
Hospital Acquired Disease Testing Market Report by Indication (UTI (Urinary Tract Infection), SSI (Surgical Site Infection), Pneumonia, Bloodstream Infections, MRSA (Methicillin-Resistant Staphylococcus Aureus), and Others), and Region 2026-2034
Hospital Acquired Disease Testing Market Overview:
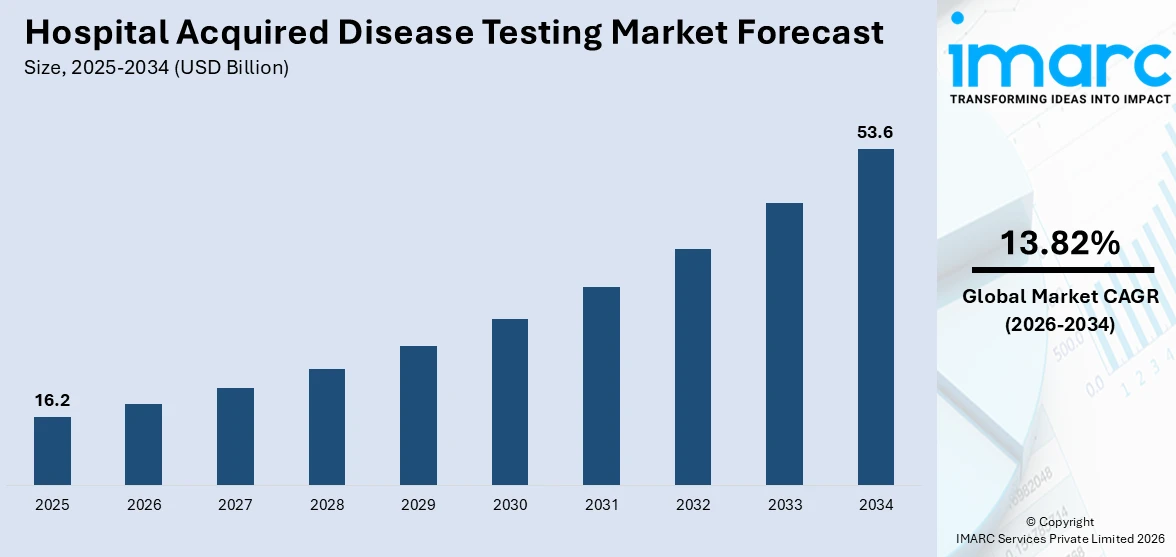
The global hospital acquired disease testing market size reached USD 16.2 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 53.6 Billion by 2034, exhibiting a growth rate (CAGR) of 13.82% during 2026-2034. There are several factors that are driving the market, which include the rising geriatric population, increasing prevalence of hospital acquired infections (HAIs) like urinary tract infections (UTIs), bloodstream infections, and pneumonia, and the growing number of people undergoing different surgical procedures.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 16.2 Billion |
|
Market Forecast in 2034
|
USD 53.6 Billion |
| Market Growth Rate 2026-2034 | 13.82% |
Hospital Acquired Disease Testing Market Analysis:
- Major Market Drivers: The market is experiencing robust growth because of stringent hospital protocols and accreditation requirements and technological advancements in diagnostic tools.
- Key Market Trends: The increasing geriatric population and the rising prevalence of hospital acquired infections (HAIs) are key market trends.
- Geographical Trends: North America exhibits a clear dominance, accounting for the biggest market share owing to the presence of specialized hospitals and clinics having modern diagnostic and treatment technologies.
- Competitive Landscape: Major market players in the hospital acquired disease testing industry are Abbott Laboratories, Alere, Inc., Becton, Dickinson and Company, bioMérieux SA, F. Hoffmann-La Roche Ltd., Hologic, Inc., QIAGEN, Siemens Healthcare, Diatherix Laboratories, Inc., Meridian Bioscience, Inc., Thermo Fisher Scientific, Inc. (Life Technologies Corporation), and Cepheid, Inc., among many others.
- Challenges and Opportunities: Sample collection and handling issues represent a key market challenge. Nonetheless, the focus on personalized medicines, along with the increasing awareness about the importance of infection control, is anticipated to overcome these challenges and offer recent market opportunities.
To get more information on this market Request Sample
Hospital Acquired Disease Testing Market Trends:
Rising Geriatric Population
The demand for hospital-acquired disease (HAD) testing is driven by the increasing geriatric population and the prevalence of chronic illnesses. Due to their complicated medical needs, patients who are elderly or have long-term illnesses like diabetes, heart disease, or respiratory disorders often require staying in the hospital for a longer period. Extended hospital stays increase the chances of acquiring infections like surgical site infections, pneumonia, and urinary tract infections (UTIs) owing to high exposure to hospital surroundings. According to the World Health Organization (WHO), there will be 426 million people 80 years of age or older in the world by 2050.
Increasing Prevalence of Hospital-Acquired Infections (HAIs)
Patient Safety Authority states that Pennsylvania's long-term care facilities reported 23,970 infection complaints in 2023. Bloodstream infections, surgical site infections, pneumonia, and UTIs are becoming more common, which is driving the expansion of the hospital acquired infections diagnostics market. Additionally, hospitals must run more diagnostic tests as to detect and treat infections more quickly because of a rise in the number of HAIs. This enhances patient outcomes and benefits in managing the spread within the hospitals. Furthermore, early infection detection through testing can aid in lowering the chances of death rates, shorten hospital stays, and lessen consequences.
Growing Number of Surgical Procedures
As per the research study of the IMARC Group, the global minimally invasive (MI) surgery market reached US$ 52.9 Billion in 2023. The rise in the number of surgeries globally directly correlates with an increased risk of HAIs, particularly surgical site infections (SSIs). More patients are being exposed to the hospital environment because of an increase in surgeries, ranging from routine procedures to complex operations. Surgical procedures usually include incisions or openings in the body, providing entry points for pathogens, increasing the susceptibility to SSIs. Early detection of SSIs benefits in preventing complications like wound dehiscence, sepsis, and delayed healing, which can thereby lead to prolonged hospital stays and increased healthcare costs.
Hospital Acquired Disease Testing Market Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the market, along with forecasts at the global and regional for 2026-2034. Our report has categorized the market based on indication.
Breakup by Indication:
Access the comprehensive market breakdown Request Sample
- UTI (Urinary Tract Infection)
- SSI (Surgical Site Infection)
- Pneumonia
- Bloodstream Infections
- MRSA (Methicillin-Resistant Staphylococcus Aureus)
- Others
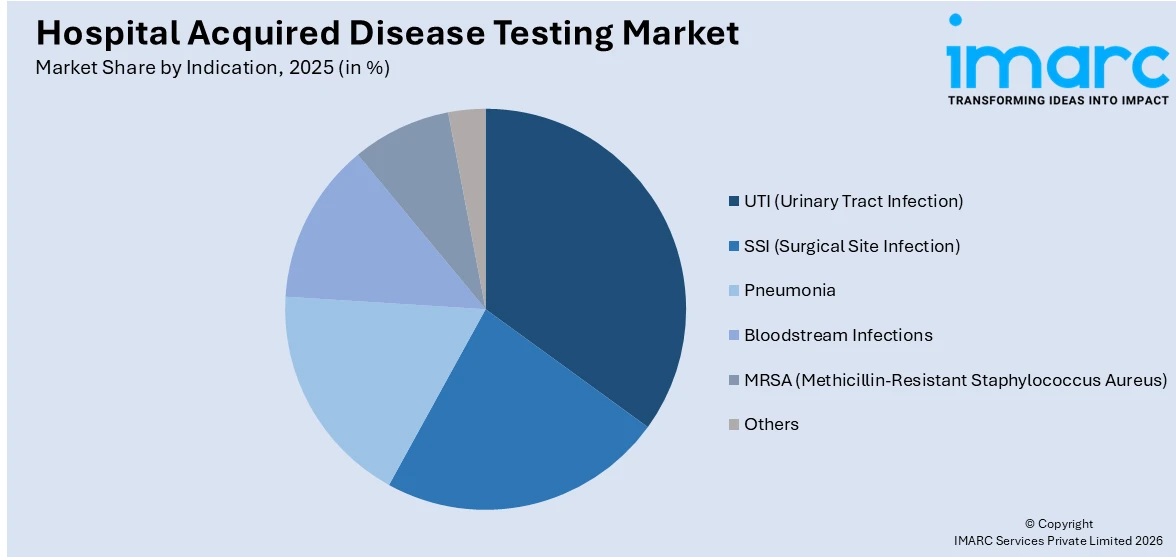
UTI (urinary tract infection) accounts for the majority of the market share
The report has provided a detailed breakup and analysis of the market based on the indication. This includes UTI (urinary tract infection), SSI (surgical site infection), pneumonia, bloodstream infections, MRSA (methicillin-resistant staphylococcus aureus), and others. According to the report, UTI (urinary tract infection) represented the largest segment.
One of the most prevalent kinds of HAIs is UTI (urinary tract infection), which mostly affects patients in intensive care units (ICUs), those with indwelling catheters, and those who stay overnight. UTIs during hospital stays are more common in the elderly, in people with compromised immune systems, and in people with long-term medical illnesses including diabetes. Because these susceptible populations are so common in healthcare settings, there is a significant need for diagnostic testing to identify UTIs. Furthermore, early discovery through testing lowers the possibility of these issues, which enhances patient outcomes and lowers medical expenses.
Breakup by Region:
- North America
- Europe
- Asia Pacific
- Middle East and Africa
- Latin America
North America leads the market, accounting for the largest hospital acquired disease testing market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America, Europe, Asia Pacific, the Middle East and Africa, and Latin America. According to the report, North America represents the largest regional market for hospital acquired disease testing.
North America boasts a highly developed healthcare system with advanced medical facilities, a high number of hospitals, and state-of-the-art technologies. In addition, favorable government initiatives are compelling hospitals and healthcare facilities to adopt frequent testing for these diseases to meet compliance standards. Apart from this, the rising geriatric population in the North American region is supporting the market growth. As reported by the Population Reference Bureau (PRB), 82 million Americans are expected to be 65 years of age or older by 2050.
Competitive Landscape:
- The market research report has also provided a comprehensive analysis of the competitive landscape in the market. Detailed profiles of all major companies have also been provided. Major market players in the hospital acquired disease testing industry are Abbott Laboratories, Alere, Inc., Becton, Dickinson and Company, bioMérieux SA, F. Hoffmann-La Roche Ltd., Hologic, Inc., QIAGEN, Siemens Healthcare, Diatherix Laboratories, Inc., Meridian Bioscience, Inc., Thermo Fisher Scientific, Inc. (Life Technologies Corporation), and Cepheid, Inc.
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
- Top players are developing faster, more accurate, and convenient diagnostic tests for HADs. They are innovating advanced molecular diagnostic tests that can quickly detect infections. They are focusing on developing rapid point-of-care (POC) diagnostic tests that can be administered directly in hospital settings, providing immediate results and facilitating early intervention. In line with this, key players are collaborating with healthcare firms, research institutions, and technology providers to create more advanced products. For instance, on 19 May 2023, researchers at the University of Texas at Dallas created a prototype diagnostic sensor that will enable users to diagnose UTIs rapidly, without having to wait 24 to 48 hours for lab test results.
Hospital Acquired Disease Testing Market News:
- 15 February 2023: Thermo Fisher Scientific unveiled real time PCR kits for the detection of HAIs among individuals in India. The company provides a complete qPCR ecosystem, which includes the Applied Biosystems QuantStudio 5 Dx Real-Time PCR System and upcoming QuantStudio 7 Pro Dx Real-Time PCR Systems designed to simplify workflows, a broad portfolio of master mixes and PCR plastics, RNA/DNA for sample preparation and biosafety cabinets, centrifuges, cold storage, pipettes, and tips to support the entire end-to-end workflow of any diagnostic lab.
Hospital Acquired Disease Testing Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Indications Covered | UTI (Urinary Tract Infection), SSI (Surgical Site Infection), Pneumonia, Bloodstream Infections, MRSA (Methicillin-Resistant Staphylococcus Aureus), Others |
| Region Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Companies Covered | Abbott Laboratories, Alere, Inc., Becton, Dickinson and Company, bioMérieux SA, F. Hoffmann-La Roche Ltd., Hologic, Inc., QIAGEN, Siemens Healthcare, Diatherix Laboratories, Inc., Meridian Bioscience, Inc., Thermo Fisher Scientific, Inc. (Life Technologies Corporation), Cepheid, Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the hospital acquired disease testing market from 2020-2034.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the global market.
- The study maps the leading, as well as the fastest-growing, regional markets.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the hospital acquired disease testing industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Frequently Asked Questions About the Hospital Acquired Disease Testing Market Report
The global hospital acquired disease testing market size was valued at USD 16.2 Billion in 2025.
According to the estimates by IMARC Group, the global hospital acquired disease testing market is expected to grow at a CAGR of 13.82% from 2026-2034.
Rising incidences of HAIs upon exposure to infected patients, contaminated linens, medical equipment, etc., are currently driving the market for hospital acquired disease testing.
Several technological advancements have led to the introduction of microarrays, solid phase hybridization, polymerase chain reaction (PCR) and real-time location systems (RTLSs) for detecting and monitoring HAIs, thereby representing the one of the key trends in the global hospital acquired disease testing market.
Sudden outbreak of the COVID-19 pandemic has led to the rising demand for advanced and effective testing devices for hospital acquired diseases based on the increasing risk of coronavirus contraction upon hospital visits.
On the basis of the indication, the market has been bifurcated into urinary tract infection, surgical site infection (SSI), pneumonia, bloodstream infections, methicillin-resistant staphylococcus aureus (MRSA), and others. Amongst these, urinary tract infection (UTI) holds the majority of the total market share.
Region-wise, the market has been classified into North America, Europe, Asia Pacific, Middle East and Africa, and Latin America, where North America dominates the global market.
The key companies in the global hospital acquired disease testing market are Abbott Laboratories, Alere, Inc., Becton, Dickinson and Company, bioMérieux SA, F. Hoffmann-La Roche Ltd., Hologic, Inc., QIAGEN, Siemens Healthcare, Diatherix Laboratories, Inc., Meridian Bioscience, Inc., Thermo Fisher Scientific, Inc. (Life Technologies Corporation) and Cepheid, Inc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












