
In-vivo CRO Market Report by Type (Rodent, Non-Rodent), GLP Type (Non-GLP, GLP Toxicology), Indication (Autoimmune/Inflammation Conditions, Pain Management, Oncology, CNS Conditions, Diabetes, Obesity, and Others), and Region 2026-2034
Global In-vivo CRO Market:
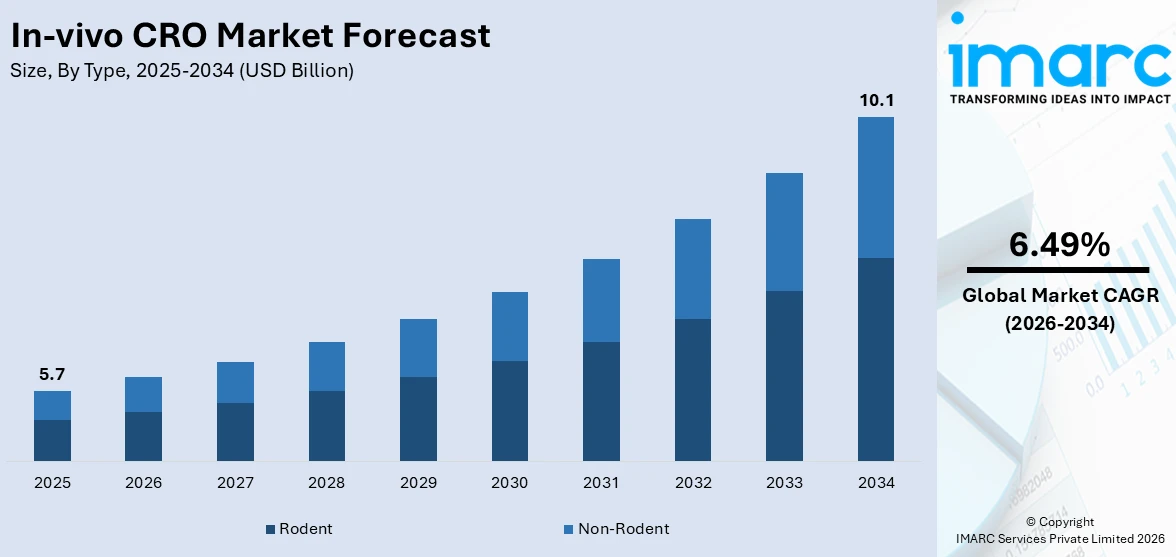
The global in-vivo CRO market size reached USD 5.7 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 10.1 Billion by 2034, exhibiting a growth rate (CAGR) of 6.49% during 2026-2034. The growing prevalence of chronic diseases worldwide and the increasing R&D expenditure by pharmaceutical companies are propelling the market growth.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 5.7 Billion |
|
Market Forecast in 2034
|
USD 10.1 Billion |
| Market Growth Rate 2026-2034 | 6.49% |
In-vivo CRO Market Analysis:
- Major Market Drivers: The rapid growth in the number of biotech startups and the rising focus on personalized and precision medicine are stimulating the market growth.
- Key Market Trends: Various outsourcing preclinical research studies and the inflating adoption of artificial intelligence in research methodologies are acting as growth-inducing factors.
- Competitive Landscape: Some of the prominent market companies include Biocytogen, Charles River Laboratories, Crown Bioscience, Eurofins Discovery, Evotec, Fortrea, Icon Plc, IQVIA Inc., Iris Pharma, Taconic Biosciences, Inc., and WuXi AppTec, among many others.
- Geographical Trends: North America dominates the market due to due to advanced healthcare infrastructure, strong pharmaceutical and biotechnology sectors, and high R&D investment.
- Challenges and Opportunities: The high costs and regulatory challenges hinder the market growth. However, adopting advanced technologies and streamlining compliance processes to enhance efficiency and reduce expenses will continue to propel the market growth over the foreseeable future.
To get more information on this market Request Sample
In-vivo CRO Market Trends:
Increased Outsourcing by Pharmaceutical Companies
Pharmaceutical companies are focusing on outsourcing to in-vivo contract research organizations to enhance research efficiency and reduce costs. These CROs provide specialized expertise in animal studies, thereby enabling faster drug development and regulatory approval while allowing companies to focus on core competencies. For example, in July 2024, IntegriChain, delivering pharma's comprehensive data, consulting, technology, and outsourcing platform for data-driven commercialization, acquired Federal Compliance Solutions (FCS), a leading pharma advisory and managed services firm, to develop more profitable drug commercialization strategies. According to the in-vivo CRO market analysis report, this is acting as a growth-inducing factor.
Rise of Specialized CRO Services
The rise of specialized in-vivo CRO services reflects a growing demand for niche expertise in preclinical studies. These CROs offer tailored services in areas like animal modeling, pharmacokinetics, and toxicology, enhancing drug development efficiency and precision in therapeutic research and development, escalating the in-vivo CRO market demand. For instance, in June 2024, Lindus Health launched an all-in-one medical device CRO service, combining in-vivo CRO expertise with advanced technology and recruitment strategies, streamlining medical device clinical trials, accelerating participant enrollment across various therapeutic areas and regulatory pathways, etc.
Growing Focus on Drug Development
The increasing emphasis on drug development has led to greater support for in-vivo CRO. These organizations provide essential services for preclinical testing, enabling pharmaceutical companies to evaluate the safety and efficacy of new drugs in living organisms, speeding up the development process while reducing costs and resource requirements. For example, in May 2024, Cannovation Clinical Research Partners launched as a CRO specializing in cannabis-based drug trials, focusing on preclinical and clinical phases, developed cannabinoid-based therapeutics through collaborations with pharmaceutical companies, universities, and DEA researchers.
Global In-vivo CRO Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global in-vivo CRO market report, along with forecasts at the global, regional and country levels from 2026-2034. Our report has categorized the market based on type, GLP type and indication.
Breakup by Type:
- Rodent
- Rats
- Mice
- Others
- Non-Rodent
Rodent dominates the in-vivo CRO market share
The report has provided a detailed breakup and analysis of the in-vivo CRO market based on the type. This includes rodent (rats, mice, and others) and non-rodent. According to the report, rodent represented the largest market segmentation.
Rodents, particularly mice and rats, dominate the market due to their genetic similarity to humans, cost-effectiveness, and rapid breeding. For example, mice are extensively used for cancer research and drug testing in preclinical trials. Moreover, companies, such as Envigo and Charles River Laboratories, developed advanced rodent models and enhanced services to support several stages of drug development.
Breakup by GLP Type:
Access the comprehensive market breakdown Request Sample
- Non-GLP
- In House
- Outsourcing
- GLP Toxicology
- In House
- Outsourcing
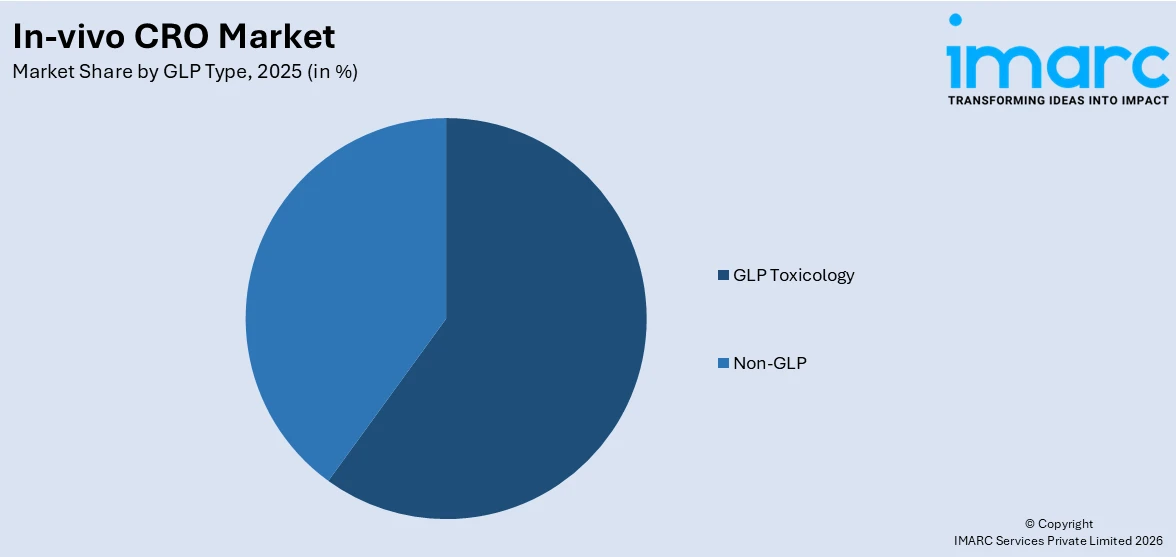
GLP toxicology exhibits a clear dominance in the market
The report has provided a detailed breakup and analysis of the market based on the GLP type. This includes non-GLP (in house and outsourcing) and GLP toxicology (in house and outsourcing). According to the report, GLP toxicology represented the largest market segmentation.
According to the in-vivo CRO market forecast report, GLP toxicology dominates the market, as companies strive to meet the growing demand for rigorous safety assessment in drug development. Advanced GLP toxicology services, including innovative study designs, specialized toxicology models, and enhanced data analytics platforms by leading organizations, like Covance and Labcorp Drug Development, are gaining traction.
Breakup by Indication:
- Autoimmune/Inflammation Conditions
- Rheumatoid Arthritis
- Multiple Sclerosis
- Osteoarthritis
- Irritable Bowel Syndrome
- Others
- Pain Management
- Chronic Pain
- Acute Pain
- Oncology
- Blood Cancer
- Solid Tumor
- Others
- CNS Conditions
- Epilepsy
- Parkinson’s Disease
- Huntington’s Disease
- Stroke
- Traumatic Brain Injury
- ALS
- Muscle Regeneration
- Others
- Diabetes
- Obesity
- Others
Oncology dominates the in-vivo CRO market outlook
The report has provided a detailed breakup and analysis of the market based on the indication. This includes autoimmune/inflammation conditions (rheumatoid arthritis, multiple sclerosis, osteoarthritis, irritable bowel syndrome, and others), pain management (chronic pain and acute pain), oncology (blood cancer, solid tumor, and others), CNS conditions (epilepsy, Parkinson’s disease, Huntington’s disease, stroke, traumatic brain injury, ALS, muscle regeneration, and others), diabetes, obesity, and others. According to the report, oncology represented the largest market segmentation.
Oncology dominates the market due to the high demand for cancer drug research and development. For example, CROs conduct animal studies to evaluate the efficacy and safety of new oncology therapies before clinical trials. Besides this, the rising innovations in supporting biotech and pharmaceutical companies to develop new cancer therapies are driving the segment's growth.
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America dominates the market
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America accounted for the largest market share.
According to the in-vivo CRO market outlook report, North America leads the market due to its advanced healthcare infrastructure, substantial R&D investment, and presence of major pharmaceutical companies. The region benefits from a strong regulatory framework, experienced researchers, and high clinical trial activity. For instance, Charles River Laboratories, a key player in the U.S., provides comprehensive in-vivo services, from early-stage research to preclinical testing, which attracts global clients seeking high-quality, reliable research outcomes. This advantage positions North America as a dominant region.
Competitive Landscape:
The in-vivo CRO market research report has provided a comprehensive analysis of the competitive landscape. Detailed profiles of all major market companies have also been provided. Some of the key players in the market include:
- Biocytogen
- Charles River Laboratories
- Crown Bioscience
- Eurofins Discovery
- Evotec
- Fortrea
- Icon Plc
- IQVIA Inc.
- Iris Pharma
- Taconic Biosciences, Inc.
- WuXi AppTec
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
In-vivo CRO Market Recent Developments:
- July 2024: IntegriChain, delivering pharma's comprehensive data, consulting, technology, and outsourcing platform for data-driven commercialization, acquired Federal Compliance Solutions (FCS), a leading pharma advisory and managed services firm, to develop more profitable drug commercialization strategies.
- June 2024: Lindus Health launched an all-in-one medical device CRO service, combining in-vivo CRO expertise with advanced technology and recruitment strategies, streamlining medical device clinical trials, and accelerating participant enrollment across various therapeutic areas and regulatory pathways.
- May 2024: Cannovation Clinical Research Partners launched as a CRO specializing in cannabis-based drug trials, focusing on preclinical and clinical phases, and developed cannabinoid-based therapeutics through collaborations with pharmaceutical companies, universities, and DEA researchers.
In-vivo CRO Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Types Covered |
|
| GLP Types Covered |
|
| Indications Covered |
|
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Biocytogen, Charles River Laboratories, Crown Bioscience, Eurofins Discovery, Evotec, Fortrea, Icon Plc, IQVIA Inc., Iris Pharma, Taconic Biosciences, Inc., WuXi AppTec, etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC's report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the in-vivo CRO market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global in-vivo CRO market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the in-vivo CRO industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Frequently Asked Questions About the In-vivo CRO Market Report
The global in-vivo CRO market was valued at USD 5.7 Billion in 2025.
We expect the global in-vivo CRO market to exhibit a CAGR of 6.49% during 2026-2034.
The growing demand for in-vivo CRO, as it assists research organizations in developing effective medications at minimal costs and optimizing workflow by avoiding delays is primarily driving the global in-vivo CRO market.
The sudden outbreak of the COVID-19 pandemic has led to the increasing need for in-vivo CRO among numerous biotechnology companies to develop effective vaccines.
Based on the type, the global in-vivo CRO market can be segmented into rodent and non-rodent, where rodent currently holds the majority of the total market share.
Based on the GLP type, the global in-vivo CRO market has been divided into non-GLP and GLP toxicology. Currently, GLP toxicology exhibits a clear dominance in the market.
Based on the indication, the global in-vivo CRO market can be categorized into autoimmune/inflammation conditions, pain management, oncology, CNS conditions, diabetes, obesity, and others. Among these, oncology accounts for the largest market share.
On a regional level, the market has been classified into North America, Asia-Pacific, Europe, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global in-vivo CRO market include Biocytogen, Charles River Laboratories, Crown Bioscience, Eurofins Discovery, Evotec, Fortrea, Icon Plc, IQVIA Inc., Iris Pharma, Taconic Biosciences, Inc., and WuXi AppTec.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












