
India Pharmaceutical Filtration Market Size, Share, Trends and Forecast by Product, Technique, Application, Scale of Operation, and Region, 2026-2034
India Pharmaceutical Filtration Market Summary:
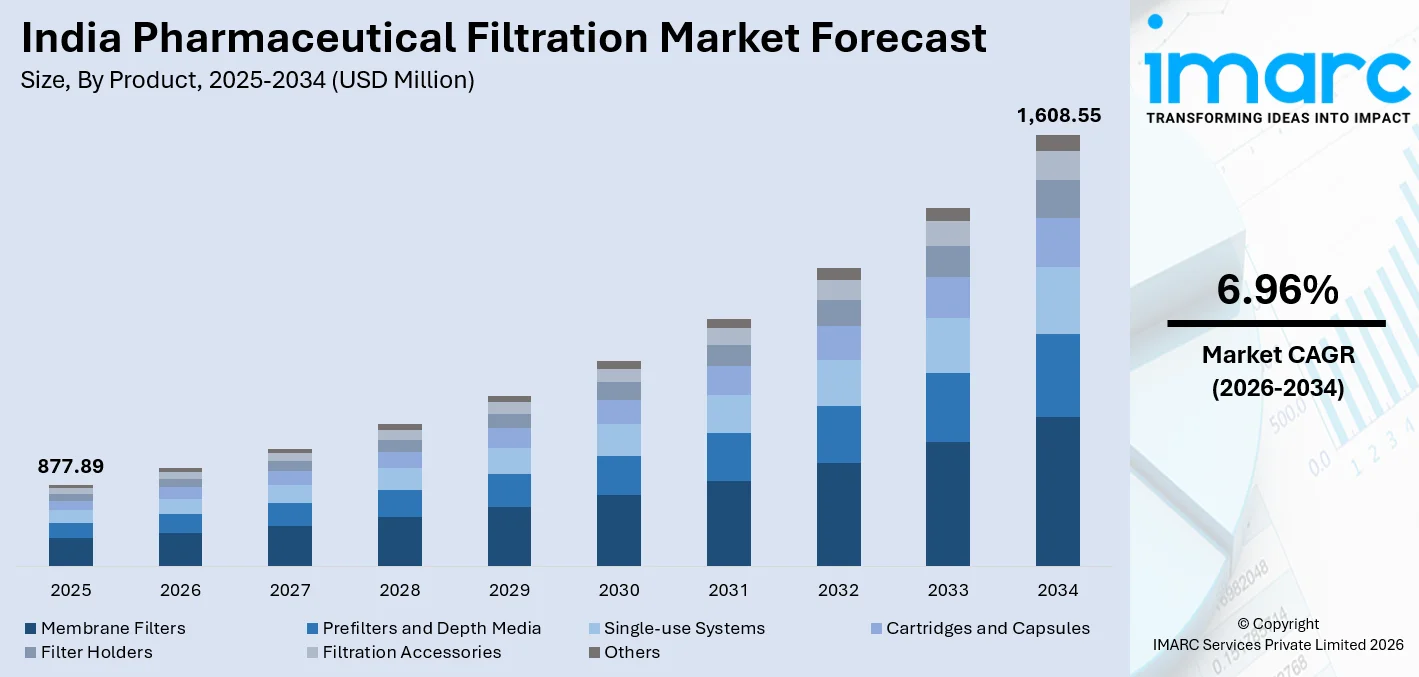
The India pharmaceutical filtration market size was valued at USD 877.89 Million in 2025 and is projected to reach USD 1,608.55 Million by 2034, growing at a compound annual growth rate of 6.96% from 2026-2034.
India's pharmaceutical filtration market is expanding steadily, driven by the country's emergence as a leading global drug manufacturing destination. Increasing production of biologics, biosimilars, and sterile injectables is generating strong demand for advanced filtration technologies. Supportive government manufacturing policies, rising pharmaceutical export volumes, and increasingly rigorous quality compliance requirements are reinforcing industry standards and infrastructure investment, collectively contributing to the growth of the India pharmaceutical filtration market share.
Key Takeaways and Insights:
- By Product: Membrane filters dominate the market with a share of 33% in 2025, owing to their widespread adoption across sterile filtration, bioprocessing, and protein purification applications throughout India’s pharmaceutical manufacturing sector.
- By Technique: Microfiltration leads the market with a share of 25% in 2025, driven by its effectiveness in removing bacteria, particulates, and contaminants across upstream and downstream pharmaceutical bioprocessing operations.
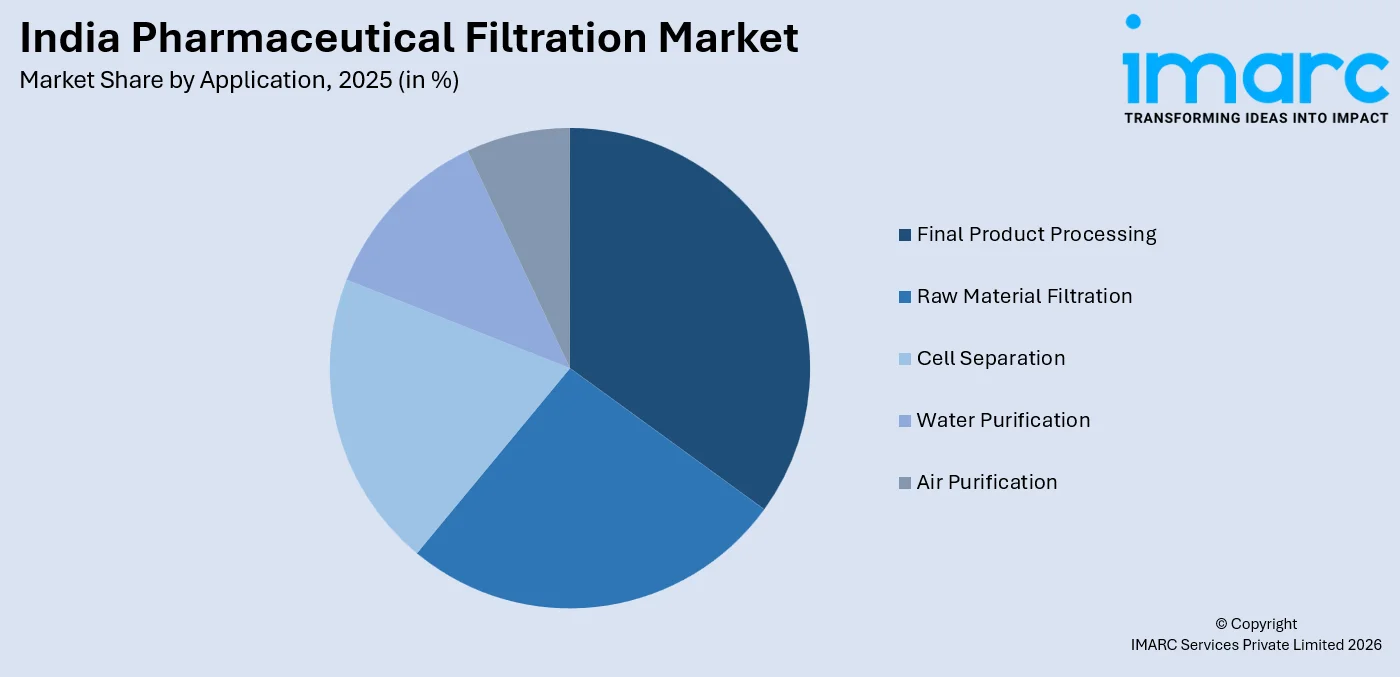
- By Application: Final product processing holds the largest share at 28% in 2025, as it encompasses critical end-stage drug manufacturing steps including sterile filtration, active pharmaceutical ingredient (API) purification, and formulation filling.
- By Scale of Operation: Manufacturing scale dominates the market with a share 50% in 2025, reflecting the large-scale production demands of India’s rapidly growing generic drug and biopharmaceutical manufacturing base.
- Key Players: The India pharmaceutical filtration market is moderately competitive, with global and domestic manufacturers offering diverse product portfolios encompassing membrane filters, single-use systems, and depth filtration solutions for pharmaceutical and biopharmaceutical applications.
To get more information on this market Request Sample
India’s pharmaceutical filtration market is steadily evolving as the industry transitions toward higher-value drug categories including biologics, biosimilars, and specialty therapeutics. In the 2026 Union Budget, the government announced a dedicated ₹10,000‑crore Biopharma SHAKTI initiative over five years to strengthen India’s biologics and biosimilars manufacturing ecosystem, bolstering infrastructure and competitiveness in advanced therapies. The growing need for contamination-free, sterile pharmaceutical production environments is reinforcing demand for precision filtration systems across the manufacturing chain. Government policy initiatives aimed at expanding domestic drug manufacturing capacity, reducing import dependence, and strengthening export competitiveness are contributing to sustained infrastructure investment. Simultaneously, the rising burden of chronic diseases within the Indian population is accelerating pharmaceutical production volumes, amplifying the need for robust filtration processes that ensure product safety and regulatory compliance. The expansion of contract manufacturing organizations and the growing influence of international regulatory standards are collectively shaping market dynamics, driving sustained investment across the filtration value chain in India.
India Pharmaceutical Filtration Market Trends:
Rising Adoption of Single-Use Filtration Technologies
India's pharmaceutical industry is increasingly integrating single-use filtration systems as manufacturing operations expand in scale and complexity. In June 2025, Mumbai‑based biotech manufacturer PharmNXT Biotech began supplying domestically produced single‑use bioprocess systems, including filtration components, to major vaccine producers like the Serum Institute of India, reducing reliance on imported consumables and accelerating local biologics production. These technologies eliminate the need for extensive cleaning validation, reduce cross-contamination risks between production batches, and offer operational flexibility suited to multi-product biopharmaceutical environments. Their growing adoption reflects a broader industry transition toward leaner, compliance-oriented production models that support faster manufacturing turnaround and more efficient facility utilization across pharmaceutical and biopharmaceutical operations.
Growing Emphasis on Compliance with International Regulatory Standards
Indian pharmaceutical manufacturers are placing heightened emphasis on meeting international regulatory requirements to maintain and grow their presence in global export markets. In December 2025, Granules India’s US packaging unit cleared a GMP inspection with zero observations, underscoring how Indian companies are investing in quality systems that satisfy stringent standards like those of the U.S. Food and Drug Administration (USFDA). This focus is accelerating investment in validated, high-performance filtration technologies that satisfy Good Manufacturing Practice standards enforced by leading regulatory authorities. Strengthening quality assurance frameworks and ensuring consistent contamination control are central priorities, directly driving the adoption of advanced filtration systems and supporting sustained India pharmaceutical filtration market growth.
Expansion of Biologics and Biosimilars Manufacturing
India's accelerating investment in biologics and biosimilars production is creating substantial demand for specialized pharmaceutical filtration technologies. For example, in 2025, Indian biotech major Biocon fully integrated its biosimilars business into the parent entity in a strategic $5.5 billion merger aimed at scaling biologics capabilities and strengthening its global presence. The manufacturing of biopharmaceuticals involves complex purification, separation, and sterilization workflows that require precise, high-performance filtration at multiple process stages. This expansion is broadening the relevance of membrane filters, crossflow filtration, nanofiltration, and ultrafiltration systems across commercial-scale biopharma facilities, reinforcing the strategic importance of filtration infrastructure throughout India's evolving pharmaceutical manufacturing landscape.
Market Outlook 2026-2034:
The India pharmaceutical filtration market is positioned for robust long-term growth, supported by the country's rapidly evolving pharmaceutical manufacturing landscape and an intensifying focus on biologics, specialty drugs, and sterile injectables. Growing regulatory demands, expanding pharmaceutical export volumes, and sustained investment in advanced drug manufacturing infrastructure are expected to drive consistent demand for filtration technologies across production scales. Rising domestic healthcare expenditure, increasing chronic disease burden, and expanding drug consumption within India's large population further reinforce strong and durable market fundamentals throughout the forecast period. The market generated a revenue of USD 877.89 Million in 2025 and is projected to reach a revenue of USD 1,608.55 Million by 2034, growing at a compound annual growth rate of 6.96% from 2026-2034.
India Pharmaceutical Filtration Market Report Segmentation:
| Segment Category | Leading Segment | Market Share |
|---|---|---|
| Product | Membrane Filters | 33% |
| Technique | Microfiltration | 25% |
| Application | Final Product Processing | 28% |
| Scale of Operation | Manufacturing Scale | 50% |
Product Insights:
- Membrane Filters
- MCE Membrane Filters
- Coated Cellulose Acetate Membrane Filters
- PTFE Membrane Filters
- Nylon Membrane Filters
- PVDF Membrane Filters
- Others
- Prefilters and Depth Media
- Glass Fiber Filters
- PTFE Fiber Filters
- Single-use Systems
- Cartridges and Capsules
- Filter Holders
- Filtration Accessories
- Others
The membrane filters dominate with a market share of 33% of the total India pharmaceutical filtration market in 2025.
Membrane filters are among the most versatile and extensively utilized filtration products in pharmaceutical manufacturing. Their proven capability to remove microorganisms, particulates, and contaminants from liquid and gas streams makes them indispensable in sterile drug production environments. Multiple variants are available, including MCE, PTFE, nylon, PVDF, and cellulose acetate membranes, each designed for specific pharmaceutical applications such as protein purification, virus filtration, sterile filtration, and buffer clarification, offering manufacturers flexibility across diverse production requirements.
In India's pharmaceutical landscape, membrane filters serve a critical role across upstream and downstream bioprocessing workflows. Their broad compatibility with solvents and biological fluids, combined with straightforward validation processes and strong regulatory acceptance, establishes them as the preferred solution for sterile filtration in injectable drug manufacturing. Expanding biologics production, growing biosimilar development activities, and increasingly stringent quality standards are further accelerating adoption among large-scale pharmaceutical manufacturers and contract development and manufacturing organizations operating across the country.
Technique Insights:
- Microfiltration
- Ultrafiltration
- Crossflow Filtration
- Nanofiltration
- Others
The microfiltration leads with a share of 25% of the total India pharmaceutical filtration market in 2025.
Microfiltration is a widely utilized separation technique in pharmaceutical manufacturing, effectively removing bacteria, particulates, and suspended solids from liquid streams without damaging sensitive biomolecules. Its applications span upstream and downstream bioprocessing workflows, encompassing cell culture clarification, process fluid purification, and sterile drug preparation. The technique's compatibility with pharmaceutical-grade membranes, operational simplicity, and ease of process scalability contribute to its broad and consistent adoption across diverse pharmaceutical manufacturing facilities and production environments.
In the context of India's rapidly expanding pharmaceutical industry, microfiltration plays a foundational role across both generic drug manufacturing and advanced biopharmaceutical production operations. The technique is particularly critical in ensuring sterility and purity in injectable drug formulations, which are subject to rigorous domestic and international regulatory scrutiny. Sustained investment in pharmaceutical manufacturing infrastructure, combined with growing compliance requirements enforced by global regulatory authorities, continues to drive strong and consistent demand for microfiltration technologies throughout India's pharmaceutical sector.
Application Insights:
Access the comprehensive market breakdown Request Sample
- Final Product Processing
- Active Pharmaceutical Ingredient Filtration
- Sterile Filtration
- Protein Purification
- Vaccines and Antibody Processing
- Formulation and Filling Solutions
- Viral Clearance
- Raw Material Filtration
- Media Buffer
- Pre-Filtration
- Bioburden Testing
- Cell Separation
- Water Purification
- Air Purification
The final product processing dominates with a market share of 28% of the total India pharmaceutical filtration market in 2025.
Final product processing encompasses critical filtration steps performed during the later stages of pharmaceutical manufacturing, including sterile filtration of injectable formulations, active pharmaceutical ingredient purification, protein purification, and viral clearance. These processes are governed by the most stringent regulatory standards, as they directly determine product safety and therapeutic efficacy. Growing production of sterile injectables and biologics across India's expanding pharmaceutical sector is intensifying demand for reliable, validated filtration solutions at this manufacturing stage.
India's pharmaceutical sector has made considerable strides in building capabilities for high-value biologics, vaccines, and antibody-based therapy production, all of which require sophisticated end-product filtration processes. Meeting international standards for sterile filtration and viral clearance is essential for sustaining access to regulated global export markets. Rising investment in fill-finish manufacturing infrastructure, combined with increasing demand for precision formulation capabilities, is driving sustained and broad-based market expansion within India's final product processing segment.
Scale of Operation Insights:
- Manufacturing Scale
- Pilot-scale
- Research and Development Scale
The manufacturing scale leads with a share of 50% of the total India pharmaceutical filtration market in 2025.
Manufacturing-scale filtration represents the largest and most investment-intensive segment within the India pharmaceutical filtration market. Large-scale production facilities demand high-throughput, rigorously validated filtration systems capable of processing substantial volumes while consistently maintaining stringent quality, sterility, and contamination control standards. The segment's dominance is closely linked to India's well-established position as the world's largest generic medicine supplier and its rapidly growing strategic role as a global manufacturing hub for biologics, biosimilars, and vaccine production.
Continued government support through production-linked incentive schemes, bulk drug park development, and dedicated pharmaceutical manufacturing zone investments is accelerating capacity expansion across large-scale operations. As India's pharmaceutical sector deepens its global integration through growing exports and expanding contract manufacturing engagements, the requirement for internationally validated, scalable filtration technologies continues to intensify. This sustained momentum generates consistent demand for advanced membrane systems, single-use filtration assemblies, and fully integrated filtration platforms across commercial manufacturing environments.
Regional Insights:
- North India
- West and Central India
- South India
- East and Northeast India
North India hosts a significant concentration of pharmaceutical manufacturing facilities, particularly across Himachal Pradesh and Uttarakhand, which serve as established centers for tablet, capsule, and injectable drug production. The region's well-developed industrial infrastructure, skilled workforce, and favorable policy environment continue to support steady pharmaceutical sector expansion and filtration technology adoption.
West and Central India, encompassing Maharashtra and Gujarat, represent major pharmaceutical and API manufacturing hubs. The dense concentration of formulation plants, bulk drug production clusters, and biotechnology facilities across these states generates substantial and diversified demand for filtration technologies spanning large-scale manufacturing and contract research operations.
South India, particularly Telangana, Andhra Pradesh, and Karnataka, has emerged as a prominent pharmaceutical and biopharmaceutical manufacturing destination. The region's expanding research and development ecosystem, export-oriented production facilities, and government-backed pharma park development are collectively driving strong demand for advanced filtration solutions across the manufacturing value chain.
East and Northeast India represent emerging pharmaceutical manufacturing territories, supported by government-led industrial and healthcare infrastructure development initiatives. Growing domestic healthcare consumption, increasing investment in regional pharmaceutical capacity, and improving connectivity are gradually broadening filtration technology adoption across these strategically important and developing markets.
Market Dynamics:
Growth Drivers:
Why is the India Pharmaceutical Filtration Market Growing?
Expansion of the Pharmaceutical Manufacturing Sector
India has firmly established itself as one of the world’s leading pharmaceutical manufacturing nations, producing a vast array of generic medicines, active pharmaceutical ingredients, biosimilars, and vaccines for both domestic consumption and global export markets. To further bolster manufacturing infrastructure and reduce import dependence, the Government of India reopened the sixth application window under its Production‑Linked Incentive (PLI) Scheme for bulk drug manufacturing in 2025, encouraging investment in APIs, intermediates, and related high‑value production facilities. The continued expansion of manufacturing capacity, supported by government-led industrial schemes, dedicated pharmaceutical zone development, and investment in bulk drug parks, is creating sustained and diversified demand for high-quality filtration solutions across the entire production chain. As facilities scale up operations to serve both domestic and international markets, the need for reliable, validated, and regulatory-compliant filtration systems increases proportionally. This ongoing manufacturing expansion is contributing to robust adoption of diverse filtration technologies, including membrane filters, single-use systems, and depth filtration platforms, across operations of all scales.
Rising Demand for Biologics and Biopharmaceuticals
The global shift toward biologics, monoclonal antibodies, and biosimilars is significantly influencing India’s pharmaceutical production landscape. For instance, in June 2024, Biocon Biologics received European Medicines Agency approval to manufacture biosimilar Bevacizumab at its new monoclonal antibodies facility in Bengaluru, underscoring India’s growing capacity to produce high‑value biologics for regulated global markets. Biopharmaceutical manufacturing is inherently filtration-intensive, requiring precise separation, purification, and sterilization processes across multiple production stages to ensure product safety, potency, and regulatory compliance. India is rapidly developing capabilities to produce high-value biologics for both domestic healthcare markets and export to developed countries, supported by targeted government investment and growing industry partnerships. This transition from small-molecule generics to complex biopharmaceuticals is creating sustained demand for more sophisticated and technically advanced filtration solutions, including ultrafiltration, nanofiltration, crossflow filtration, and sterile-grade membrane systems.
Stringent Regulatory Requirements and Quality Standards
The pharmaceutical manufacturing landscape in India is increasingly governed by stringent domestic and international regulatory frameworks. In 2025, Indian pharmaceutical companies saw a notable decrease in serious adverse findings (“Official Action Indicated” or OAI) during U.S. Food and Drug Administration inspections, with OAI cases nearly halving compared to the previous year, reflecting stronger compliance with global quality expectations. Compliance with standards set by regulatory authorities for good manufacturing practices, sterility assurance, and contamination control requires pharmaceutical manufacturers to invest in reliable, validated filtration solutions across every stage of production. The growing emphasis on pharmaceutical export quality, driven by scrutiny from international regulatory agencies, is compelling Indian manufacturers to adopt internationally recognized filtration technologies. Organizations pursuing regulatory approvals for export to regulated markets must demonstrate consistent and documented filtration performance as part of their quality management systems. This regulatory-driven dynamic is a fundamental and enduring catalyst for sustained filtration adoption across both established pharmaceutical manufacturers and newer entrants in India’s industry.
Market Restraints:
What Challenges the India Pharmaceutical Filtration Market is Facing?
High Cost of Advanced Filtration Technologies
Advanced pharmaceutical filtration systems, particularly those designed for biopharmaceutical applications, involve significant capital and operational expenditure. Smaller and mid-sized pharmaceutical manufacturers may face affordability barriers when seeking to upgrade or implement internationally compliant filtration infrastructure. The cost of specialized membrane materials, single-use assemblies, and validated filtration systems can constrain adoption, particularly among emerging manufacturers operating with limited budgets and less access to capital for infrastructure investment and process modernization.
Dependence on Imported Filtration Components
India’s pharmaceutical filtration market relies substantially on imported membranes, filter media, and specialized consumables from global suppliers. This dependence introduces supply chain vulnerabilities, longer lead times, and exposure to currency fluctuation risks, which can disrupt production schedules and increase operational costs. Developing a robust domestic filtration component supply chain remains a critical priority for strengthening market self-reliance and securing continuity for pharmaceutical manufacturing.
Filter Fouling and Maintenance Requirements
Filter fouling represents a persistent operational challenge in pharmaceutical filtration processes, where accumulated particulates and microbial matter reduce membrane permeability, flow rates, and filtration efficiency. Frequent filter replacement and the associated validation, quality testing, and documentation requirements increase operational burdens and costs. Managing fouling effectively requires specialized process knowledge and skilled personnel, posing capacity constraints for manufacturers with limited technical expertise and process engineering resources.
Competitive Landscape:
The India pharmaceutical filtration market features a dynamic competitive environment shaped by the presence of global filtration technology providers and emerging domestic manufacturers. Leading international players offer comprehensive portfolios spanning membrane filters, single-use systems, depth filtration products, and integrated filtration platforms designed specifically for biopharmaceutical and sterile drug manufacturing. These organizations leverage their global research and development capabilities, established regulatory expertise, and broad distribution networks to maintain strong market positions across India’s pharmaceutical manufacturing hubs. Domestic manufacturers and regional suppliers are increasingly carving out competitive space by offering cost-effective alternatives tailored to India’s large generic drug manufacturing base. Competitive dynamics are being shaped by investments in product innovation, expansion of technical service capabilities, and strategic partnerships with pharmaceutical manufacturers. Growing demand for customized, scalable filtration solutions across both commercial manufacturing and contract development operations is intensifying market competition, with players prioritizing technological differentiation, process efficiency, and regulatory compliance support.
Recent Developments:
- In February 2026, India’s pharma innovator Zydus Lifesciences launched Anyra™, the country’s first indigenously developed Aflibercept 2 mg biosimilar for retinal diseases, boosting access to biologics and expected to spur demand for advanced filtration in large‑scale biologics manufacturing.
India Pharmaceutical Filtration Market Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Million USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Products Covered |
|
| Techniques Covered | Microfiltration, Ultrafiltration, Crossflow Filtration, Nanofiltration, Others |
| Applications Covered |
|
| Scale of Operations Covered | Manufacturing Scale, Pilot-scale, Research and Development Scale |
| Regions Covered | North India, West and Central India, South India, East and Northeast India |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report
The India pharmaceutical filtration market size was valued at USD 877.89 Million in 2025.
The India pharmaceutical filtration market is expected to grow at a compound annual growth rate of 6.96% from 2026-2034 to reach USD 1,608.55 Million by 2034.
Membrane filters held the largest share at 33%, driven by their extensive adoption across sterile filtration, protein purification, viral clearance, and bioprocessing applications. Their versatility, broad regulatory acceptance, and compatibility with diverse pharmaceutical manufacturing workflows establish them as the preferred filtration product across India's pharmaceutical and biopharmaceutical production operations.
Key factors driving the India pharmaceutical filtration market include expanding pharmaceutical manufacturing capacity, rising demand for biologics and biosimilars, stringent regulatory compliance requirements, government support through production incentive schemes, and growing pharmaceutical exports.
Major challenges include high capital costs of advanced filtration technologies, dependence on imported filtration components and materials, filter fouling and maintenance requirements, and the need for skilled technical personnel to manage complex filtration validation processes.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












