
India Aseptic Sampling Market Size, Share, Trends and Forecast by Product Type, Sampling Technique, Application, End User, and Region, 2026-2034
India Aseptic Sampling Market Summary:
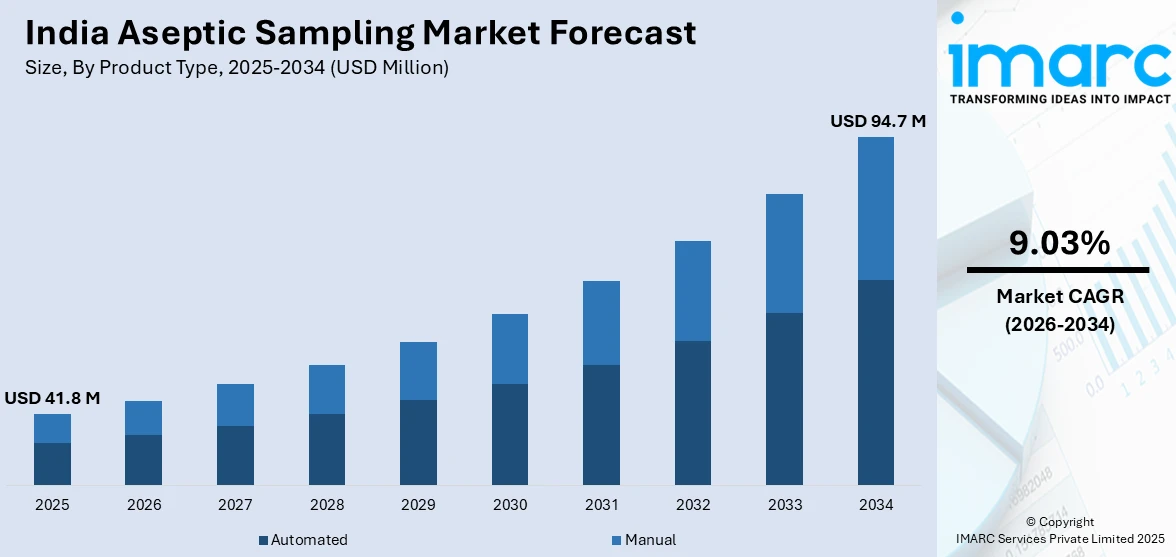
The India aseptic sampling market size was valued at USD 41.8 Million in 2025 and is projected to reach USD 94.7 Million by 2034, growing at a compound annual growth rate of 9.03% from 2026-2034.
The India aseptic sampling market is witnessing sustained expansion driven by the country’s rapidly growing biopharmaceutical manufacturing sector, increasing adoption of single-use bioprocessing technologies, and stringent regulatory mandates for sterile production environments. Government-led initiatives promoting domestic pharmaceutical self-reliance, coupled with rising investments in biologics, biosimilars, and vaccine production facilities, are strengthening demand for advanced contamination-free sampling solutions. The expanding role of contract development and manufacturing organizations and growing emphasis on quality assurance across upstream and downstream bioprocessing workflows continue to elevate India aseptic sampling market share.
Key Takeaways and Insights:
- By Product Type: Automated dominates the market with a share of 54.6% in 2025, driven by increasing demand for reproducible, high-throughput sampling in biopharmaceutical production and the shift toward Industry 4.0 manufacturing workflows.
- By Sampling Technique: Valve-based sampling leads the market with a share of 48.8% in 2025, owing to its reliability in maintaining sterile integrity during large-scale fermentation and purification operations.
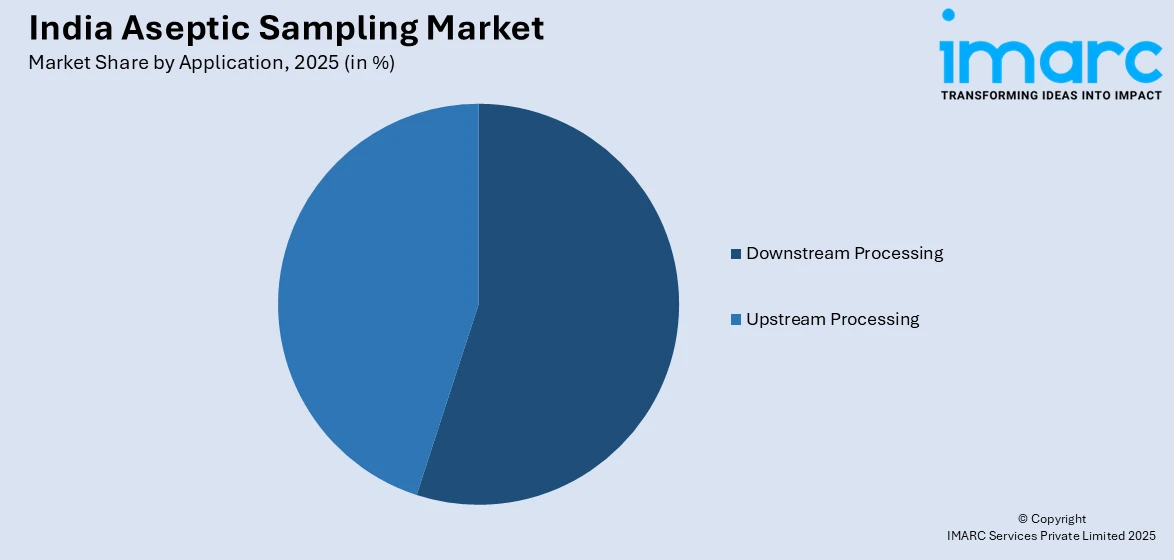
- By Application: Downstream processing represents the largest segment with a market share of 42.1% in 2025, attributed to the growing complexity of purification and formulation stages requiring frequent sterile quality checks.
- By End User: Pharmaceutical & biopharmaceutical manufacturers leads the market with a share of 56.2% in 2025, supported by large-scale investments in biologics, vaccines, and sterile drug manufacturing facilities across India.
- By Region: West India dominated the market with 34.3% revenue share in 2025, driven by the concentration of major pharmaceutical manufacturing clusters and bioprocessing infrastructure in Maharashtra and Gujarat.
- Key Players: The India aseptic sampling market exhibits a moderately consolidated competitive structure, with global bioprocessing technology providers competing alongside specialized regional suppliers. Market participants differentiate through product innovation, integration with single-use systems, and strategic partnerships with domestic biopharmaceutical manufacturers.
To get more information on this market Request Sample
The India aseptic sampling market is evolving through a combination of regulatory upgrades, technological progress, and rapid expansion of biomanufacturing capabilities. Strengthened pharmaceutical manufacturing regulations have brought domestic standards closer to global GMP norms, placing greater emphasis on sterile processing, contamination control, and in-process quality assurance. This regulatory shift has encouraged manufacturers to adopt advanced aseptic sampling systems that ensure product integrity across critical production stages. At the same time, supportive industrial policies have stimulated investments in biologics, vaccines, and sterile drug manufacturing, leading to the development of modern production facilities with higher compliance requirements. Global life sciences companies are also deepening their presence in India by establishing technical and innovation centers that highlight advanced bioprocessing and aseptic technologies. Together, these trends highlight India’s transition toward a more sophisticated biopharmaceutical manufacturing landscape, where reliable, contamination-free sampling solutions are becoming an essential component of operational excellence and regulatory compliance.
India Aseptic Sampling Market Trends:
Rising Adoption of Single-Use Aseptic Sampling Technologies
Indian biopharmaceutical manufacturers are increasingly moving away from traditional stainless-steel sampling systems toward single-use alternatives that lower cross-contamination risks and simplify validation processes. This transition is especially evident in multi-product facilities focused on biosimilars and vaccines, where fast and flexible batch changeovers are critical. Disposable aseptic sampling bags, connectors, and valve assemblies are gaining preference due to their operational efficiency and reduced downtime. As new biomanufacturing facilities come online under supportive industrial initiatives, single-use aseptic sampling solutions are becoming an integral part of modern upstream and downstream processing workflows.
Integration of Automated Sampling with Digital Biomanufacturing Platforms
The integration of automated aseptic sampling with real-time process analytical tools and digital manufacturing execution systems is transforming quality control practices across Indian bioprocessing facilities. Automated sampling allows continuous, contamination-free monitoring of critical parameters during fermentation and purification, reducing manual intervention and enhancing regulatory compliance. Growing adoption of advanced bioprocessing equipment reflects the industry’s shift toward more integrated and efficient production environments. As manufacturers increasingly adopt smart factory approaches for biologics and advanced therapies, digitized and closed-loop sampling workflows are gaining traction, enabling improved process visibility, faster decision-making, and more consistent product quality across complex manufacturing operations.
Expansion of Contract Manufacturing Driving Specialized Sampling Demand
India’s contract development and manufacturing organization sector is undergoing rapid expansion, driving rising demand for aseptic sampling equipment. As global pharmaceutical companies seek to diversify supply chains, Indian CDMOs are increasingly handling complex outsourcing programs involving biologics, sterile injectables, and antibody-drug conjugates. Ongoing investments in drug development and manufacturing capacity are leading to the addition of advanced, GMP-compliant sterile production lines. Managing multiple client projects within these facilities requires reliable aseptic sampling systems to ensure contamination control, consistent quality assurance, and regulatory compliance across parallel manufacturing operations.
Market Outlook 2026-2034:
The India aseptic sampling market is expected to witness strong growth, supported by increasing biopharmaceutical investments, ongoing regulatory upgrades, and India’s growing role as a global hub for biologics and sterile therapy manufacturing. Supportive government initiatives aimed at strengthening advanced biomanufacturing are encouraging the expansion of modern sterile production facilities across the country. At the same time, manufacturers are increasingly adopting automated and closed-system sampling technologies to improve contamination control and compliance. Expanding CDMO activity and the rising complexity of outsourced sterile manufacturing programs are likely to sustain long-term demand for reliable aseptic sampling solutions. The market generated a revenue of USD 41.8 Million in 2025 and is projected to reach a revenue of USD 94.7 Million by 2034, growing at a compound annual growth rate of 9.03% from 2026-2034.
India Aseptic Sampling Market Report Segmentation:
|
Segment Category |
Leading Segment |
Market Share |
|
Product Type |
Automated |
54.6% |
|
Sampling Technique |
Valve-Based Sampling |
48.8% |
|
Application |
Downstream Processing |
42.1% |
|
End User |
Pharmaceutical & Biopharmaceutical Manufacturers |
56.2% |
|
Region |
West India |
34.3% |
Product Type Insights:
- Manual
- Automated
Automated dominates with a market share of 54.6% of the total India aseptic sampling market in 2025.
Automated aseptic sampling systems have emerged as the predominant product type in the Indian market, driven by their ability to deliver consistent, reproducible results while minimizing human error and contamination risk in sterile manufacturing environments. These systems integrate seamlessly with process analytical technology frameworks, enabling real-time quality monitoring during critical bioprocessing stages. The growing number of large-scale biologics facilities being established under government investment programs is fueling demand for automated sampling solutions that support high-throughput production workflows.
The growing preference for automated systems is reinforced by their strong alignment with evolving regulatory expectations that emphasize validated and digitally documented manufacturing processes. Indian biopharmaceutical manufacturers engaged in the production of monoclonal antibodies, vaccines, and biosimilars increasingly depend on automated sampling solutions to ensure consistent compliance with both domestic and international quality standards. Broader industry adoption of advanced bioprocessing platforms highlights the rising importance of automation and contamination-controlled sampling.
Sampling Technique Insights:
- Valve-Based Sampling
- Needle & Septum-Based Sampling
- Bag-Based Sampling
- Others
Valve-based sampling leads the market with a share of 48.8% of the total India aseptic sampling market in 2025.
Valve-based aseptic sampling maintains its leadership position owing to its proven reliability in maintaining sterile barrier integrity during repeated sampling cycles in large-volume bioreactors and purification trains. These systems are particularly favored in established stainless-steel manufacturing facilities where they integrate with existing piping infrastructure while providing validated sterile access points. The technique’s widespread adoption across India’s mature pharmaceutical manufacturing base, including vaccine production facilities operated by major domestic producers, underpins its dominant market position.
The continued expansion of sterile injectable and biologics manufacturing capacity across India is sustaining demand for valve-based sampling solutions. As facilities scale production to meet both domestic healthcare needs and growing export obligations, the requirement for robust, reusable sampling valves capable of withstanding sterilization-in-place and clean-in-place cycles remains strong. The integration of valve-based systems with automated actuators and electronic monitoring further enhances their appeal in facilities transitioning toward smart manufacturing environments aligned with Industry 4.0 principles.
Application Insights:
Access the comprehensive market breakdown Request Sample
- Downstream Processing
- Upstream Processing
Downstream processing represents the largest share with 42.1% of the total India aseptic sampling market in 2025.
The downstream processing segment commands the largest application share, driven by the inherent complexity of purification, filtration, chromatography, and formulation stages that demand frequent sterile quality assessments. As Indian biopharmaceutical manufacturers expand their biosimilar and monoclonal antibody production portfolios, the number of critical quality checkpoints in downstream workflows has increased substantially. Each purification step requires verified aseptic sampling to ensure product integrity and regulatory compliance before proceeding to subsequent processing stages.
The increasing focus on implementing process analytical technologies in Indian bioprocessing facilities is driving higher demand for downstream aseptic sampling solutions. Modern purification approaches, including continuous processing and advanced filtration techniques, require frequent in-process sampling under strict sterile conditions. As downstream bioprocessing infrastructure becomes more sophisticated, manufacturers must ensure reliable contamination control throughout purification and formulation stages. This growing complexity in downstream operations is strengthening the need for robust aseptic sampling systems that support real-time monitoring, process consistency, and compliance with quality and regulatory expectations.
End User Insights:
- Pharmaceutical & Biopharmaceutical Manufacturers
- CMOs / CDMOs
- Others
Pharmaceutical and biopharmaceutical manufacturers lead the market with a share of 56.2% of the total India aseptic sampling market in 2025.
Pharmaceutical and biopharmaceutical manufacturers represent the dominant end-user segment, reflecting India’s substantial base of vertically integrated drug production companies operating sterile manufacturing facilities for vaccines, biologics, and injectable formulations. These manufacturers maintain dedicated quality assurance laboratories requiring continuous aseptic sampling capabilities across multiple production lines. The PLI scheme for pharmaceuticals has catalyzed significant capacity expansion, with cumulative sales of approved pharmaceutical products surpassing INR 2.66 lakh crore by March 2025, driving proportional demand for sampling infrastructure.
India’s position as the world’s largest vaccine supplier, producing over sixty percent of global vaccines, creates sustained baseline demand for aseptic sampling across biopharmaceutical manufacturing operations. The country hosts over 500 US FDA-approved pharmaceutical manufacturing facilities, the highest count for any country outside the United States, each requiring validated sterile sampling protocols. Growing domestic biologics pipelines targeting oncology, autoimmune disorders, and rare diseases continue to expand the installed base of aseptic sampling equipment within this end-user category.
Regional Insights:
- North India
- South India
- East India
- West India
West India exhibits a clear dominance with a 34.3% share of the total India aseptic sampling market in 2025.
West India maintains its leadership position driven by the concentration of pharmaceutical manufacturing hubs in Maharashtra and Gujarat, which collectively host a significant proportion of India’s sterile drug production and biologics manufacturing infrastructure. Maharashtra’s established pharmaceutical corridor, anchored around Pune and Mumbai, accommodates numerous large-scale sterile manufacturing facilities producing vaccines, injectables, and biotherapeutics that require extensive aseptic sampling capabilities. Gujarat’s emergence as a center for bulk drug manufacturing, supported by the government-approved Bulk Drug Park, further strengthens the region’s demand for sterile processing and sampling equipment.
The region benefits from well-developed logistics networks, proximity to major ports facilitating equipment imports, and an established ecosystem of pharmaceutical suppliers and service providers. The Gujarat Bulk Drug Park, one of three parks approved under the central government scheme with total project costs exceeding INR 6,300 crore, is expected to create additional sterile manufacturing capacity that will generate incremental demand for aseptic sampling solutions. Additionally, the presence of major CDMO operations and biopharmaceutical research centers in western India continues to sustain the region’s dominant market position.
Market Dynamics:
Growth Drivers:
Why is the India Aseptic Sampling Market Growing?
Government-Led Regulatory Modernization and GMP Harmonization
India’s pharmaceutical regulatory framework is undergoing a major transformation that is directly driving demand for aseptic sampling technologies. Updated good manufacturing practice requirements have brought domestic standards closer to global benchmarks, placing stronger emphasis on sterile manufacturing controls, robust quality systems, and risk-based compliance approaches. Manufacturers are now required to adopt validated processes supported by reliable documentation and digital traceability. These changes are compelling pharmaceutical producers, particularly those engaged in sterile and vaccine manufacturing, to strengthen contamination control measures. As a result, the adoption of validated aseptic sampling procedures is becoming essential for achieving regulatory compliance and sustaining long-term operational readiness.
Rapid Expansion of Biopharmaceutical and Vaccine Manufacturing Capacity
India’s biopharmaceutical manufacturing sector is undergoing rapid capacity expansion, supported by incentive-driven industrial policies, rising global demand for cost-efficient biologics, and the country’s growing role as a preferred outsourcing destination. Supportive government initiatives are encouraging the development of new biologics production facilities and the scaling of existing operations. This expansion is strengthening India’s position in the global biopharmaceutical supply chain while increasing the complexity of manufacturing environments. India’s pharmaceutical exports have grown by ninety-two percent over six financial years, reaching INR 2,45,962 crore in FY 2024-25, with sterile products representing an increasing share of this output. Each new biologics, vaccine, or sterile injectable production line requires integrated aseptic sampling systems for in-process quality control, creating a direct correlation between manufacturing capacity expansion and sampling equipment demand.
Growing CDMO Sector Demanding Advanced Sterile Processing Infrastructure
The explosive growth of India’s contract development and manufacturing organization sector is generating significant incremental demand for aseptic sampling equipment. Global pharmaceutical companies are actively diversifying supply chains, with Indian CDMOs witnessing up to fifty percent year-on-year increases in requests for proposals during 2024 as clients seek alternatives to geographically concentrated manufacturing dependencies. The India CDMO sector is projected to double in size over the coming years, with firms expanding into high-value modalities including biologics, antibody-drug conjugates, and sterile injectables. For instance, in January 2025, Shilpa Biologicals inaugurated a new bioconjugation manufacturing facility in Karnataka, joining the global league of CDMOs offering integrated antibody-drug conjugate services. Each CDMO facility operating sterile production lines requires dedicated aseptic sampling systems to maintain compliance across multiple client programs, multiplying equipment demand proportionally with sector expansion.
Market Restraints:
What Challenges the India Aseptic Sampling Market is Facing?
High Capital Investment Requirements for Advanced Automated Systems
The significant upfront costs associated with procuring, installing, and validating automated aseptic sampling systems present a barrier for small and medium-sized pharmaceutical manufacturers in India. These enterprises, which constitute a substantial portion of the domestic manufacturing base, often face financial constraints when upgrading from manual sampling methods to automated alternatives that require specialized infrastructure, trained personnel, and ongoing maintenance expenditure.
Limited Availability of Skilled Technical Personnel
The effective operation and maintenance of sophisticated aseptic sampling systems demands specialized technical expertise that remains in limited supply across the Indian pharmaceutical workforce. High attrition rates in the pharmaceutical manufacturing sector compound this challenge, making it difficult for facilities to retain trained operators capable of managing advanced bioprocessing equipment and ensuring compliance with stringent aseptic protocols.
Supply Chain Dependencies on Imported Components
A substantial proportion of critical aseptic sampling components and consumables, including specialized membranes, connectors, and sensor technologies, are sourced from international suppliers. This import dependency exposes Indian manufacturers to supply chain disruptions, currency fluctuations, and extended procurement lead times that can impact production continuity and increase overall operational costs for sterile manufacturing facilities.
Competitive Landscape:
The India aseptic sampling market is characterized by a moderately consolidated competitive structure in which established global bioprocessing technology providers hold significant market positions through comprehensive product portfolios and integrated service offerings. These international players leverage extensive research and development capabilities, global manufacturing networks, and established distribution partnerships with Indian pharmaceutical companies to maintain competitive advantages. Market participants are increasingly differentiating through the development of integrated automated sampling solutions compatible with single-use bioprocessing workflows, digital manufacturing platforms, and continuous processing technologies. Strategic investments in local manufacturing, application centers, and customer support infrastructure are becoming critical competitive levers as companies seek to deepen their presence in the rapidly expanding Indian biopharmaceutical market. Regional distributors and niche domestic suppliers complement the competitive landscape by offering cost-effective alternatives and localized technical support to smaller manufacturers.
India Aseptic Sampling Market Report Coverage:
|
Report Features |
Details |
|
Base Year of the Analysis |
2025 |
|
Historical Period |
2020-2025 |
|
Forecast Period |
2026-2034 |
|
Units |
USD Million |
|
Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
|
Product Types Covered |
Manual, Automated |
|
Sampling Techniques Covered |
Valve-Based Sampling, Needle & Septum-Based Sampling, Bag-Based Sampling, Others |
|
Applications Covered |
Downstream Processing, Upstream Processing |
|
End Users Covered |
Pharmaceutical & Biopharmaceutical Manufacturers, CMOs / CDMOs, Others |
|
Regions Covered |
North India, South India, East India, West India |
|
Customization Scope |
10% Free Customization |
|
Post-Sale Analyst Support |
10-12 Weeks |
|
Delivery Format |
PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report
The India aseptic sampling market size was valued at USD 41.8 Million in 2025.
The India aseptic sampling market is expected to grow at a compound annual growth rate of 9.03% from 2026-2034 to reach USD 94.7 Million by 2034.
The automated aseptic sampling represents the largest market share of 54.6% in 2025, driven by increasing demand for reproducible, contamination-free sampling in biopharmaceutical manufacturing and the shift toward digitized quality control workflows.
Key factors driving the India aseptic sampling market include government-led GMP regulatory modernization aligned with WHO standards, rapid expansion of biopharmaceutical and vaccine manufacturing capacity under PLI incentive programs, and growing CDMO sector demand for advanced sterile processing infrastructure.
Major challenges include high capital investment requirements for automated sampling systems, limited availability of skilled technical personnel for operating advanced bioprocessing equipment, and supply chain dependencies on imported components and consumables.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












