
Hypoactive Sexual Desire Disorder Treatment Market Report by Therapy (Buspirone, Bupropion, Testosterone, Flibanserin, Bremelanotide, Cognitive Behavior Therapy), Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), Application (Hospitals, Clinics, and Others), and Region 2026-2034
Hypoactive Sexual Desire Disorder Treatment Market Size:
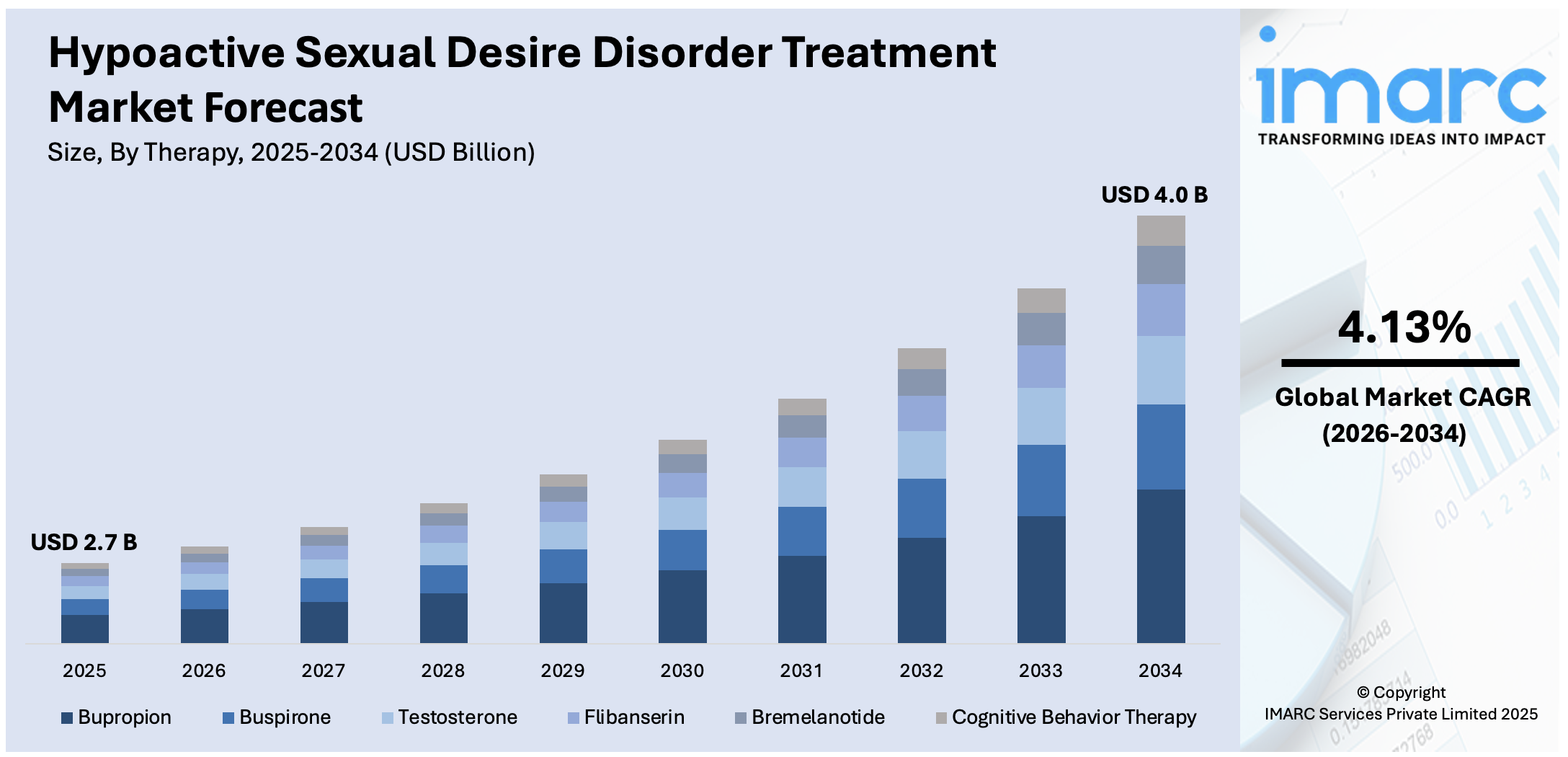
The global hypoactive sexual desire disorder treatment market size reached USD 2.7 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 4.0 Billion by 2034, exhibiting a growth rate (CAGR) of 4.13% during 2026-2034. The escalating consumer awareness about hypoactive sexual desire disorder (HSDD) and its rising prevalence among women, ongoing advancements in treatment options, extensive research activities, improved healthcare infrastructure, supportive government initiatives, and enhanced patient access to novel therapies are some of the key factors strengthening the market growth.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 2.7 Billion |
|
Market Forecast in 2034
|
USD 4.0 Billion |
| Market Growth Rate 2026-2034 | 4.13% |
Hypoactive Sexual Desire Disorder Treatment Market Analysis:
- Major Market Drivers: Increasing awareness and diagnosis of HSDD through educational initiatives and public awareness campaigns drive the market growth by encouraging more individuals to seek treatment. In line with this, the development and approval of specialized drugs like flibanserin and bremelanotide are strengthening the market growth. Moreover, expanding healthcare infrastructure and patient access to medical services due to the advent of telemedicine and improved insurance coverage facilitating better diagnosis and treatment are contributing to the market expansion.
- Key Market Trends: Growing investment in research and development (R&D) leading to the introduction of novel therapies with improved efficacy and safety profiles is one of the prominent market trends influencing market growth. Furthermore, the hypoactive sexual desire disorder treatment market recent opportunities like mergers and acquisitions among pharmaceutical companies to enhance their geographic reach and diversify their product offerings are fostering market expansion. Besides this, the market is witnessing increased participation from both established pharmaceutical giants and emerging biotech firms, which is supporting the market demand.
- Geographical Trends: North America leads the HSDD treatment market due to its advanced healthcare infrastructure, high awareness, and strong presence of pharmaceutical companies engaged in developing HSDD treatments. Along with this, proactive approaches towards sexual health, supported by educational campaigns and robust insurance coverage in the region are impelling the market growth: Furthermore, early access to new therapies and substantial investment in R&D are also boosting the hypoactive sexual desire disorder treatment market revenue.
- Competitive Landscape: Some of the major market players in the hypoactive sexual desire disorder treatment industry include Allergan Plc, AMAG Pharmaceuticals, Inc., Emotional Brain BV, Endoceutics, Inc., GlaxoSmithKline Plc, Ovoca Bio Plc, Pivot Pharmaceuticals, Inc., Palatin Technologies, Inc., S1 Biopharma, Inc., Sprout Pharmaceuticals, Inc., among many others.
- Challenges and Opportunities: Social stigma and underreporting of sexual health issues pose significant challenges, potentially limiting diagnosis and treatment rates. Besides this, financial barriers and limited awareness in some regions also hinder market growth, necessitating targeted educational and affordability initiatives. Meanwhile, the opportunities lie in expanding telemedicine services and digital health platforms, improving patient access to diagnosis and treatment, especially in remote or underserved areas.
To get more information on this market Request Sample
Hypoactive Sexual Desire Disorder Treatment Market Trends:
Increasing awareness and diagnosis
The identification and diagnosis of HSDD have greatly improved due to a rise in awareness campaigns and educational initiatives. Historically, sexual health issues, especially those affecting women, have been underreported and undertreated due to social stigma and lack of awareness. However, in recent years, efforts by healthcare providers, patient advocacy groups, and pharmaceutical companies have brought attention to the importance of sexual health. Campaigns to raise public awareness, professional development for healthcare workers, and the creation of diagnostic standards and instruments are fostering market expansion. Besides this, the increased visibility of HSDD in medical literature and mainstream media has encouraged more individuals to seek help and led to higher diagnosis rates, strengthening the hypoactive sexual desire disorder treatment market growth.
Advancements in pharmaceutical treatments
The pharmaceutical landscape for HSDD has evolved with the development of new and effective treatments. Traditional approaches often involved off-label use of medications not specifically approved for HSDD, which limited treatment options and effectiveness. However, the approval of drugs specifically designed to address HSDD, such as flibanserin (marketed as Addyi) and bremelanotide (marketed as Vyleesi), has revolutionized the market. These drugs have offered focused therapeutic alternatives and have demonstrated promise in elevating sexual desire and lowering HSDD-related distress. Moreover, extensive R&D initiatives focused on developing innovative treatments with improved safety profiles, administration methods, and efficacy are aiding in market expansion. The introduction of these specialized drugs has expanded treatment options and heightened interest and investment in the HSDD market, contributing to the hypoactive sexual desire disorder treatment demand.
Expansion of healthcare infrastructure and patient access
Improved healthcare infrastructure and increased patient access to medical services also play a crucial role in the market growth. Improved healthcare systems have made it easier to diagnose and treat HSDD, especially in developed nations. Digital health platforms and telemedicine have also become essential tools, giving patients easier access to medical professionals and treatment options—particularly in remote areas. In line with this, the expansion of health insurance coverage for mental and sexual health issues has reduced financial barriers, allowing more patients to seek and receive appropriate care. Government initiatives and policies aimed at improving women's health and promoting sexual health awareness by ensuring that more individuals have access to the necessary resources and treatments are bolstering the market demand.
Hypoactive Sexual Desire Disorder Treatment Market Segmentation
IMARC Group provides an analysis of the key trends in each segment of the market, along with forecasts at the global, regional, and country levels for 2026-2034. Our report has categorized the market based on therapy, distribution channel, and application.
Breakup by Therapy:
- Buspirone
- Bupropion
- Testosterone
- Flibanserin
- Bremelanotide
- Cognitive Behavior Therapy
Bupropion accounts for the majority of the market share
The report has provided a detailed breakup and analysis of the market based on the therapy. This includes buspirone, bupropion, testosterone, flibanserin, bremelanotide, and cognitive behavior therapy. According to the report, bupropion represented the largest segment.
As per the hypoactive sexual desire disorder treatment market forecast, the demand for bupropion in the HSDD treatment market is spurred by its established efficacy, favorable safety profile, and off-label use in treatment. Originally an antidepressant, bupropion has shown effectiveness in enhancing sexual desire and arousal, making it a preferred choice among healthcare providers. Co-occurring depression, which is common in HSDD patients, can also be managed due to its dual function as an antidepressant and HSDD treatment. Additionally, bupropion's relatively low cost and wide availability contribute to its extensive demand and sales among patients seeking treatment for HSDD.
Breakup by Distribution Channel:
Access the comprehensive market breakdown Request Sample
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
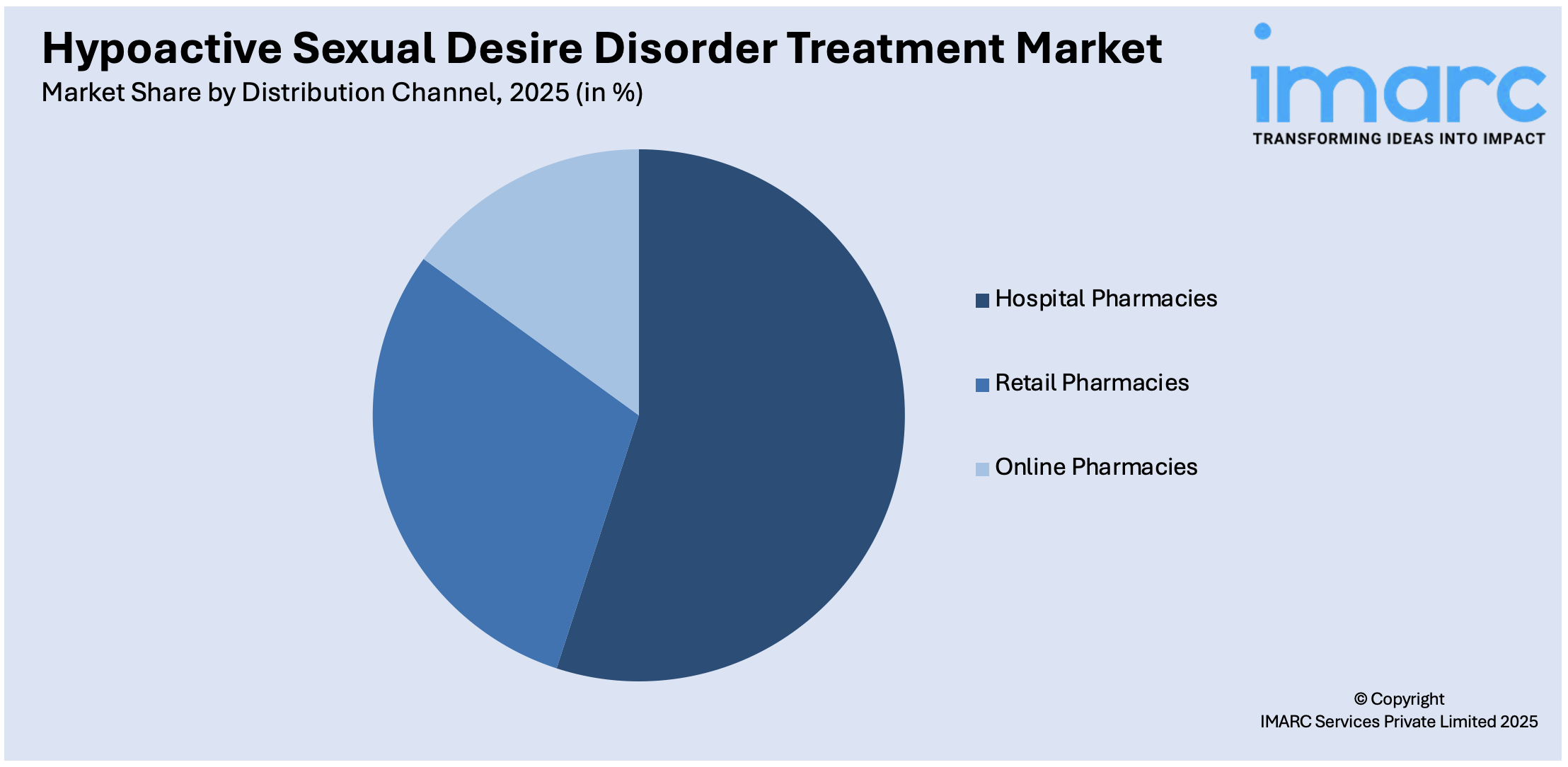
Hospital pharmacies holds the largest share of the industry
A detailed breakup and analysis of the market based on the distribution channel have also been provided in the report. This includes hospital pharmacies, retail pharmacies, and online pharmacies. According to the report, hospital pharmacies accounted for the largest market share.
Hypoactive sexual desire disorder treatment market overview shows that hospital pharmacies dominate the market due to their central role in the healthcare system, providing comprehensive care and ensuring accurate diagnosis and treatment. Hospitals are frequently the primary points of care (POC) for the diagnosis of HSDD, particularly in their specialized mental health and gynecology departments. They have direct access to specialized medications and can offer immediate dispensing, ensuring that patients receive their treatments promptly. Moreover, hospital pharmacies are more likely to stock a wider range of HSDD medications, including newer or less common treatments, facilitating better management of the disorder. This accessibility and comprehensive care make hospital pharmacies a crucial distribution channel for HSDD treatments.
Breakup by Application:
- Hospitals
- Clinics
- Others
Hospitals represent the leading market segment
The report has provided a detailed breakup and analysis of the market based on the application. This includes hospitals, clinics, and others. According to the report, hospitals represented the largest segment.
The increasing product demand across hospitals owing to their comprehensive diagnostic and treatment capabilities is propelling the market forward. Hospitals include house-specialized departments such as gynecology and psychiatry, where HSDD is frequently diagnosed and managed. They offer access to a wide array of treatments, including advanced and experimental therapies, ensuring patients receive the most effective care. The presence of multidisciplinary teams allows for holistic treatment approaches, addressing both physical and psychological aspects of HSDD. Furthermore, hospitals often participate in clinical trials, contributing to the development of new treatments and ensuring early access to innovative therapies, which is positively impacting the hyperactive sexual desire disorder treatment market outlook.
Breakup by Region:
- North America
- United States
- Canada
- Europe
- United Kingdom
- Germany
- France
- Spain
- Italy
- Russia
- Others
- Asia Pacific
- China
- Japan
- India
- South Korea
- Others
- Middle East and Africa
- Saudi Arabia
- Iran
- Turkey
- Others
- Latin America
- Brazil
- Mexico
- Others
North America leads the market, accounting for the largest hypoactive sexual desire disorder treatment market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (United Kingdom, Germany, France, Spain, Italy, Russia, and others); Asia Pacific (China, Japan, India, South Korea, and others); Middle East and Africa ( Saudi Arabia, Iran, Turkey, and others); and Latin America (Brazil, Mexico, and others). According to the report, North America represents the largest regional market for hypoactive sexual desire disorder treatment.
North America leads the market with its advanced healthcare infrastructure, high awareness, and better diagnostic capabilities, leading to more frequent identification and treatment of HSDD. Concurrently, the region has a strong presence of pharmaceutical companies engaged in the development and commercialization of HSDD treatments, which ensures early access to new therapies. Moreover, the proactive approach towards sexual health, supported by educational campaigns and robust insurance coverage, facilitates enhanced patient access to necessary treatments. Apart from this, the region's substantial investment in R&D drives innovation, further contributing to the hypoactive sexual desire disorder treatment market recent developments.
Competitive Landscape:
- The market research report has also provided a comprehensive analysis of the competitive landscape in the market. Detailed profiles of all major companies have also been provided. Some of the major market players in the hypoactive sexual desire disorder treatment industry include Allergan Plc, AMAG Pharmaceuticals, Inc., Emotional Brain BV, Endoceutics, Inc., GlaxoSmithKline Plc, Ovoca Bio Plc, Pivot Pharmaceuticals, Inc., Palatin Technologies, Inc., S1 Biopharma, Inc., Sprout Pharmaceuticals, Inc., etc.
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
- The competitive landscape is characterized by a mix of established pharmaceutical giants and emerging biotech firms vying for market share. Prominent manufacturers are leading the market with FDA-approved treatments specifically for HSDD. In addition to this, hypoactive sexual desire disorder treatment companies are investing heavily in marketing, educational campaigns, and research to expand their market presence and address unmet needs. They are continuously introducing new treatments with improved efficacy and safety profiles. The market is also shaped by strategic partnerships, mergers, and acquisitions among key players to enhance product portfolios and geographic reach. Besides this, the increasing awareness and changing societal attitudes towards sexual health drive demand, prompting companies to innovate and differentiate their offerings to capture a larger share of this growing market.
Hypoactive Sexual Desire Disorder Treatment Market News:
- In August 2023, Palatin Technologies partnered with UpScriptHealth to offer its FDA-approved treatment, Vyleesi for HSDD, through UpScriptHealth's Women's Telehealth Platform. This collaboration aims to enhance awareness and accessibility of Vyleesi to a broad patient base.
- In January 2024, Cosette Pharmaceuticals acquired Vyleesi® from Palatin Technologies, securing five patents with protection until 2041. The transition will ensure uninterrupted access for patients and healthcare professionals. This move is aimed at enhancing access to Vyleesi® for women with HSDD.
Hypoactive Sexual Desire Disorder Treatment Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Therapies Covered | Buspirone, Bupropion, Testosterone, Flibanserin, Bremelanotide, Cognitive Behavior Therapy |
| Distribution Channels Covered | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies |
| Applications Covered | Hospitals, Clinics, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, United Kingdom, Germany, France, Spain, Italy, Russia, China, Japan, India, South Korea, Saudi Arabia, Iran, Turkey, Brazil, Mexico |
| Companies Covered | Allergan Plc, AMAG Pharmaceuticals, Inc., Emotional Brain BV, Endoceutics, Inc., GlaxoSmithKline Plc, Ovoca Bio Plc, Pivot Pharmaceuticals, Inc., Palatin Technologies, Inc., S1 Biopharma, Inc., Sprout Pharmaceuticals, Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the hypoactive sexual desire disorder treatment market from 2020-2034.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the global hypoactive sexual desire disorder treatment market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the hypoactive sexual desire disorder treatment industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Frequently Asked Questions About the Hypoactive Sexual Desire Disorder Treatment Market Report
The hypoactive sexual desire disorder treatment market was valued at USD 2.7 Billion in 2025.
We expect the global hypoactive sexual desire disorder treatment market to exhibit a CAGR of 4.13% during 2026-2034.
The rising prevalence of Hypoactive Sexual Desire Disorder (HSDD), along with the increasing focus on R&D of new drugs and therapies for more effective treatment of HSDD, is primarily driving the global hypoactive sexual desire disorder treatment market.
The sudden outbreak of the COVID-19 pandemic had led to the postponement of elective treatment procedures for HSDD to reduce the risk of the coronavirus infection upon hospital visits and interaction with healthcare professionals or medical equipment.
Based on the therapy, the global hypoactive sexual desire disorder treatment market can be segmented into buspirone, bupropion, testosterone, flibanserin, bremelanotide, and cognitive behavior therapy. Currently, bupropion holds the majority of the total market share.
Based on the distribution channel, the global hypoactive sexual desire disorder treatment market has been divided into hospital pharmacies, retail pharmacies, and online pharmacies. Among these, hospital pharmacies currently exhibit a clear dominance in the market.
Based on the application, the global hypoactive sexual desire disorder treatment market can be categorized into hospitals, clinics, and others. Currently, hospitals account for the largest market share.
On a regional level, the market has been classified into North America, Europe, Asia Pacific, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global hypoactive sexual desire disorder treatment market include Allergan Plc, AMAG Pharmaceuticals, Inc., Emotional Brain BV, Endoceutics, Inc., GlaxoSmithKline Plc, Ovoca Bio Plc, Pivot Pharmaceuticals, Inc., Palatin Technologies, Inc., S1 Biopharma, Inc., and Sprout Pharmaceuticals, Inc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












