
Hernia Repair Market Report by Product Type (Hernia Mesh, Hernia Fixation Devices), Surgery Type (Open Tension Repair Surgery, Tension-Free Repair Surgery, Laparoscopic Surgery), Hernia Type (Inguinal Hernia, Ventral Hernia, Femoral Hernia, Umbilical Hernia, Hiatal Hernia, and Others), End-User (Ambulatory Surgery Centers, Hospitals and Clinics, and Others), and Region 2026-2034
Hernia Repair Market Size:
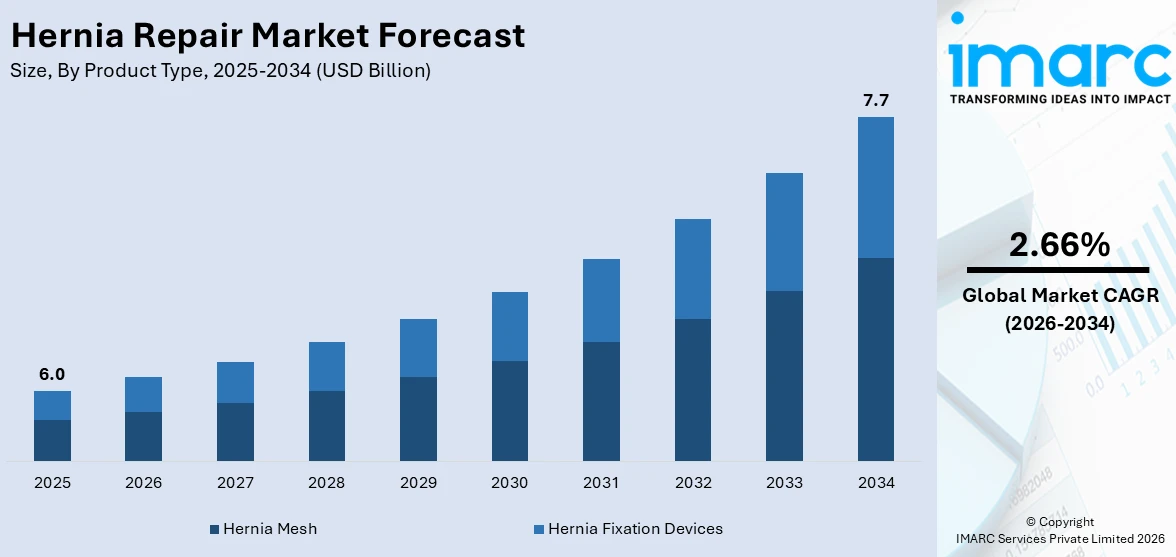
The global hernia repair market size reached USD 6.0 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 7.7 Billion by 2034, exhibiting a growth rate (CAGR) of 2.66% during 2026-2034. The market is primarily driven by the escalating prevalence of hernia cases worldwide, technological advancements in surgical techniques, increasing healthcare investments, growing awareness about hernia treatments, and rising adoption of advanced treatment options.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 6.0 Billion |
|
Market Forecast in 2034
|
USD 7.7 Billion |
| Market Growth Rate 2026-2034 | 2.66% |
Hernia Repair Market Analysis:
- Major Market Drivers: Some of the major factors driving the market include growing geriatric population, rising awareness about treatment alternatives, and numerous technological innovations. These factors are creating a positive hernia repair market overview.
- Key Market Trends: Abdominal core health is becoming more and more important, with an emphasis on physical therapy, diet, and exercise to strengthen core muscles and avoid hernias. Additionally, the growing trend toward minimally invasive procedures, such as laparoscopic and robotic-assisted surgeries is propelling the hernia repair market demand.
- Geographical Trends: North America dominates the market due to the increasing rates of new technology adoption, extensive treatment research and development, and the presence of vital key factors in the region. Europe is the second largest market driven by the growing elderly population and sedentary lifestyles.
- Competitive Landscape: Some of the major market players in the hernia repair industry include B.Braun Melsungen Aktiengesellschaft, Baxter International Inc., BD (Becton, Dickinson and Company), Cook Medical Inc., CooperSurgical Inc., Ethicon Inc. (Johnson & Johnson), Herniamesh S.r.l., Lifecell Corporation (Allergan Plc), Maquet (Getinge), Medtronic Inc., Olympus Corporation, Via Surgical Ltd., W. L. Gore & Associates Inc. among many others.
- Challenges and Opportunities: Opportunities for key players in the industry include increasing hernia prevalence. Challenges include the unavailability of expertise in laparoscopic surgery.
To get more information on this market Request Sample
Hernia Repair Market Trends:
Rising Geriatric Population
The growing geriatric population has led to a rise in the use of hernia repair. According to a report published by the WHO, 1 in 6 individuals on the planet will be 60 years of age or older by 2030. At this point, there will be 1.4 billion individuals over the age of 60, up from 1 billion in 2020. The prevalence of hernias, particularly inguinal hernias, which are more common in elderly individuals, is positively driving the future of hernia repair market. Hernia repair methods are especially important in the elderly due to the weakening abdominal muscles and tissues, thus creating a positive hernia repair market outlook. The need for hernia repair tools and treatments is also fueled by the aging population's increased need for surgical interventions for a range of medical issues, thereby positively influencing the hernia repair market revenue.
Introduction of minimally invasive (MI) hernia repair devices
One major factor driving the hernia repair market is the development of minimally invasive (MI) hernia repair technologies. By providing less intrusive alternatives with less post-operative discomfort, quicker recovery periods, and a lower risk of complications than open surgeries, these devices have completely changed the way hernia repair treatments are performed, thus driving the hernia repair market growth. With the use of specialized instruments, cutting-edge imaging, and smaller incisions, MI methods enable surgeons to precisely repair hernias with the least amount of tissue damage. A hernia repair market recent news includes W. L. Gore & Associates introduced the GORE SYNECOR Intraperitoneal Biomaterial, a tri-layer hybrid solution intended to enable single, successful hernia surgery in complex patients. With its quick vascularity and long-lasting strength combined with a low profile for efficient hernia repair, this novel device fills gaps in the market for complex hernia repair. The biomaterial can be easily handled during laparoscopic, robotic, and open surgical procedures since it is flexible and conformable.
Improving Healthcare Infrastructure Globally
The worldwide healthcare infrastructure is one of the major hernia repair market drivers. Governments across the globe are investing in healthcare infrastructure due to which, there is a growing capacity for hernia repair tools and techniques. For instance, the United States spent 18.3% of its GDP on healthcare in 2021, compared to 19.7% in 2022, thus improving the hernia repair statistics. In addition to giving patients easier access to high-quality care, this infrastructure enhancement in the healthcare industry also fosters the advancement and acceptance of cutting-edge hernia repair technology.
Hernia Repair Market Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the market, along with forecasts at the global, regional, and country levels for 2026-2034. Our report has categorized the market based on product type, surgery type, hernia type, end-user.
Breakup by Product Type:
- Hernia Mesh
- Synthetic Mesh
- Biological Material Mesh
- Others
- Hernia Fixation Devices
- Sutures
- Absorbable and Non-Absorbable Tack Fixation Devices
- Glue Applicators
- Endoscopy Equipment
- Others
The report has provided a detailed breakup and analysis of the market based on the product type. This includes hernia mesh (synthetic mesh, biological material mesh, and others) and hernia fixation devices (sutures, absorbable and non-absorbable tack fixation devices, glue applicators, endoscopy equipment, and others).
Hernia repair utilizes two types of biomaterials, mesh biomaterials and fixation biomaterials to close the defect. Synthetic meshes are produced from materials such as polypropylene or polyester and are used to support damages tissues around a hernia as it heals.
Hernia fixation devices are primarily used to secure hernia mesh during surgeries. These tools are available in a range of varieties, including absorbable tacks, helical titanium tacks, and staplers made specifically for attaching prosthetic materials to soft tissue. With alternatives for both laparoscopic and open hernia repair treatments, they offer dependable strength and support for long-lasting hernia repairs.
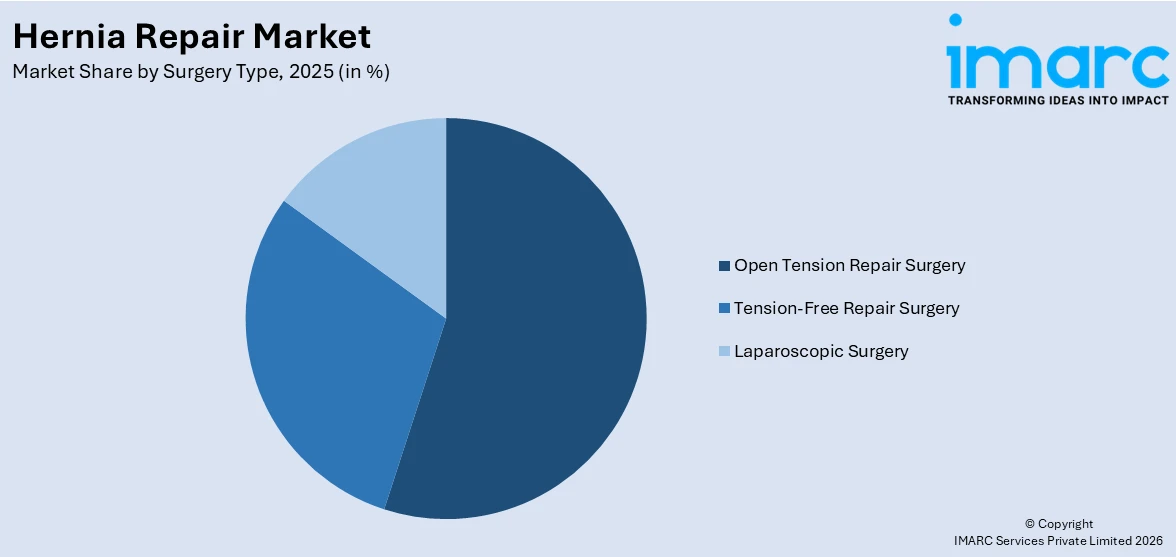
Breakup by Surgery Type:
Access the comprehensive market breakdown Request Sample
- Open Tension Repair Surgery
- Tension-Free Repair Surgery
- Laparoscopic Surgery
A detailed breakup and analysis of the market based on the surgery type have also been provided in the report. This includes open tension repair surgery, tension-free repair surgery, laparoscopic surgery.
During open tension repair surgery, the abdominal cavity is punctured over the hernia site, the projecting tissue is pushed back inside, and the incision is sutured shut. In order to seal the defect, this conventional approach uses sutures, which may cause discomfort and increase the chance of recurrence.
In tension-free repair procedures, mesh is used to bridge the hernia defect, which are regarded as the gold standard by the American College of Surgeons. When compared to tension repair techniques, this treatment lowers recurrence rates, postoperative pain, and discomfort.
In order to inspect and repair the hernia from behind the abdominal wall, laparoscopic hernia surgery requires small incisions and a laparoscope. Compared to open operations, this minimally invasive procedure has benefits such as quicker recovery periods and less pain following surgery.
Breakup by Hernia Type:
- Inguinal Hernia
- Ventral Hernia
- Femoral Hernia
- Umbilical Hernia
- Hiatal Hernia
- Others
The report has provided a detailed breakup and analysis of the market based on the hernia type. This includes inguinal hernia, ventral hernia, femoral hernia, umbilical hernia, hiatal hernia, and others.
An inguinal hernia is a protrusion in the groin or scrotum caused by tissue pushing through a weak point in the groin area. It usually needs surgery to be fixed and might cause pressure, burning, or pain.
Ventral hernias, which are frequently caused by post-surgical muscle weakening, occur along the anterior abdominal wall away from the umbilicus. The suggested course of treatment, in order to avoid complications, is surgery for symptoms that include nausea, vomiting, bloating, and abdominal discomfort.
Femoral hernias are more common in women and appear close to the groin crease, when abdominal tissue protrudes through a weak area close to the femoral vein. In order to prevent problems such as imprisonment or strangling, early repair is essential.
An umbilical hernia appears as a bulge close to the belly button, where tissues or the intestines poke through a weak area in the muscles that surround the stomach. If the hernia does not heal on its own, surgery could be suggested to avoid problems.
When a portion of the stomach protrudes into the chest cavity through the diaphragm, it is known as a hiatal hernia and can cause symptoms, such as heartburn and indigestion. To effectively manage the illness, treatment options include medication, lifestyle modifications, and occasionally surgery.
Breakup by End-User:
- Ambulatory Surgery Centers
- Hospitals and Clinics
- Others
A detailed breakup and analysis of the market based on the end user have also been provided in the report. This includes ambulatory surgery centers, hospitals and clinics, and others.
Ambulatory surgical centers are outpatient facilities where same-day surgeries are performed without overnight stays. They offer diagnostic and preventive procedures, including hernia repairs, cataract surgery, and gall bladder removal. Factors such as the rise in surgeries, growth in minimally invasive procedures, and technological advancements drive the market growth.
With a sizable portion of the market for hernia repairs, hospitals and clinics are important players in the industry. To meet the increasing demand for hernia repair operations, they provide a comprehensive range of hernia repair goods and services. Their dominance in the market is attributed to a number of factors, including the launch of innovative goods, attractive reimbursement policies, and cutting-edge healthcare infrastructure.
Breakup by Region:
- North America
- United States
- Canada
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa.
North America's hernia repair market is well-established, distinguished by a sophisticated healthcare infrastructure, substantial healthcare spending, and a notable presence of major industry participants such as Medtronic Plc and Ethicon Inc.
The increased prevalence of hernias, rising healthcare expenditures, and technological improvements are driving the European hernia repair market. France, Germany, and the United Kingdom are important suppliers to this market.
Due to a sizable target patient population, rising rates of diagnosis and treatment, and the uptake of cutting-edge treatment alternatives, the Asia Pacific hernia repair market is expanding quickly. Major market expansion potential are there in nations such as China, India, and Japan.
The existence of major players such as B. Braun SE, growing knowledge of hernia treatments, and an improved healthcare infrastructure are the characteristics of the Latin American hernia repair market. Mexico and Brazil are two of the region's most important marketplaces.
The Middle East and Africa hernia repair market is gradually expanding due to rising healthcare spending, a greater understanding of the condition, and the use of cutting-edge hernia repair technology. The market's potential for expansion in the GCC countries is especially noteworthy.
Competitive Landscape:
- The market research report has also provided a comprehensive analysis of the competitive landscape in the market. Detailed profiles of all major companies have also been provided. Some of the major market players in the hernia repair industry include B. Braun Melsungen Aktiengesellschaft, Baxter International Inc., BD (Becton, Dickinson and Company), Cook Medical Inc., CooperSurgical Inc., Ethicon Inc. (Johnson & Johnson), Herniamesh S.r.l., Lifecell Corporation (Allergan Plc), Maquet (Getinge), Medtronic Inc., Olympus Corporation, Via Surgical Ltd., W. L. Gore & Associates Inc.
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
- Important companies in the hernia repair business are continuously pursuing strategic plans to improve their offerings and market share. To bolster their positions companies are concentrating on new product releases, acquisitions, partnerships, and product innovation. For instance, Orlando Health established a Complex Hernia Center in December 2022 to offer specialist care for complex hernias, highlighting the significance of a multidisciplinary approach for the best outcomes. To create cutting-edge hernia repair devices that provide better results, less invasive treatments, and enhanced patient care, businesses are also investing in technological developments, research, and development. This is propelling growth and innovation in the hernia repair market.
Hernia Repair Market News:
- 2021: A minimally invasive technique known as transversus abdominis release (TAR), which involves surgeons using robots for hernia repair in the deep abdominal muscles, has been introduced at HOUSTON METHODIST. This technique has reduced recurrence and complication rates, particularly beneficial for immediate intervention in obese patients.
- March 2023: Recent advancements have been made by AMERICAN COLLEGE OF SURGEONS in hernia repair by using advanced mesh materials that control breakdown and prevent complications. These advancements have made hernia repair surgery safer and more predictable, with innovations in laparoscopes, robotics, and machine learning techniques. Ongoing developments in mesh technology are enhancing surgical outcomes and patient recovery.
Hernia Repair Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Product Types Covered |
|
| Surgery Types Covered | Open Tension Repair Surgery, Tension-Free Repair Surgery, Laparoscopic Surgery |
| Hernia Types Covered | Inguinal Hernia, Ventral Hernia, Femoral Hernia, Umbilical Hernia, Hiatal Hernia, Others |
| End Users Covered | Ambulatory Surgery Centers, Hospitals and Clinics, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | B. Braun Melsungen Aktiengesellschaft, Baxter International Inc., BD (Becton, Dickinson and Company), Cook Medical Inc., CooperSurgical Inc., Ethicon Inc. (Johnson & Johnson), Herniamesh S.r.l., Lifecell Corporation (Allergan Plc), Maquet (Getinge), Medtronic Inc., Olympus Corporation, Via Surgical Ltd., W. L. Gore & Associates Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the hernia repair market from 2020-2034.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the global hernia repair market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the hernia repair industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Frequently Asked Questions About the Hernia Repair Market Report
The global hernia repair market was valued at USD 6.0 Billion in 2025.
We expect the global hernia repair market to exhibit a CAGR of 2.66% during 2026-2034.
The increasing prevalence of hernia, along with the introduction of biologic allograft mesh, which aids in minimizing post-surgery pain and offers a faster recovery rate, is primarily driving the global hernia repair market.
The sudden outbreak of the COVID-19 pandemic had led to the postponement of elective hernia repair treatment procedures to reduce the risk of the coronavirus infection upon hospital visits and interaction with healthcare professionals or medical equipment.
On a regional level, the market has been classified into North America, Asia Pacific, Europe, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global hernia repair market include B. Braun Melsungen Aktiengesellschaft, Baxter International Inc., BD (Becton, Dickinson and Company), Cook Medical Inc., CooperSurgical Inc., Ethicon Inc. (Johnson & Johnson), Herniamesh S.r.l., Lifecell Corporation (Allergan Plc), Maquet (Getinge), Medtronic Inc., Olympus Corporation, Via Surgical Ltd., W. L. Gore & Associates Inc., etc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












