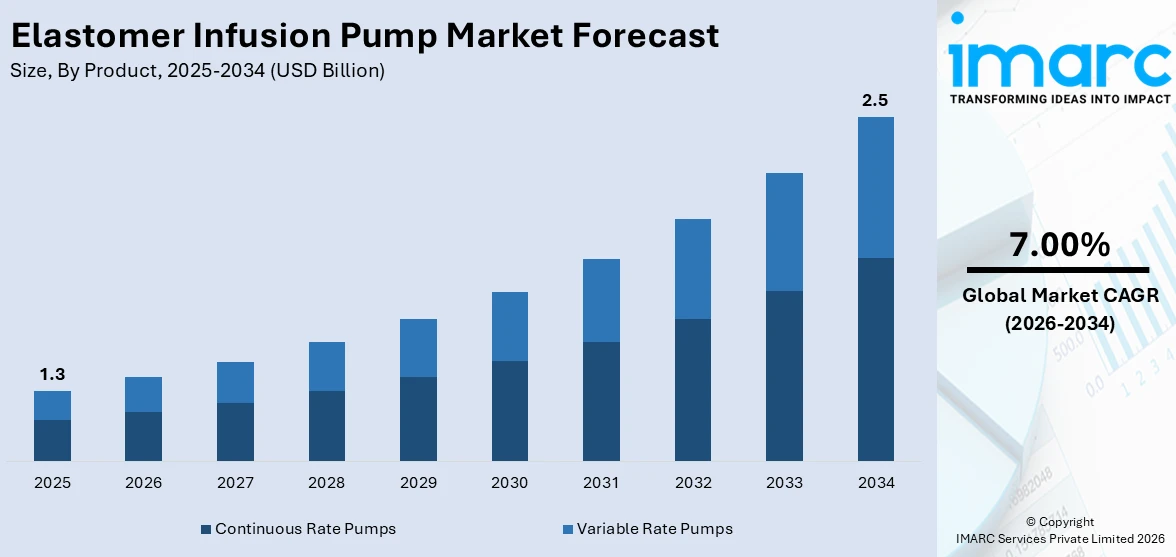
The global elastomer infusion pump market size was valued at USD 1.3 Billion in 2025. The market is projected to reach USD 2.5 Billion by 2034, exhibiting a CAGR of 7.00% from 2026-2034. North America currently dominates the market, holding a market share of over 41.7% in 2025. The market is fueled by intensified patient-focused care demand, home therapy, and accurate drug delivery systems. Expanding use in pain treatment, post-surgery care, and treatment of chronic diseases, along with technology advancements and emphasis on treatment efficiency and safety, are also driving adoption. Portable, easy-to-use, and disposable infusion pumps are becoming a necessity in hospitals, clinics, and home care. All of these factors together are adding to the growth of the elastomer infusion pump market share.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025 |
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
| Market Size in 2025 | USD 1.3 Billion |
| Market Forecast in 2034 | USD 2.5 Billion |
| Market Growth Rate (2026-2034) | 7.00% |
The growing worldwide emphasis on efficient and sustainable healthcare solutions is serving as an important driving force behind the implementation of improved medical devices and drug delivery systems. Governments and healthcare systems globally, across regions, are placing greater focus on innovations that improve patient safety, reduce the chance of cross-contamination risks, and maximize treatment efficiency. There is increasing recognition among healthcare providers for the significance of proper administration of medications and minimizing errors, which is inducing adoption of precision-based infusion devices. According to the sources, in April 2025, B. Braun's introduction of the Spaceplus and elastomeric infusion pump systems to the U.S. reinforced the elastomer infusion pump market, improving accuracy, safety, and clinical effectiveness. Moreover, the emerging trend toward patient-centric care models, such as home therapy and outpatient services, is driving demand for portable and user-friendly devices that are capable of controlled and consistent drug delivery. The focus on enhancing treatment effectiveness and decreasing resource utilization helps meet global healthcare priorities, stimulating investment in cutting-edge infusion solutions. This confluence of regulation, professional interest, and patient-oriented care is propelling strong market growth globally and determining the course of worldwide medical device adoption.
To get more information on this market Request Sample
In the United States, the market for elastomer infusion pumps with 87.50% share in 2024 is growing robustly, fueled by boosting demand for personalized care and patient-centered treatment protocols. Healthcare professionals are placing more and more importance on customized treatment regimens that require accurate and consistent drug delivery technology. The increasing attention to patient safety, convenience, and comfort has further spurred the use of portable and easy-to-use infusion devices both in clinical settings and home care. Furthermore, the United States healthcare is transitioning towards outpatient treatments and home therapies as means to decrease hospitalizations, maximize the utilization of resources, and improve patient satisfaction. Medical technology breakthroughs, changing models of care, and increasing expectations for the accuracy of treatment are all solidifying the incorporation of elastomeric infusion systems. Combining patient-centered care and clinical effectiveness, the United States remains a growth area of prime importance for sophisticated infusion devices, with uptake forecast to grow steadily.
Elastomer Infusion Pump Market Trends:
Rising Incidence of Cancer Driving Chemotherapy Market
Increasing incidence of cancer is largely propelling the demand for chemotherapy, driven by physical inactivity, environmental pollution, and worldwide consumption of tobacco and alcohol. In the United States alone, 2025 will witness 2,041,910 new cancer diagnoses and 618,120 fatalities, indicating the pressing need for effective therapeutic options. Worldwide, urbanization, lifestyle, and environmental factors are also responsible for increasing cancer rates, thus creating a priority for chemotherapy among healthcare professionals. Sophisticated drug delivery systems, especially elastomeric infusion pumps, are indispensable to guarantee treatment precision, safety, and patient comfort. They enable intermittent and continuous drug delivery, eliminating errors in dosing and enhancing therapy consistency. In the context of elastomer infusion pump market trends, growing up-take of safe, reliable, and patient-centric infusion solutions is apparent, indicative of the greater significance of these technologies to oncology treatment, particularly in outpatient and home-based treatment environments where convenience and accuracy are of greatest concern.
Geriatric Population Amplifying Treatment Needs
The world geriatric population is growing fast, with those over 65 years set to reach 2.2 billion in the late 2070s, outnumbering the population of persons under 18 years. Aging also doubles cancer risk when cellular repair efficiency is lost, leading to amplified rates of chronic disease. As such, elderly patients increasingly need frequent chemotherapy and supportive therapy. Single-infusion elastomeric pumps that are disposable are especially useful in preventing cross-contamination risks and delivering safe, precise drug therapy, particularly for home-care therapy. The accelerating demand for patient-focused and minimally invasive treatment methods further underscores the importance of advanced infusion systems. In line with market trends, the confluence of a growing elderly population and greater emphasis on safe, effective, and patient-friendly therapies is propelling consistent elastomer infusion pump market growth in the U.S. Sophisticated infusion equipment is becoming integral to handling geriatric treatment needs, providing both convenience and treatment accuracy in contemporary healthcare service delivery.
Home-Based Care and Post-Operative Pain Management
The growth in surgical treatments, especially orthopedic and post-operative treatments, is driving demand for effective pain management systems. Elastomeric infusion pumps ensure controlled single-infusion administration of analgesics, eliminating dosing errors and cross-contamination hazards to patients and healthcare personnel. Hospitals are also pushing home care more and more to decrease inpatient stays, enhance patient comfort, and better handle mounted patient volumes. Portable, light, and user-friendly elastomeric pumps are replacing conventional electric infusion systems in outpatient as well as inpatient environments. The trend towards outpatient and home-oriented therapy, along with increasing focus on safety, convenience, and treatment efficacy, is supporting adoption of these systems. In accordance with trends in the market, healthcare providers are seeking consistent, patient-compliant, and accurate drug-delivery solutions. This trend is playing an important role in the development of elastomeric infusion pump use in the U.S., especially for post-operative treatment and home therapy programs.
Elastomer Infusion Pump Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global elastomer infusion pump market, along with forecast at the global, regional, and country levels from 2026-2034. The market has been categorized based on product, application, and end user.
Analysis by Product:
- Continuous Rate Pumps
- Variable Rate Pumps
Continuous rate pumps are a prominent segment in the elastomer infusion pump market outlook with a share of 59.2% in 2025, as they are capable of delivering drugs with high precision and uniformity over a prolonged duration. The pumps are commonly used in clinical environments as they provide controlled drug delivery and minimize the risk of dosing errors, which helps in enhancing patient outcomes. Their ability to deliver continuous infusion without the need for repeated adjustments makes them ideal for critical therapies, such as chemotherapy, antibiotics, and pain relief. Continuous rate pumps are also very compatible with hospital and home-care settings, with portability and convenience for patients needing treatment over long periods or as outpatients. Further, improvements in elastomer technology have increased reliability and safety in these pumps, reducing contamination risks and ease of operation. The emphasis on patient-driven care, in addition to the demand for efficient and easy medication administration, is fueling growth and acceptance of continuous rate pumps through worldwide healthcare systems.
Analysis by Application:
Access the comprehensive market breakdown Request Sample
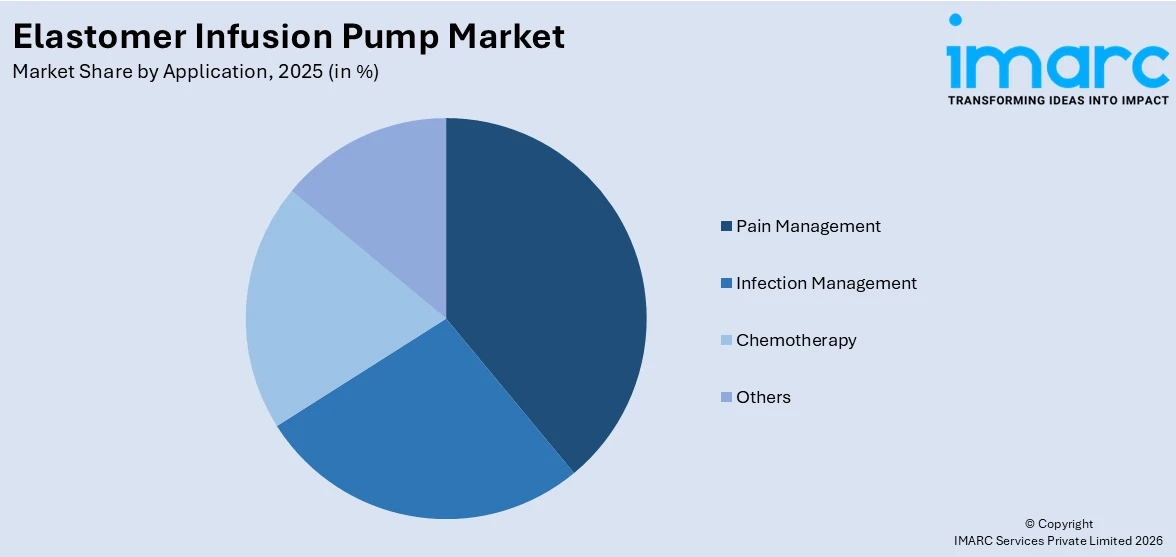
- Pain Management
- Infection Management
- Chemotherapy
- Others
Pain management is one of the most important uses propelling the elastomer infusion pump market with a market share of share of 39.8% in 2025. The equipment provides accurate, controlled analgesic delivery, delivering effective post-operative and chronic pain management with decreased dependence on oral drugs. Elastomer infusion pumps are especially valuable in post-surgical care, allowing healthcare professionals to control pain effectively and customize treatment for unique patient requirements. Their portability enables patients to continue therapy in home-care or outpatient locations, enhancing comfort and compliance. Single-use, disposable designs further minimize infection risks, which is especially important in pain management situations that frequently involve extended infusion. With hospitals and clinics increasingly focusing on patient-centered strategies and improved recovery protocols, elastomer infusion pumps have become a critical component of successful pain control therapies. The heightened emphasis on quality improvement, shorter hospital stays, and fewer complications highlights the role of pain management as the primary growth driver for this segment.
Analysis by End User:
- Hospitals and Clinics
- Ambulatory Care
- Others
According to elastomer infusion pump market analysis, in 2025 with a share of 47.6%, hospitals and clinics are a prevalent end-user segment of the market because these institutions have a great need for efficient and effective drug delivery systems. Hospitals and clinics provide various types of therapies, such as chemotherapy, antibiotics, and pain control, that necessitate continuous and accurate infusion. Elastomer pumps have numerous benefits in healthcare settings, such as ease of portability, simplicity of use, and lower cross-contamination risks due to their disposable nature. Furthermore, they aid outpatient and home-care programs, consistent with contemporary patient-focused treatment models and hospital efficiency goals. The growing volume of surgical procedures and chronic disease management also stimulates demand for infusion solutions within hospitals and clinics. With healthcare facilities concentrating on enhancing the quality of treatment, reducing errors, and maximizing workflow efficiency, elastomer infusion pumps became a critical tool. Their broad acceptance guarantees precise, safe, and easy therapy administration in all clinical settings, making this segment a key market driver.
Regional Analysis:
To get more information on the regional analysis of this market Request Sample
- North America
- United States
- Canada
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
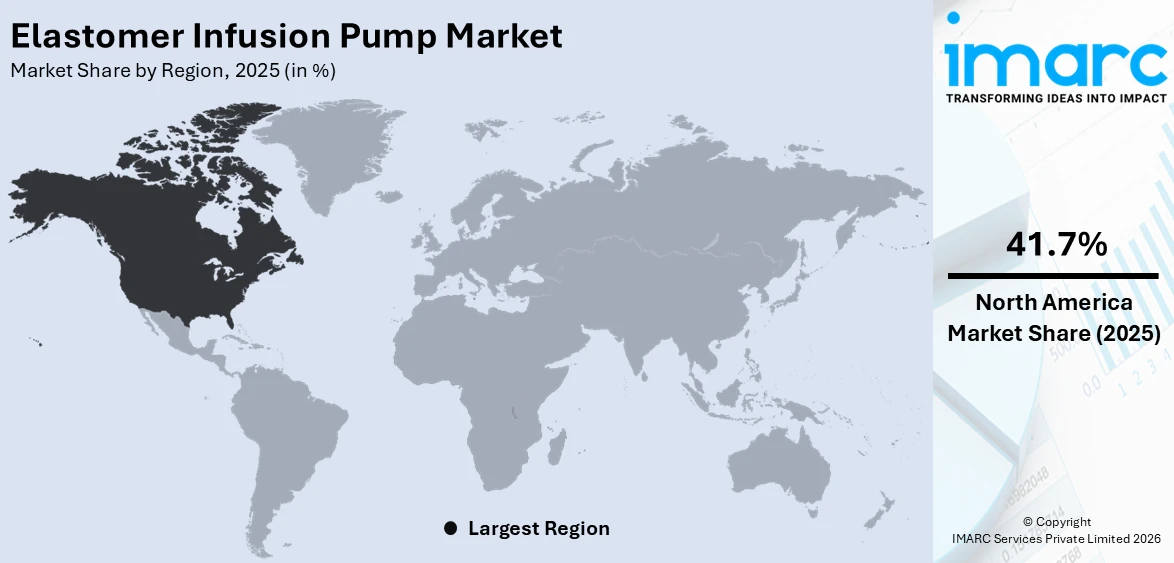
North America is a key region in the elastomer infusion pump market with a share of share of 41.7% in 2025, boosting from advanced healthcare infrastructure, heightening patient awareness, and use of advanced drug delivery systems. The concentration of the region on patient-focused care, home-based therapy, and outpatient care has enhanced demand for user-friendly, portable infusion devices. Clinics and hospitals in North America increasingly opt for elastomer pumps due to their reliability, safety, and potential for lowering cross-contamination, especially in post-operative care and pain management. Regulatory endorsement, technological innovations, and strong healthcare spending also underpin broad usage throughout the U.S. and Canada. North America is also reinforced by an expanding elderly population that demands more long-term therapies demanding precise infusion solutions. Ongoing efforts to drive treatment efficiency, patient safety, and comfort place the region at the forefront of a critical market for elastomer infusion pumps. The convergence of innovation, infrastructure, and shifting care models guarantees consistent growth and leadership of North America in this market.
Key Regional Takeaways:
United States Elastomer Infusion Pump Market Analysis
United States is witnessing increasing elastomer infusion pump adoption due to shifting preferences towards home-based care. For instance, every year, approximately 12 Million Americans receive home health care services. As patients seek convenient and cost-effective treatment options, home-based care is becoming a favorable alternative to hospital stays, especially for post-operative and chronic pain management. This shift is driving demand for portable, user-friendly medical devices like elastomer infusion pumps, which offer continuous drug delivery without the need for electronic components. Advancements in outpatient care models and an aging population preferring in-home recovery are further boosting adoption. Healthcare providers are also encouraging early discharge strategies that support self-administration therapies, enhancing the demand for such infusion solutions.
Asia Pacific Elastomer Infusion Pump Market Analysis
Asia-Pacific is experiencing increased elastomer infusion pump usage due to the rising number of orthopedic procedures and surgeries. For instance, the India orthopedic devices market size was valued at USD 2.62 Billion in 2024. Looking forward, IMARC Group estimates the market to reach USD 4.53 Billion by 2033, exhibiting a CAGR of 5.80% from 2025-2033. The surge in musculoskeletal conditions, road injuries, and sports-related trauma has led to higher orthopedic interventions, demanding effective pain management tools. Elastomer infusion pumps, offering reliable, continuous drug delivery, are becoming vital in post-surgical recovery. Growing investment in healthcare infrastructure and the expansion of surgical facilities across this region are further strengthening the market. Healthcare professionals are favoring these devices for their portability and ease of use in outpatient settings.
Europe Elastomer Infusion Pump Market Analysis
Europe is registering growing adoption of elastomer infusion pumps owing to the rising geriatric population. According to WHO, the population aged 60 and older is rapidly growing in the WHO European Region. In 2021, there were 215 Million; by 2030, it is projected to be 247 Million, and by 2050, over 300 Million. The elderly demographic is more susceptible to chronic diseases, surgeries, and long-term pain, necessitating reliable infusion solutions. Elastomer infusion pumps, known for their simplicity and reduced risk of complications, are ideal for home and hospital-based elderly care. Healthcare systems are focusing on aging-related care models that support non-invasive, continuous drug delivery solutions. With an emphasis on improving quality of life for older patients, demand for user-friendly infusion options is increasing.
Latin America Elastomer Infusion Pump Market Analysis
Latin America shows rising elastomer infusion pump adoption due to boosting cancer cases. For instance, in Brazil, the National Cancer Institute (INCA) estimates that, for each year of the 2023-2025 triennium, 704 thousand new cancer cases have occurred. The growing oncology burden demands efficient and continuous drug delivery for pain management and chemotherapy. Elastomer infusion pumps provide a non-electric, portable option for delivering analgesics and anti-cancer drugs. With limited healthcare access in some areas, these pumps support outpatient and home-based cancer care, enhancing patient comfort.
Middle East and Africa Elastomer Infusion Pump Market Analysis
Middle East and Africa is witnessing increasing elastomer infusion pump adoption due to growing healthcare facilities. For instance, in 2025, the UAE is currently home to over 150 hospitals and has more than 5,000 healthcare facilities. As medical infrastructure expands, demand for effective post-surgical and chronic pain management tools is increasing. Elastomer infusion pumps offer cost-effective, easy-to-use solutions for hospitals and outpatient centers. Healthcare investments and the development of specialized care units are further driving the usage of these devices for patient-controlled infusion therapy.
Competitive Landscape:
The market competition of elastomer infusion pumps is defined by ongoing innovation, technological developments, and strategic action to improve product performance and availability. Major players are working on developing portable, user-friendly, and disposable infusion systems in order to address the growing need for patient-centered care, home therapy, and outpatient services. Market outlook suggests continued growth led by intensifying adoption of elastomeric infusion solutions by hospitals, clinics, and home-care facilities. Focusing on enhancing accuracy of treatment, reducing cross-contamination, and increasing patient comfort is driving product development initiatives and prompting larger market penetration. Cooperation among healthcare professionals, technology developers, and regulatory agencies is also supporting developments in pump design, material safety, and infusion efficiency. With the elastomer infusion pump market forecast anticipating steady growth in the years to come, innovation, reliability, and flexibility are becoming the main drivers shaping the competitive landscape, allowing businesses to take advantage of established and rising healthcare markets.
The report provides a comprehensive analysis of the competitive landscape in the elastomer infusion pump market with detailed profiles of all major companies, including:
- Avanos Medical Inc.
- B. Braun Melsungen AG
- Baxter International Inc.
- Daiken Medical Co. Ltd.
- Nipro Corporation
- O&M Halyard Inc
- Palex Medical S.A.
- Pfizer Inc. (Hospira Inc.)
- pfm medical ag.
- Samtronic Industria e Comercio LTDA
- Smiths Group plc.
- Woo Young Medical Co. Ltd.
Latest News and Developments:
- April 2025: B. Braun advanced regional anesthesia and infusion therapy by integrating innovations like the ON-Q® elastomer infusion pump, which supported continuous postoperative pain relief, and reinforced its global leadership through AI-enhanced systems, EU MDR compliance, and strategic collaborations.
- April 2025: the Ministry of Health’s Medical Technology Advisory Committee recommended elastomeric infusion pumps for outpatient parenteral antimicrobial therapy, endorsing their use in home or OPAT clinic settings under multidisciplinary supervision and proper patient or caregiver training.
- January 2025: The Centers for Medicare and Medicaid Services (CMS) expanded reimbursement to non-opioid solutions, including the ON-Q* elastomeric infusion pump and ambIT* infusion pump, under the NOPAIN Act. Effective January 1, 2025, both elastomer infusion pumps qualified for separate payment, marking them as the only pain pumps covered by the policy.
- January 2025: SMARTeZ® disposable elastomeric infusion pumps emerged as a safe, accurate, and maintenance-free alternative to electronic and ambulatory pump sets, offering enhanced drug stability, broader fill volumes, and superior accuracy for home, chemotherapy, and antibiotic therapies. These pumps supported greater patient mobility and reduced ownership costs with zero maintenance, calibration, or alarm issues.
- January 2025: The Accufuser range of elastomer infusion pumps, now available in NRFit and listed on the TGA Prescribed List, was launched to offer accurate, single-use solutions for home or hospital-based chemotherapy, antibiotics, and pain medication. Featuring selectable flow rates, built-in filters, and safety mechanisms, these pumps enabled patient mobility without electronic devices.
Elastomer Infusion Pump Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Products Covered | Continuous Rate Pumps, Variable Rate Pumps |
| Applications Covered | Pain Management, Infection Management, Chemotherapy, Others |
| End Users Covered | Hospitals and Clinics, Ambulatory Care, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Avanos Medical Inc., B. Braun Melsungen AG, Baxter International Inc., Daiken Medical Co. Ltd., Nipro Corporation, O&M Halyard Inc, Palex Medical S.A., Pfizer Inc. (Hospira Inc.), pfm medical ag., Samtronic Industria e Comercio LTDA, Smiths Group plc. and Woo Young Medical Co. Ltd. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the elastomer infusion pump market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global elastomer infusion pump market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's Five Forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the elastomer infusion pump industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Key Questions Answered in This Report
The elastomer infusion pump market was valued at USD 1.3 Billion in 2025.
The elastomer infusion pump market is projected to exhibit a CAGR of 7.00% during 2026-2034, reaching a value of USD 2.5 Billion by 2034.
Driving forces for the elastomer infusion pump market are the escalating need for patient-focused and home-based treatment, greater use of portable and ease-of-use infusion devices, greater need for accurate and controlled drug delivery, and increasing applications in pain administration, post-surgical treatment, and treatment of chronic diseases. Market growth is additionally stimulated by technological innovation and the focus on treatment efficiency and safety.
North America currently dominates the elastomer infusion pump market holding a market share of 41.7% in 2025. The market is driven by rising cancer incidence, expanding geriatric population, increasing adoption of home-based care, growing demand for safe and precise drug delivery systems, favorable healthcare infrastructure, advanced technological adoption, and supportive reimbursement policies for infusion therapies.
Some of the major players in the elastomer infusion pump market include Avanos Medical Inc., B. Braun Melsungen AG, Baxter International Inc., Daiken Medical Co. Ltd., Nipro Corporation, O&M Halyard Inc, Palex Medical S.A., Pfizer Inc. (Hospira Inc.), pfm medical ag., Samtronic Industria e Comercio LTDA, Smiths Group plc. and Woo Young Medical Co. Ltd., etc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












