
Formulation Development Outsourcing Market Report by Service (Pre-formulation Services, Formulation Optimization), Dosage Form (Injectable, Oral, Topical, and Others), Application (Oncology, Genetic Disorders, Neurology, Infectious Diseases, Respiratory, Cardiovascular, and Others), End User (Pharmaceutical and Biopharmaceutical Companies, Government and Academic Institutes), and Region 2026-2034
Formulation Development Outsourcing Market Size:
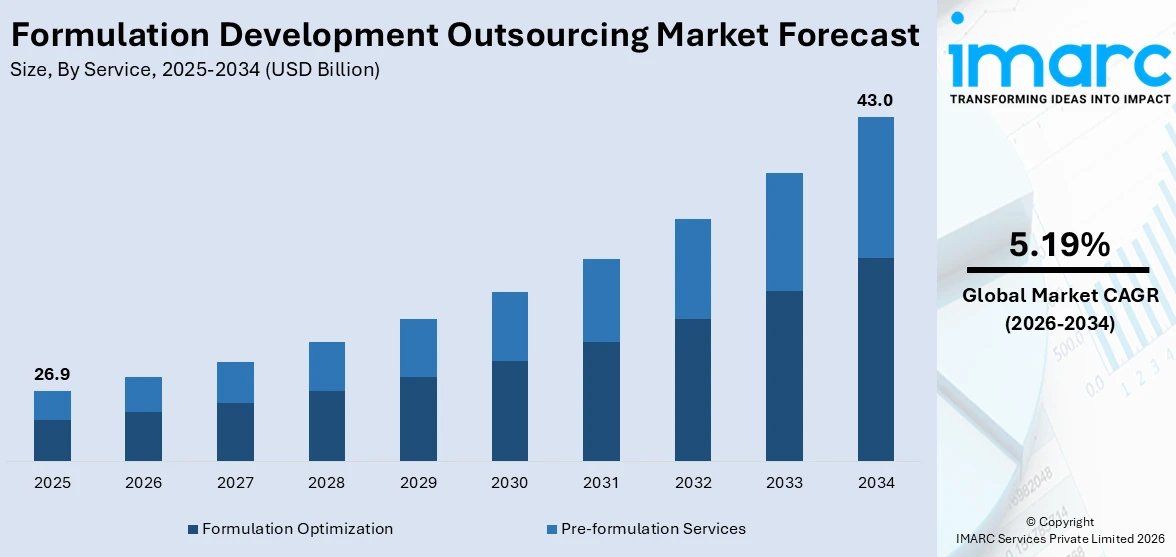
The global formulation development outsourcing market size reached USD 26.9 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 43.0 Billion by 2034, exhibiting a growth rate (CAGR) of 5.19% during 2026-2034. The increasing research and development (R&D) costs, the rising need for specialized expertise, accelerated drug development timelines, growing regulatory complexities, and the growing pharmaceutical sector are some of the major factors propelling the growth of the market.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025 |
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
| Market Size in 2025 | USD 26.9 Billion |
| Market Forecast in 2034 | USD 43.0 Billion |
| Market Growth Rate (2026-2034) | 5.19% |
Formulation Development Outsourcing Market Analysis:
- Major Market Drivers: The rising research and development (R&D) costs and the growing need for specialized expertise represent the major drivers of the market. Pharmaceutical companies increasingly outsource to reduce expenses and access advanced technologies that enhance drug development efficiency.
- Key Market Trends: The widespread adoption of advanced technologies such as artificial intelligence (AI) and machine learning (ML) to optimize drug formulations represents the key trend of the market. The market also experiences a shift toward strategic partnerships and long-term collaborations between pharmaceutical companies and outsourcing providers.
- Geographical Trends: According to the formulation development outsourcing market overview, North America accounts for the largest region in the market. The high research and development (R&D) costs, the rising need for specialized expertise, strict regulatory requirements, and the increasing focus on accelerating drug development are driving the growth of the market across the region.
- Competitive Landscape: Some of the major market players in the formulation development outsourcing industry include Aizant Drug Research Solutions Private Limited, Catalent Inc., Charles River Laboratories, Dr. Reddy’s Laboratories Ltd., Emergent BioSolutions Inc., Intertek Group plc, Irisys LLC (Recro Pharma), Laboratory Corporation of America Holdings, Lonza Group AG, Piramal Pharma Solutions, Quotient Sciences, Syngene International Limited (Biocon Limited), and Thermo Fisher Scientific Inc., among many others.
- Challenges and Opportunities: The market faces various challenges including regulatory compliance complexities, intellectual property concerns, and the need for stringent quality control. However, the market also faces several opportunities such as the increasing demand for advanced drug formulations, the growth of personalized medicine, and technological advancements like artificial intelligence (AI) and machine learning (ML).
To get more information on this market Request Sample
Formulation Development Outsourcing Market Trends:
Rising Research and Development (R&D) Costs
Pharmaceutical companies face high costs in research and development (R&D), prompting them to outsource formulation development to reduce expenses and allocate resources more efficiently. For instance, according to industry reports, Merck & Co’s revenues reached $60.1bn in FY 2023, with research and development (R&D) investments peaking at $30.5bn. Displaying a staggering 126% growth over the course of one year, the company’s research and development (R&D) investments now represent 50.8% of its total revenue for 2023. Similarly, in 2023 Johnson & Johnson (J&J) displayed strong financial growth and was the leading pharmaceutical company in terms of revenues. The pharma giant ranked third in terms of R&D expenditure, investing approximately $15.1bna record-breaking figure for the company. The pharmaceutical industry’s expansion is positively enhancing the formulation development outsourcing market revenue.
Increasing Need for Specialized Expertise
The growing complexity of drug formulations requires specialized expertise and advanced technologies, which many pharmaceutical companies may lack internally. For instance, according to industry reports, Indian pharma’s global presence in generic medicine exports among others like biosimilars will indicate a strategic shift toward further expansion in the international markets, particularly the US. To this end, the guidance from the US FDA on formulation development serves as a crucial enabler for Indian pharma companies in this pursuit. The draft guidance on formulation development outlines the regulatory requirements and best practices for developing drug formulations, especially those intended for the US market. Adhering to these guidelines not only ensures compliance but also enhances the quality and efficacy of pharmaceutical products. According to Indian pharma industry experts, for Indian pharmaceutical firms eyeing global expansion, such guidance provides a roadmap for developing high-quality formulations that meet international standards, thereby bolstering their credibility and competitiveness in the global arena. Outsourcing partners offer the necessary skills and cutting-edge technologies to develop sophisticated drug formulations that enhance the quality and efficacy of pharmaceutical products, thereby creating a positive formulation development outsourcing market outlook.
Growing Regulatory Compliance
Stringent regulatory requirements necessitate comprehensive and compliant formulation processes, which outsourcing partners with regulatory expertise can effectively manage. For instance, according to industry reports, Indian pharmaceutical companies must meet new manufacturing standards in 2024, as per a Union Health Ministry notification, although small companies have asked for a delay, citing their debt load. Under the revised Schedule M guidelines, pharmaceutical companies will have to inform the licensing authority about recalling a drug and report product defects, deterioration, or faulty production. Till now there was no provision for informing the licensing authority about drug recall. This is further expected to facilitate the market growth.
Formulation Development Outsourcing Market Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the market, along with forecasts at the global, regional, and country levels for 2026-2034. Our report has categorized the market based on service, dosage form, application, and end user.
Breakup by Service:
- Pre-formulation Services
- Discovery and Preclinical Services
- Analytical Services
- Formulation Optimization
- Phase I
- Phase II
- Phase III
- Phase IV
Formulation Optimization accounts for the majority of the market share
The report has provided a detailed breakup and analysis of the market based on the service. This includes pre-formulation services (discovery and preclinical services, analytical services) and formulation optimization (Phase I, Phase II, Phase III, and Phase IV). According to the report, formulation optimization represented the largest segment.
The demand for formulation optimization in the market is driven by the need for enhanced drug efficacy, stability, and bioavailability. Increasing regulatory scrutiny necessitates robust and compliant formulations. Additionally, the rise of complex and personalized medicines requires precise formulation techniques. Outsourcing provides access to specialized expertise and advanced technologies, enabling efficient optimization. Moreover, the pressure to accelerate time-to-market and reduce development costs further fuels the demand for optimized formulations in the pharmaceutical industry. For instance, in July 2024, Pfizer announced that it had selected its preferred once-daily modified-release formulation for danuglipron, an oral glucagon-like peptide-1 (GLP-1) receptor agonist. Looking ahead, Pfizer plans to conduct dose optimization studies in the second half of 2024, evaluating multiple doses of the preferred modified release formulation to inform the registration enabling studies.
Breakup by Dosage Form:
- Injectable
- Oral
- Topical
- Others
Injectable holds the largest share of the industry
A detailed breakup and analysis of the market based on the dosage form have also been provided in the formulation development outsourcing market report. This includes injectable, oral, topical, and others. According to the report, injectable accounted for the largest market share.
The demand for injectable dosage forms in the market is driven by the growing prevalence of chronic diseases and the need for targeted and controlled drug delivery. Injectable forms offer rapid absorption and bioavailability, essential for critical care and biologic drugs. Additionally, the rise in biologics and biosimilars, which are predominantly administered via injection, fuels this demand. Outsourcing provides access to specialized manufacturing capabilities and regulatory expertise, ensuring high-quality and compliant injectable formulations. This, in turn, is enhancing the overall formulation development outsourcing market value.
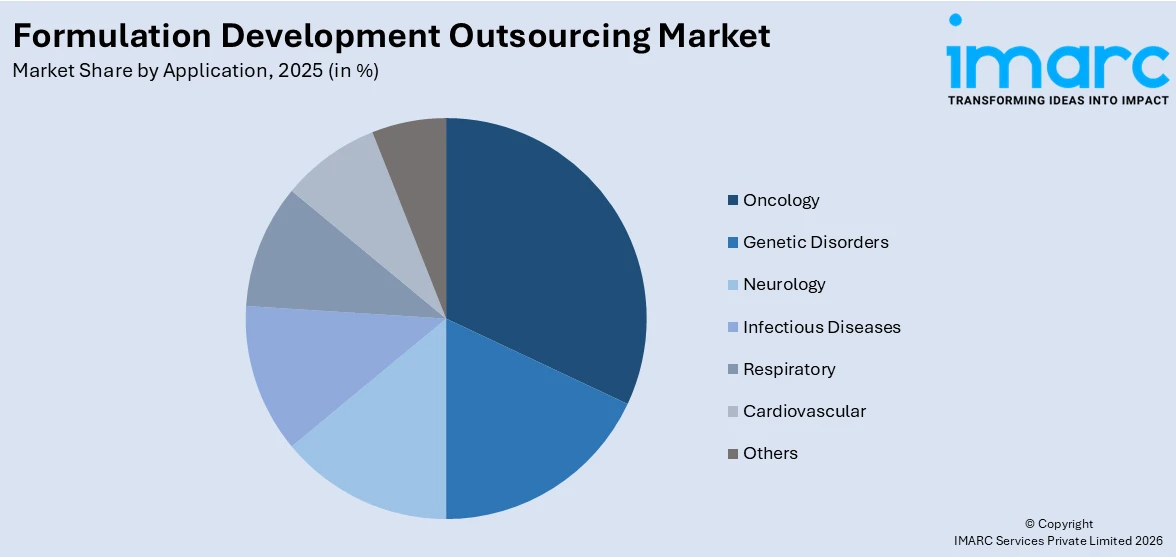
Breakup by Application:
Access the comprehensive market breakdown Request Sample
- Oncology
- Genetic Disorders
- Neurology
- Infectious Diseases
- Respiratory
- Cardiovascular
- Others
Oncology represents the leading market segment
The report has provided a detailed breakup and analysis of the market based on the application. This includes oncology, genetic disorders, neurology, infectious diseases, respiratory, cardiovascular, and others. According to the report, oncology represented the largest segment.
The demand for formulation development outsourcing in oncology is driven by the complexity of cancer treatments and the need for specialized expertise. Advancements in targeted therapies and personalized medicine require innovative formulation techniques. High research and development (R&D) costs and stringent regulatory requirements also necessitate outsourcing to manage these challenges efficiently which is driving the formulation development outsourcing demand. Additionally, the urgency to accelerate time-to-market for new cancer drugs pushes pharmaceutical companies to seek external partners with advanced technologies and experience in oncology formulation development.
Breakup by End User:
- Pharmaceutical and Biopharmaceutical Companies
- Government and Academic Institutes
Pharmaceutical and biopharmaceutical companies exhibits a clear dominance in the market
A detailed breakup and analysis of the market based on the end user have also been provided in the report. This includes pharmaceutical and biopharmaceutical companies and government and academic institutes. According to the report, pharmaceutical and biopharmaceutical companies accounted for the largest market share.
The demand for formulation development outsourcing in pharmaceutical and biopharmaceutical companies is driven by high research and development (R&D) costs, the need for specialized expertise, and advanced technologies. Outsourcing helps accelerate drug development timelines and ensures regulatory compliance. The complexity of new drug formulations, including biologics and personalized medicines, necessitates external expertise which is further influencing formulation development outsourcing market growth positively. Additionally, outsourcing allows companies to focus on core competencies while leveraging innovative solutions from specialized partners to enhance drug efficacy, stability, and market readiness.
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America leads the market, accounting for the largest formulation development outsourcing market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America represents the largest regional market for formulation development outsourcing.
The high research and development (R&D) costs and the need for specialized expertise to develop complex drug formulations are driving the market growth in the region. The region's stringent regulatory environment necessitates compliance with rigorous standards, which outsourcing partners can manage efficiently. Additionally, the rise of biotechnology and personalized medicine requires advanced formulation solutions, further boosting the demand for outsourcing. According to the formulation development outsourcing market forecast across the region, the pressure to accelerate drug development timelines and reduce time-to-market also contributes to the growth of this market. For instance, in May 2024, Coriolis Pharma, a globally operating service provider for R&D and GMP drug product development, R&D and GMP analytics, and sterile drug product manufacturing support, announced the launch of a U.S. entity that allows customers to combine the convenience of a local service with the advantages of international expertise. U.S. customers will be served by the U.S.-based Coriolis commercial organization, which consists of business development and client project management professionals.
Competitive Landscape:
- The market research report has also provided a comprehensive analysis of the competitive landscape in the market. Detailed profiles of all major companies have also been provided. Some of the major market players in the formulation development outsourcing industry include Aizant Drug Research Solutions Private Limited, Catalent Inc., Charles River Laboratories, Dr. Reddy’s Laboratories Ltd., Emergent BioSolutions Inc., Intertek Group plc, Irisys LLC (Recro Pharma), Laboratory Corporation of America Holdings, Lonza Group AG, Piramal Pharma Solutions, Quotient Sciences, Syngene International Limited (Biocon Limited) and Thermo Fisher Scientific Inc.
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
- The market for formulation development outsourcing is highly competitive, with numerous players offering specialized services. These formulation development outsourcing companies include Catalent, Lonza, and Charles River Laboratories, known for their extensive expertise and advanced technologies. Emerging firms and niche providers also compete by offering tailored solutions and innovative approaches. For instance, in April 2024, Charles River Laboratories launched its Alternative Methods Advancement Project (AMAP), an initiative to develop alternatives to reduce animal testing and to drive a new standard for drug discovery and development. The company’s initial investment was $200 million over the past four years and its five-year goal is to invest an additional $300 million. This investment spans a portfolio of technology innovations, partnerships, and advocacy efforts to reduce the use of animal testing.
Formulation Development Outsourcing Market News:
- In February 2023, Quotient Sciences, the drug development and manufacturing accelerator, announced that it had completed an expansion of its early-phase formulation development capabilities for oral dosage forms at its Nottingham, UK, facility. The expanded services build upon the site’s existing formulation capabilities and increase capacity to support fully integrated drug development programs through the company’s flagship platform, Translational Pharmaceutics.
- In May 2024, AGC Biologics, a CDMO, and BioConnection, a CMO, agreed to partner to provide end-to-end biopharmaceutical development and manufacturing capabilities for drug substance and drug products using the development and manufacturing expertise of AGC Biologics and the specialized aseptic filling capabilities of BioConnection.
- In October 2023, Lonza, a global manufacturing partner to the pharmaceutical, biotech, and nutraceutical markets, announced the extension of a collaboration with a major biopharmaceutical partner for the commercial-scale filling of ADCs.
Formulation Development Outsourcing Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Services Covered |
|
| Dosage Forms Covered | Injectable, Oral, Topical, Others |
| Applications Covered | Oncology, Genetic Disorders, Neurology, Infectious Diseases, Respiratory, Cardiovascular, Others |
| End Users Covered | Pharmaceutical and Biopharmaceutical Companies, Government and Academic Institutes |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Aizant Drug Research Solutions Private Limited, Catalent Inc., Charles River Laboratories, Dr. Reddy’s Laboratories Ltd., Emergent BioSolutions Inc., Intertek Group plc, Irisys LLC (Recro Pharma), Laboratory Corporation of America Holdings, Lonza Group AG, Piramal Pharma Solutions, Quotient Sciences, Syngene International Limited (Biocon Limited), Thermo Fisher Scientific Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the formulation development outsourcing market from 2020-2034.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the global formulation development outsourcing market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the formulation development outsourcing industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Frequently Asked Questions About the Formulation Development Outsourcing Market Report
The formulation development outsourcing market was valued at USD 26.9 Billion in 2025.
The formulation development outsourcing market is projected to exhibit a CAGR of 5.19% during 2026-2034, reaching a value of USD 43.0 Billion by 2034.
The market is driven by the increasing demand for cost-effective and time-efficient drug development solutions, the growing complexity of drug formulations, and the need for specialized expertise. Additionally, the rising adoption of contract research organizations (CROs), regulatory pressure, and the focus on accelerating time-to-market contribute to the market's growth.
North America currently dominates the formulation development outsourcing market in 2025. The dominance is fueled by the presence of top pharmaceutical firms and cutting-edge healthcare infrastructure, high R&D spending, and the growing trend of outsourcing by pharmaceutical firms. Additionally, favorable regulatory policies and access to top-tier talent drive market growth in the region.
Some of the major players in the formulation development outsourcing market include Aizant Drug Research Solutions Private Limited, Catalent Inc., Charles River Laboratories, Dr. Reddy’s Laboratories Ltd., Emergent BioSolutions Inc., Intertek Group plc, Irisys LLC (Recro Pharma), Laboratory Corporation of America Holdings, Lonza Group AG, Piramal Pharma Solutions, Quotient Sciences, Syngene International Limited (Biocon Limited), and Thermo Fisher Scientific Inc., among others.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












