
India Real World Evidence Solutions Market Size, Share, Trends and Forecast by Component, Therapeutic Area, End User, and Region, 2026-2034
India Real World Evidence Solutions Market Summary:
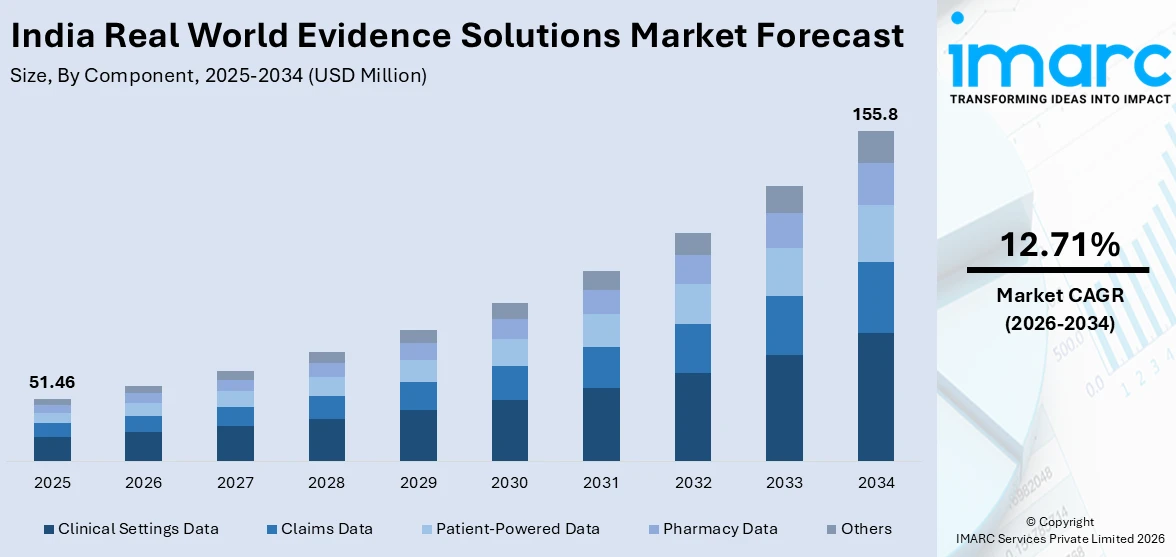
The India real world evidence solutions market was valued at USD 51.46 Million in 2025 and is projected to reach USD 155.8 Million by 2034, growing at a compound annual growth rate of 12.71% during 2026-2034.
The market is witnessing strong growth fueled by the government's bold Ayushman Bharat Digital Mission, which is establishing unparalleled collections of longitudinal patient information across both public and private healthcare institutions. The growing burden of chronic and non-communicable diseases, coupled with the pharmaceutical sector's intensifying focus on evidence-based drug development and regulatory submissions, is driving the demand for real world evidence (RWE) solutions. Increasing investments by multinational and domestic pharmaceutical companies in clinical research and post-market surveillance further expand the India real world evidence solutions market share.
Key Takeaways and Insights:
- By Component: Clinical settings data holds the largest share of 34.6% in 2025, driven by the widespread integration of electronic health records across hospital networks and the digitization of clinical encounters under national health programs.
- By Therapeutic Area: Oncology dominates the market with a 29.8% share in 2025, underpinned by India's significant and growing cancer burden, expanding oncology clinical trials activity, and pharma companies' urgent need for RWE to support drug approvals in this therapeutic area.
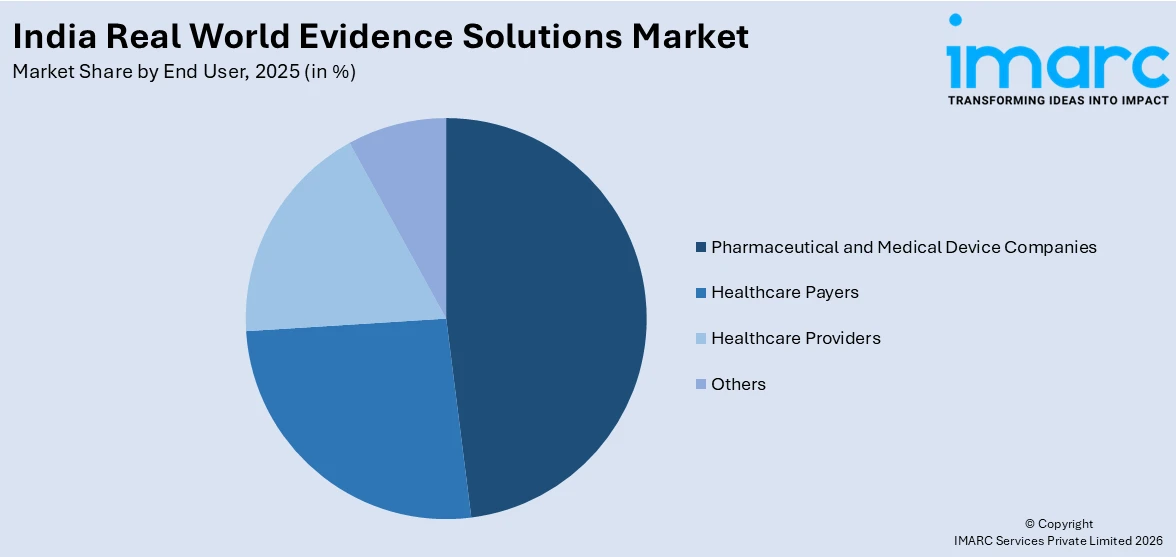
- By End User: Pharmaceutical and medical device companies represent the largest segment with a market share of 47.2% in 2025, as these organizations are the primary consumers of RWE for regulatory submissions, post-market surveillance, and market access strategies.
- By Region: South India represents the largest regional share at 31.5% in 2025, attributed to the concentration of pharmaceutical and biotechnology companies, advanced tertiary hospital infrastructure, and active clinical research ecosystems in cities such as Hyderabad, Chennai, and Bengaluru.
- Key Players: The Indian market features a competitive mix of global analytics leaders, contract research organizations, and specialized health informatics firms leveraging advanced analytics and AI-enabled platforms to serve pharmaceutical and healthcare clients.
To get more information on this market Request Sample
The India real world evidence solutions market is undergoing a fundamental transformation as the confluence of digital health policy, pharmaceutical innovation, and data science capabilities reshapes how clinical and health outcomes evidence is generated. The national push toward digitization of health records is creating structured data ecosystems that were previously unavailable, providing rich longitudinal datasets essential for generating credible RWE. Moreover, the government is increasingly investing in drug approval procedures. In 2026, the Union government is developing a strategy to reform drug sampling and testing methods to curb corruption the pharmaceutical regulatory process. The evolving regulatory environment, with growing awareness of RWE's role in drug approvals and safety monitoring at the Central Drugs Standard Control Organisation level, is prompting pharmaceutical companies and healthcare payers to invest in robust real-world data collection and analytics infrastructure. Simultaneously, India's emergence as a leading global destination for clinical trials is amplifying demand for solutions that can bridge traditional trial data with real-world patient outcomes across diverse population subgroups.
India Real World Evidence Solutions Market Trends:
Rapid Expansion of Government Policies Creating Structured Real-World Data Repositories
India's Ayushman Bharat Digital Mission (ABDM) is emerging as a foundational catalyst for RWE data generation. As of February 2025, more than 73.98 Crore Ayushman Bharat Health Account (ABHA) IDs had been created, with over 49.06 Crore health records linked across more than 1.59 Lakh ABDM-enabled health facilities. This national-scale digitization of patient encounters is producing longitudinal health records that serve as high-value real-world data sources, enabling pharmaceutical companies and researchers to conduct population-level evidence studies with unprecedented geographic and demographic coverage.
Integration of Artificial Intelligence (AI) and Machine Learning (ML) into RWE Analytics Platforms
The incorporation of artificial intelligence and machine learning is fundamentally enhancing how real-world data is processed and translated into actionable evidence in India. In 2026, Union Minister of State for Health and Family Welfare Anupriya Patel emphasized the revolutionary impact of artificial intelligence (AI) in the healthcare sector. During the AI Impact Summit in the national capital, Patel stated that India's governance framework sees AI as a facilitator and enhancer, moving the nation nearer to achieving inclusivity and health equity. Within the One Health Mission, she emphasized the rollout of an AI instrument by the Indian Council of Medical Research that examines genomes and forecasts zoonotic outbreaks prior to their transfer from animals to humans. She also mentioned the use of AI-assisted handheld X-rays in tuberculosis screening initiatives as an instance of AI enhancing healthcare efforts.
India's Rise as a Premier Global Clinical Trials Destination Elevating RWE Demand
India has rapidly ascended to become the third-largest destination globally for clinical trials as of 2024, growing at a compound annual growth rate of 80% from 2019. Phase II and Phase III clinical trial registrations rose by 20% year-on-year from 2019 to 2024, with 79% of registered trials addressing non-communicable diseases including malignant neoplasms and cardiovascular conditions. This clinical research expansion is substantially elevating demand for RWE solutions that complement trial data, support regulatory filings, and enable post-market surveillance across diverse patient populations within India's vast healthcare ecosystem.
Market Outlook 2026-2034:
The India real world evidence solutions market is poised for sustained expansion throughout the forecast period, supported by deepening digital health infrastructure, an increasingly evidence-conscious regulatory environment, and robust growth in pharmaceutical R&D activity. The market generated a revenue of USD 51.46 Million in 2025 and is projected to reach a revenue of USD 155.8 Million by 2034, growing at a compound annual growth rate of 12.71% from 2026-2034. Emerging opportunities in oncology evidence generation, patient-reported outcome studies, and health technology assessment for market access are expected to diversify the application landscape, while national digital health policies will continue creating the data infrastructure necessary to sustain long-term market growth.
India Real World Evidence Solutions Market Report Segmentation:
|
Segment Category |
Leading Segment |
Market Share |
|
Component |
Clinical Settings Data |
34.6% |
|
Therapeutic Area |
Oncology |
29.8% |
|
End User |
Pharmaceutical and Medical Device Companies |
47.2% |
|
Region |
South India |
31.5% |
Component Insights:
- Clinical Settings Data
- Claims Data
- Patient-Powered Data
- Pharmacy Data
- Others
Clinical settings data dominates with a market share of 34.6% of the total India real world evidence solutions market in 2025.
Clinical settings data encompasses electronic health records, discharge summaries, clinical notes, laboratory test results, and imaging data generated within hospital and outpatient care environments. As India's public and private health facilities progressively integrate ABDM-enabled hospital management information systems, the volume and quality of structured clinical data available for RWE generation has expanded considerably. The ABDM's Digital Health Incentive Scheme has incentivized health facilities to adopt digital health platforms as of late 2024, creating interconnected data pools that support longitudinal patient outcome tracking. Pharmaceutical companies and clinical research organizations increasingly rely on this institutional clinical data to construct synthetic control arms, conduct comparative effectiveness studies, and support submissions to regulatory authorities.
Claims data, while at an earlier stage of maturity relative to Western markets, is gaining traction as India's health insurance penetration grows. Patient-powered data from wearables, patient registries, and self-reported outcomes is an emerging segment increasingly deployed by specialty disease programs. Pharmacy data offers insights into prescribing patterns and treatment adherence across urban and semi-urban markets, supporting drug utilization studies and pharmacovigilance activities. Together, these component types are broadening the scope of real-world data available to stakeholders across the healthcare value chain in India.
Therapeutic Area Insights:
- Oncology
- Immunology
- Neurology
- Cardiovascular Disease
- Others
Oncology leads with a share of 29.8% of the total India real world evidence solutions market in 2025.
Oncology is the largest therapeutic area in the market, driven by the rising cancer burden across the country, along with increasing demand for better treatment pathways and outcomes monitoring. With India seeing steady growth in cancer incidence, healthcare providers and pharmaceutical companies are placing greater focus on using real-world data to understand how oncology therapies perform outside controlled clinical trials. RWE plays a major role in evaluating the effectiveness and safety of cancer drugs in diverse patient populations, especially given variations in access to care, genetic profiles, and treatment adherence.
Oncology also benefits from the rapid adoption of precision medicine, targeted therapies, and immuno-oncology drugs, all of which require continuous evidence generation after approval. In India, cancer registries, hospital networks, and digital health records are increasingly being used to support oncology-focused RWE studies. These insights help stakeholders improve clinical decision-making, support regulatory submissions, and design value-based care strategies. As investments in oncology research expand and new therapies enter the market, oncology is expected to remain the leading segment driving growth in India’s RWE solutions landscape.
End User Insights:
Access the comprehensive market breakdown Request Sample
- Pharmaceutical and Medical Device Companies
- Healthcare Payers
- Healthcare Providers
- Others
Pharmaceutical and medical device companies exhibit a clear dominance with a 47.2% share of the total India real world evidence solutions market in 2025.
Pharmaceutical and medical device companies represent the largest end-user segment, driven by their growing need to generate evidence beyond traditional clinical trials. As the healthcare landscape becomes more outcome-focused, these companies are increasingly relying on real-world data to demonstrate the safety, effectiveness, and value of their products in routine clinical practice. In India, the rapid expansion of patient populations, diverse treatment settings, and rising adoption of advanced therapies make RWE especially important for drug development and lifecycle management. Pharmaceutical firms use RWE to support regulatory approvals, identify new indications, and monitor post-market safety of therapies.
Similarly, medical device manufacturers leverage real-world insights to assess device performance, patient outcomes, and long-term benefits across different healthcare environments. The demand is also fueled by increasing competition and the need for cost-effective market access strategies. By using RWE, companies can strengthen pricing and reimbursement discussions, optimize clinical trial designs, and better understand unmet medical needs. With improvements in digital health infrastructure, electronic medical records, and data analytics capabilities, India is becoming a strong hub for RWE studies. This makes pharmaceutical and medical device companies key drivers of growth in the country’s evolving RWE solutions market.
Regional Insights:
- North India
- South India
- East India
- West India
South India leads with a share of 31.5% of the total India real world evidence solutions market in 2025.
South India's prominence in the RWE solutions market is anchored by its exceptional concentration of pharmaceutical manufacturing, biotechnology companies, and world-class tertiary care hospitals. Hyderabad, recognized as the country's pharmaceutical hub and home to the Genome Valley cluster, hosts numerous global pharmaceutical and contract research organizations that are major consumers of RWE solutions. Chennai and Bengaluru contribute significantly through their IT and health informatics capabilities, enabling the development and deployment of sophisticated analytics platforms. The region is also home to major oncology research centers, including institutions affiliated with the ICMR's Phase 1 Clinical Trial Network, further reinforcing RWE activity in South India.
North India, anchored by Delhi-NCR and major AIIMS institutions, represents a significant center for clinical research and healthcare policy, driving steady demand for RWE solutions among academic medical centers and government health programs. West India, particularly Mumbai and Pune, benefits from a dense pharmaceutical and CRO ecosystem, contributing substantial RWE activity. East India is at an earlier stage of market development but presents growing opportunity as healthcare infrastructure investment increases and tertiary care capacity expands in Kolkata and Bhubaneswar.
Market Dynamics:
Growth Drivers:
Why is the India Real World Evidence Solutions Market Growing?
Government-Led Digital Health Infrastructure Generating High-Quality Real-World Data
India's Ayushman Bharat Digital Mission is constructing the foundational digital infrastructure that makes large-scale RWE generation possible. The mission's interoperable architecture connects patients, hospitals, laboratories, pharmacies, and insurance providers through a consent-based health data exchange framework. The resulting longitudinal patient records, linked to unique health identifiers across diverse care settings, represent an unprecedented real-world data resource for pharmaceutical researchers, health technology assessment bodies, and clinical decision makers. Government incentives such as the Digital Health Incentive Scheme are accelerating facility onboarding, while the National Health Authority's collaboration with IIT Kanpur to develop a federated learning platform under ABDM signals a maturing approach to privacy-preserving analytics. This infrastructure is transforming India's previously fragmented data landscape into a structured ecosystem capable of supporting credible, regulatory-grade RWE at population scale.
Expanding Pharmaceutical R&D Investment and Clinical Trial Ecosystem
India's pharmaceutical sector is experiencing a sustained period of R&D intensification that is directly amplifying demand for RWE solutions. The country's clinical trials ecosystem has grown to make India the third-largest global destination for trials, supported by the New Drugs and Clinical Trials Rules reforms that streamlined approval processes. Global pharmaceutical companies are establishing Global Capability Centers in India, engaging local talent for drug development activities ranging from early-stage research through regulatory filings. Post-market surveillance obligations under Indian regulatory frameworks require pharmaceutical and medical device companies to collect and analyze real-world safety and effectiveness data, creating recurring demand for RWE services. In parallel, the National Biopharma Mission is strengthening clinical trial capacities, while domestic companies are investing in in-house RWE capabilities to compete in increasingly evidence-driven therapeutic markets. The National Biopharma Mission (NBM) – Innovate in India (i3) commenced in May 2017, intending to convert India into a $100 billion top global biotech sector by 2025 and secure 5% of the worldwide pharmaceutical market.
Rising Chronic Disease Burden Demanding Real-World Evidence for Treatment Optimization
India's epidemiological profile is shifting decisively toward non-communicable diseases, creating persistent and growing demand for RWE solutions across oncology, cardiovascular, neurological, and metabolic disease areas. With more than 100 million individuals living with diabetes and cancer incidence rising sharply across multiple states, healthcare stakeholders require evidence from real clinical practice settings to understand treatment patterns, outcome disparities, and adherence challenges across heterogeneous patient populations. RWE is increasingly recognized as essential for informing clinical guidelines, healthcare payer coverage policies, and public health resource allocation in the face of this growing disease burden. Personalized medicine initiatives require population-level real-world data to identify patient subgroups and tailor interventions, further expanding the scope and value of RWE solutions across the country's health system.
Market Restraints:
What Challenges the India Real World Evidence Solutions Market is Facing?
Data Quality, Completeness, and Standardization Deficiencies
Despite significant digitization progress, inconsistency in data collection standards, coding practices, and electronic health record system architectures across India's fragmented healthcare landscape introduces substantial variability in real-world dataset quality. Incomplete patient records, inconsistent diagnostic coding, and gaps in follow-up data compromise the reproducibility and credibility of RWE studies. Regulatory authorities and research sponsors therefore face significant challenges in validating real-world data for use in regulatory submissions and clinical decision-making, constraining the pace at which the market can mature toward high-value applications.
Regulatory Uncertainty and Evolving Data Privacy Frameworks
India's regulatory environment for real-world data use in drug and device approvals remains at an early stage of formalization, with the Central Drugs Standard Control Organisation yet to issue comprehensive guidance on systematic RWE integration into regulatory submissions. Simultaneously, the Digital Personal Data Protection Act of 2023 introduces new compliance requirements for health data processing that are still being interpreted and operationalized across the healthcare ecosystem. This dual uncertainty around regulatory acceptance of RWE and evolving data governance obligations increases compliance complexity for market participants and slows investment in large-scale RWE platform deployments.
Infrastructure Disparities and Limited Rural Healthcare Data Capture
Pronounced disparities in digital health infrastructure between urban and rural healthcare settings result in significant geographic gaps in real-world data coverage. Rural primary care facilities, which serve the majority of India's population, have lower rates of digital health adoption, electronic health record implementation, and connectivity, limiting the representativeness of nationally aggregated real-world datasets. RWE studies conducted primarily on urban hospital data may not accurately reflect treatment patterns and outcomes for rural populations, introducing selection bias that can reduce the generalizability and regulatory acceptability of evidence generated from such data.
Competitive Landscape:
The India real world evidence solutions market features a moderately concentrated competitive landscape, with a combination of global analytics organizations, contract research organizations, health informatics specialists, and technology-enabled data analytics firms competing for market share. Leading global players leverage expansive proprietary real-world data assets, advanced AI and machine learning analytics capabilities, and established regulatory expertise to serve multinational pharmaceutical clients conducting drug development activities in India. Indian technology conglomerates and specialized health analytics companies are deploying domestically curated datasets and cost-competitive service models to serve both global and local pharmaceutical clients. Strategic alliances between global RWE platform providers and Indian hospital networks, technology companies, and academic institutions are an increasingly common competitive strategy, enabling richer data access and locally relevant evidence generation capabilities.
India Real World Evidence Solutions Market Report Coverage:
|
Report Features |
Details |
|
Base Year of the Analysis |
2025 |
|
Historical Period |
2020-2025 |
|
Forecast Period |
2026-2034 |
|
Units |
Million USD |
|
Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
|
Components Covered |
Clinical Settings Data, Claims Data, Patient-Powered Data, Pharmacy Data, Others |
|
Therapeutic Areas Covered |
Oncology, Immunology, Neurology, Cardiovascular Disease, Others |
|
End Users Covered |
Pharmaceutical and Medical Device Companies, Healthcare Payers, Healthcare Providers, Others |
|
Regions Covered |
North India, South India, East India, West India |
|
Customization Scope |
10% Free Customization |
|
Post-Sale Analyst Support |
10-12 Weeks |
|
Delivery Format |
PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report
The India real world evidence solutions market size was valued at USD 51.46 Million in 2025.
The India real world evidence solutions market is expected to grow at a compound annual growth rate of 12.71% from 2026-2034 to reach USD 155.8 Million by 2034.
Clinical settings data dominated the component segment with a market share of 34.6% in 2025, supported by the rapid digitization of hospital and clinical encounter records across India's healthcare network under the Ayushman Bharat Digital Mission.
Key factors driving the India real world evidence solutions market include the government's Ayushman Bharat Digital Mission creating large-scale longitudinal health data repositories, expanding pharmaceutical R&D and clinical trials activity, rising chronic and non-communicable disease burden, and growing regulatory awareness of RWE's role in drug approvals and post-market surveillance.
Major challenges include data quality and standardization deficiencies across fragmented healthcare settings, regulatory uncertainty regarding CDSCO's formal guidance on RWE acceptance, evolving data privacy compliance requirements under the Digital Personal Data Protection Act of 2023, and infrastructure gaps between urban and rural care settings that limit the representativeness of nationally aggregated real-world datasets.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












