
Influenza Diagnostics Market Report by Product (Test Kit and Reagents, Instruments, and Others), Test Type (Molecular Diagnostic Tests, Traditional Diagnostic Tests), Type of Flu (Type A Flu, Type B Flu, Type C Flu), End User (Hospitals, Diagnostic Laboratories, and Others), and Region 2026-2034
Market Overview:
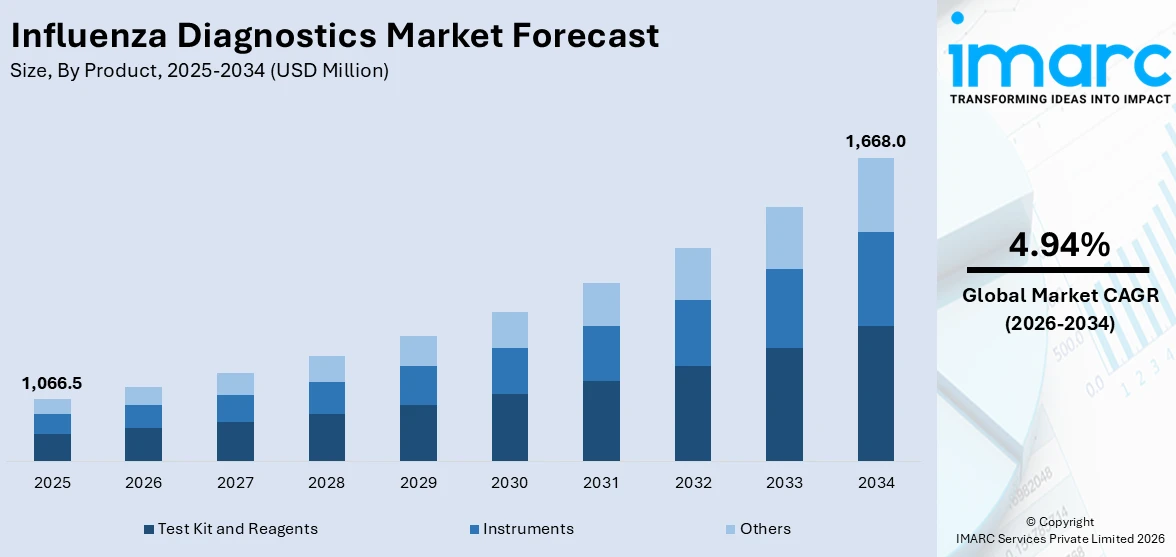
The global influenza diagnostics market size reached USD 1,066.5 Million in 2025. Looking forward, IMARC Group expects the market to reach USD 1,668.0 Million by 2034, exhibiting a growth rate (CAGR) of 4.94% during 2026-2034. The increasing vaccination efforts, the convenience of point-of-care tests, the rising research into flu viruses, and the growing awareness through public health programs are some of the factors propelling the market.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 1,066.5 Million |
|
Market Forecast in 2034
|
USD 1,668.0 Million |
| Market Growth Rate 2026-2034 | 4.94% |
Influenza, commonly known as the flu, is a viral infection primarily targeting the respiratory system, encompassing the nose, throat, and lungs. Influenza diagnostics encompasses an array of immunoassay tests designed to identify the presence of influenza A and B viral nucleoprotein antigens within respiratory samples. Among the diagnostic options available are rapid antigen testing, reverse transcription polymerase chain reaction (RT-PCR), immunofluorescence assays, serology, and rapid molecular assays. The medical fraternity relies on these tests to detect and categorize influenza strains promptly, aiding in effective treatment decisions and public health management. The continual evolution of diagnostic techniques and the ongoing research into influenza viruses enhance the efficiency and reliability of these tests. As healthcare providers strive to combat seasonal flu outbreaks and potential pandemics, accurate diagnostics remain a cornerstone, enabling swift intervention and informed medical strategies.
To get more information on this market Request Sample
The global market is majorly driven by the increasing occurrence of flu outbreaks. In line with this, the rising global concerns about potential pandemics drive investments in diagnostic capabilities, significantly contributing to the market. Furthermore, rapid technological innovations improve the accuracy and speed of diagnostic tests, positively influencing the market. Apart from this, the growing public awareness campaigns encourage more individuals to seek timely testing, catalyzing the market. Moreover, the escalating healthcare spending supports the adoption of advanced diagnostic methods, propelling the market. Besides, elderly individuals are more susceptible to severe flu, boosting demand for diagnostics. Additionally, the increasing urban populations contribute to higher infection rates, requiring robust diagnostics. Frequent travel accelerates the spread of the flu, necessitating efficient diagnostics. This, in turn, offers numerous opportunities for the market.
Influenza Diagnostics Market Trends/Drivers:
Easy access to healthcare
The easy access to healthcare is stimulating market growth. Improved accessibility to medical facilities encourages individuals to seek timely diagnostic testing for flu infections. When healthcare services are easily reachable, people are more inclined to undergo testing at the onset of symptoms, leading to early detection and appropriate intervention. This timely approach not only aids in preventing the spread of the virus but also facilitates better patient outcomes. Furthermore, convenient access to healthcare facilities supports the adoption of various diagnostic methods, including point-of-care testing, which provides rapid results without requiring extensive laboratory infrastructure. This accessible healthcare environment creates a favorable ecosystem for diagnostic manufacturers, healthcare providers, and patients, ultimately contributing to market growth.
Increasing prevalence of several chronic diseases
The increasing prevalence of several chronic diseases is catalyzing the market. Chronic diseases, such as diabetes, cardiovascular disorders, and respiratory conditions, weaken the immune system and make individuals more susceptible to severe flu infections. As the number of individuals affected by these chronic conditions rises, the demand for accurate and early influenza diagnostics intensifies. Patients with chronic diseases are at a higher risk of complications from the flu, necessitating prompt detection and tailored medical interventions. Healthcare providers prioritize testing these vulnerable populations to mitigate potential complications and provide timely treatment. Moreover, the interconnectedness of chronic diseases and flu underscores the need for comprehensive diagnostic approaches. Accurate flu diagnosis in individuals with pre-existing conditions enables healthcare professionals to tailor treatment plans, manage complications effectively, and improve patient outcomes. As chronic diseases continue to increase globally, the demand for influenza diagnostics becomes integral in safeguarding the health of individuals with underlying conditions. This ongoing demand significantly contributes to the market.
Rapid integration of artificial intelligence
The rapid integration of artificial intelligence (AI) is favorably impacting the market. AI technologies are revolutionizing diagnostic capabilities by enhancing flu detection accuracy, speed, and efficiency. AI-powered algorithms can analyze vast amounts of data from various sources, including patient histories, symptoms, and test results, to provide more precise and tailored diagnostic insights. This accelerates the identification of flu cases, enabling timely medical interventions and public health responses. Machine learning and AI algorithms also improve predictive modeling for flu outbreaks. By analyzing historical data, patterns, and environmental factors, these technologies aid in forecasting potential flu epidemics, assisting healthcare organizations in preparing and allocating resources effectively. Additionally, AI-driven image analysis assists in evaluating medical images, such as chest X-rays, to identify flu-related complications and monitor disease progression accurately. The rapid pace of AI integration is transforming the diagnostic landscape, offering healthcare professionals advanced tools for more informed decision-making and precise patient care. This trend contributes significantly to the market.
Influenza Diagnostics Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global influenza diagnostics market report, along with forecasts at the global, regional and country levels from 2026-2034. Our report has categorized the market based on product, test type, type of flu and end user.
Breakup by Product:
- Test Kit and Reagents
- Instruments
- Others
Test Kit and Reagents dominates the market
The report has provided a detailed breakup and analysis of the market based on the product. This includes test kit and reagents, instruments, and others. According to the report, test kit and reagents represented the largest segment.
Test kit and reagents form the foundation of diagnostic procedures, providing the necessary chemical components for identifying viral antigens and antibodies in patient samples. These kits enable healthcare professionals to swiftly and accurately diagnose influenza infections, facilitating timely medical interventions and public health responses. The continuous innovation in test kit technology, including improved sensitivity and specificity, contributes to heightened accuracy in flu detection.
On the other hand, instruments encompass the equipment for processing and analyzing samples, automating diagnostic workflows, and delivering reliable results. These instruments, ranging from PCR machines to automated immunoassay platforms, streamline the testing process, reduce human errors, and enhance the overall efficiency of diagnostics.
Breakup by Test Type:
- Molecular Diagnostic Tests
- Polymerase Chain Reaction
- Isothermal Nucleic Acid Amplification Tests
- Other Molecular Tests
- Traditional Diagnostic Tests
- Rapid Influenza Diagnostic Tests
- Viral Culture Tests
- Direct Fluorescent Antibody Test
- Serological Tests
Traditional Diagnostic Test dominates the market
The report has provided a detailed breakup and analysis of the market based on the test type. This includes molecular diagnostic tests (polymerase chain reaction, isothermal nucleic acid amplification tests, other molecular tests) and traditional diagnostic tests (rapid influenza diagnostic tests, viral culture tests, direct fluorescent antibody test, serological tests). According to the report, traditional diagnostic tests represented the largest segment.
Traditional diagnostic tests encompass methods like rapid antigen testing and immunofluorescence assays. While they may not offer the same level of sensitivity as molecular tests, they provide rapid results, making them invaluable in scenarios where swift diagnosis is critical for patient management and public health responses.
Molecular diagnostic tests on the other hand, utilize advanced molecular techniques such as polymerase chain reaction (PCR) and nucleic acid amplification to detect the influenza virus's genetic material directly. These tests offer high sensitivity and specificity, enabling early and accurate identification of viral infections. The increasing adoption of molecular diagnostic tests is driven by their ability to detect multiple flu strains and their potential to differentiate between seasonal and novel influenza viruses.
Breakup by Type of Flu:
- Type A Flu
- Type B Flu
- Type C Flu
Type A Flu dominates the market
The report has provided a detailed breakup and analysis of the market based on the type of flu. This includes type A flu, type B flu, and type C flu. According to the report, type A flu represented the largest segment.
"Type A Flu" includes various influenza viruses, some responsible for seasonal flu outbreaks and potential pandemics. Accurate differentiation of specific Type A strains is essential for targeted treatment and surveillance efforts.
Furthermore, "Type B Flu" encompasses a distinct group of viruses contributing to seasonal flu epidemics. Detecting and categorizing Type B strains aid in tailoring medical interventions and vaccine development to mitigate flu-related complications.
Moreover, "Type C Flu," although typically milder, still requires accurate diagnosis for effective patient management. While it is less likely to cause large outbreaks, its inclusion highlights the comprehensive approach to influenza diagnostics.
Breakup by End User:
Access the comprehensive market breakdown Request Sample
- Hospitals
- Diagnostic Laboratories
- Others
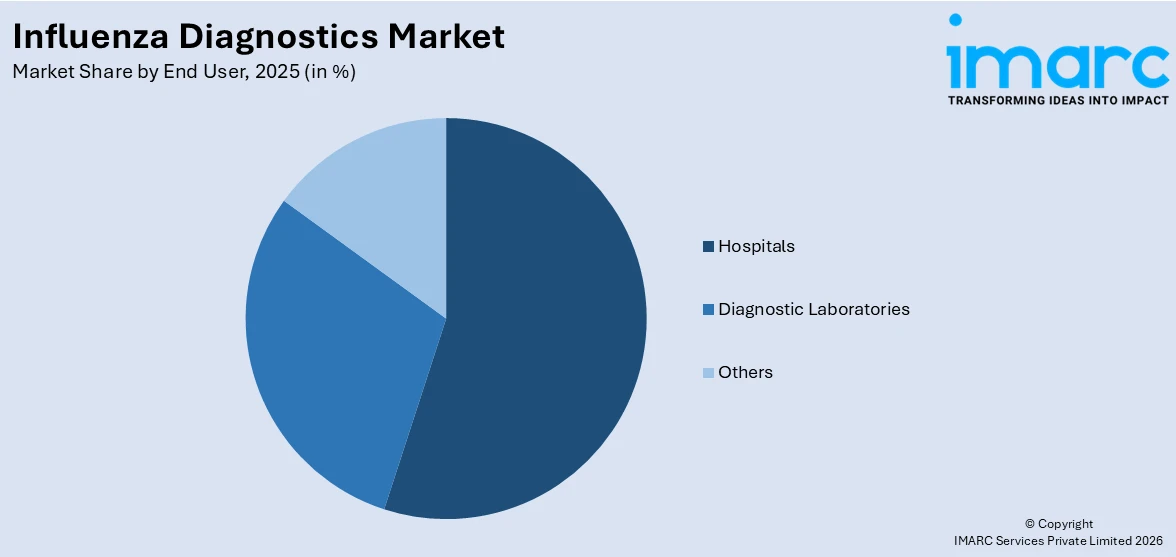
Hospitals dominates the market
The report has provided a detailed breakup and analysis of the market based on the end user. This includes hospitals, diagnostic laboratories, and others. According to the report, hospitals represented the largest segment.
Hospitals encompass a significant portion of the market demand, offering immediate access to diagnostic services for patients with flu-like symptoms. Timely diagnosis within hospitals enables rapid treatment decisions and infection control measures, which are crucial for managing patient health and preventing virus spread within healthcare facilities.
Furthermore, diagnostic Laboratories are specialized hubs for accurate and thorough flu testing. These laboratories often employ advanced equipment and experienced personnel to conduct comprehensive diagnostic procedures. By focusing on high-quality diagnostics, they support healthcare providers in delivering accurate results, essential for effective patient management and public health interventions.
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America exhibits a clear dominance, accounting for the largest market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America accounted for the largest market share.
North America's emphasis on research and innovation fosters the development of cutting-edge diagnostic technologies and methodologies. This region leads in adopting advanced testing solutions that enhance the accuracy and efficiency of flu detection. Its high prevalence of chronic diseases and respiratory conditions underscores the need for accurate influenza diagnostics. With a focus on timely detection, healthcare providers in North America rely on a wide range of diagnostic tests to ensure prompt treatment and prevent escalating flu-related complications.
Government initiatives, awareness campaigns, and strong healthcare policies drive the demand for influenza diagnostics. The region's robust public health infrastructure emphasizes the importance of accurate diagnostics in managing seasonal outbreaks and potential pandemics. The prevalence of international travel and globalization also heightens the demand for accurate flu testing in North America, as early detection becomes crucial in preventing the importation and spread of viral strains. The region's commitment to accurate and timely diagnostics positions it as a key driver in shaping the industry's expansion.
Competitive Landscape:
Top companies are bolstering the market through their innovative approaches and expertise. These companies leverage their resources and research capabilities to drive market expansion. They invest significantly in research and development, leading to the creation of advanced diagnostic technologies. These innovations, including AI-powered platforms and rapid testing solutions, enhance the accuracy and efficiency of flu detection. Top companies collaborate with healthcare providers and institutions to ensure widespread product access. These partnerships facilitate the integration of cutting-edge diagnostic solutions into existing healthcare systems, expanding market reach. Moreover, these companies often conduct awareness campaigns to educate healthcare professionals and the public about the importance of timely flu testing. These efforts drive increased demand for accurate diagnostics. Furthermore, their global presence and distribution networks enable them to provide essential diagnostic tools to regions with varying levels of healthcare infrastructure, contributing to overall market growth.
The report has provided a comprehensive analysis of the competitive landscape in the influenza diagnostics market. Detailed profiles of all major companies have also been provided. Some of the key players in the market include:
- Abbott Laboratories
- Becton Dickinson and Company
- Cepheid (Danaher Corporation)
- Coris BioConcept
- DiaSorin S.p.A.
- F. Hoffmann-La Roche Ltd
- Hologic, Inc.
- Meridian Bioscience Inc.
- Qiagen N.V
- QuidelOrtho Corporation
- SEKISUI Diagnostics
- Siemens Healthcare Diagnostics GmbH
- Thermo Fisher Scientific Inc.
Recent Developments:
- In August 2020, Abbott launched the BinaxNOW COVID-19 Ag Card, a rapid antigen test for detecting SARS-CoV-2, the virus causing COVID-19.
- In August 2020, BD received Emergency Use Authorization from the FDA for a rapid point-of-care antigen test to detect SARS-CoV-2, providing results in 15 minutes.
- In April 2020, Cepheid, a subsidiary of Danaher, received FDA Emergency Use Authorization for its Xpert Xpress SARS-CoV-2 test, designed to deliver rapid results for detecting COVID-19.
Influenza Diagnostics Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Million USD |
| Scope of the Report | Exploration of Historical and Forecast Trends, Industry Catalysts and Challenges, Segment-Wise Historical and Predictive Market Assessment:
|
| Products Covered | Test Kit and Reagents, Instruments, Others |
| Test Types Covered |
|
| Types of Flu Covered | Type A Flu, Type B Flu, Type C Flu |
| End Users Covered | Hospitals, Diagnostic Laboratories, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Abbott Laboratories, Becton Dickinson and Company, Cepheid (Danaher Corporation), Coris BioConcept, DiaSorin S.p.A., F. Hoffmann-La Roche Ltd, Hologic, Inc., Meridian Bioscience Inc., Qiagen N.V, QuidelOrtho Corporation, SEKISUI Diagnostics, Siemens Healthcare Diagnostics GmbH, Thermo Fisher Scientific Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the influenza diagnostics market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global influenza diagnostics market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the influenza diagnostics industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Frequently Asked Questions About the Influenza Diagnostics Market Report
The global influenza diagnostics market was valued at USD 1,066.5 Million in 2025.
We expect the global influenza diagnostics market to exhibit a CAGR of 4.94% during 2026-2034.
The rising prevalence of respiratory tract infections, along with the growing integration of Artificial Intelligence (AI) with influenza diagnostics to minimize the timelines for sample processing and facilitate error-free results, is primarily driving the global influenza diagnostics market.
The sudden outbreak of the COVID-19 pandemic has led to the increasing adoption of numerous influenza diagnostics tests, such as rapid antigen testing and Reverse Transcription Polymerase Chain Reaction (RT-PCR), to detect the coronavirus infection among individuals.
Based on the product, the global influenza diagnostics market has been segmented into test kit and reagents, instruments, and others. Currently, test kit and reagents hold the majority of the total market share.
Based on the test type, the global influenza diagnostics can be divided into molecular diagnostic tests and traditional diagnostic tests, where traditional diagnostic tests currently exhibit a clear dominance in the market.
Based on the type of flu, the global influenza diagnostics market has been categorized into type A flu, type B flu, and type C flu. Currently, type A flu accounts for the majority of the global market share.
Based on the end user, the global influenza diagnostics market can be segregated into hospitals, diagnostic laboratories, and others. Among these, hospitals hold the largest market share.
On a regional level, the market has been classified into North America, Asia-Pacific, Europe, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global influenza diagnostics market include Abbott Laboratories, Becton Dickinson and Company, Cepheid (Danaher Corporation), Coris BioConcept, DiaSorin S.p.A., F. Hoffmann-La Roche Ltd, Hologic, Inc., Meridian Bioscience Inc., Qiagen N.V, QuidelOrtho Corporation, SEKISUI Diagnostics, Siemens Healthcare Diagnostics GmbH, and Thermo Fisher Scientific Inc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












