
Interventional Cardiology Devices Market Report by Product (Stents, Interventional Catheters, PTCA Guidewires, Balloon Inflation Device, PTCA Balloons), End User (Hospitals, Ambulatory Surgical Centers, and Others), and Region 2026-2034
Global Interventional Cardiology Devices Market:
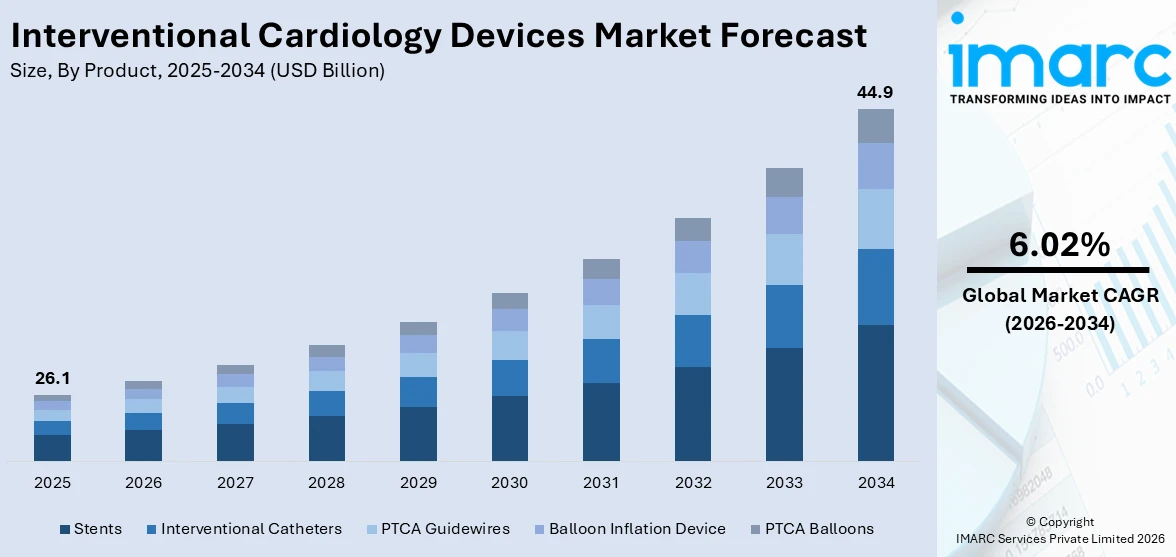
The global interventional cardiology devices market size reached USD 26.1 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 44.9 Billion by 2034, exhibiting a growth rate (CAGR) of 6.02% during 2026-2034. The rising prevalence of cardiovascular diseases, aging population, ongoing technological advancements, and increasing shift towards minimally invasive procedures are primarily driving the market's growth.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 26.1 Billion |
|
Market Forecast in 2034
|
USD 44.9 Billion |
| Market Growth Rate 2026-2034 | 6.02% |
Interventional Cardiology Devices Market Analysis:
- Major Market Drivers: The increasing prevalence of diabetes, hypertension, and cardiovascular disorders, such as coronary artery disease (CAD, are some of the key factors driving the growth of the market. Moreover, the rising geriatric population, which is more susceptible to such ailments, is providing a thrust to the interventional cardiology devices market growth.
- Key Market Trends: The increasing utilization of advanced imaging solutions, such as intravenous ultrasound (IVUS), fractional flow reserve (FFR), and optical coherence tomography (OCT) for early diagnosis and timely treatment of cardiovascular ailments, is contributing to the interventional cardiology devices market share. Other factors, including rising healthcare expenditure capacities of the masses, along with extensive improvements in the medical infrastructure, especially in developing economies, are anticipated to drive the market towards growth.
- Competitive Landscape: Some of the prominent interventional cardiology devices market companies include Abbott Laboratories, Alvimedica, B. Braun Melsungen AG, Biosensors International Group Ltd. (Bluesail Medical Co. Ltd.), BIOTRONIK Inc., Boston Scientific Corporation, Cook Group Incorporated, ENDOCOR GmbH, Koninklijke Philips N.V., Medinol Ltd., Medtronic plc, and Terumo Corporation, among many others.
- Geographical Trends: According to the interventional cardiology devices market dynamics, North America dominates the overall market. Cardiovascular diseases, such as coronary artery disease (CAD), myocardial infarction, and stroke, are some of the leading causes of death in North America. The increasing incidence of CVDs has driven the demand for interventional cardiology procedures, such as angioplasty, stenting, and catheter-based treatments.
- Challenges and Opportunities: The high cost associated with the devices and the shortage of trained professionals are hampering the interventional cardiology devices market share. However, with the increasing incidence of cardiovascular diseases worldwide, particularly driven by aging populations, rising obesity rates, diabetes, and hypertension, the demand for interventional cardiology procedures continues to grow.
To get more information on this market Request Sample
Interventional Cardiology Devices Market Trends:
Rising Prevalence of Cardiovascular Disease
Cardiovascular diseases, such as coronary artery disease, myocardial infarction, and stroke, are the leading causes of death globally. For instance, according to a World Heart Federation (WHF) report, cardiovascular disease (CVD) deaths increased globally from nearly 12.1 million in 1990 to 20.5 million in 2021. With aging populations, rising obesity rates, sedentary lifestyles, and increasing rates of hypertension and diabetes, the number of people affected by CVD continues to rise, driving demand for interventional procedures. These factors are expected to propel the interventional cardiology devices market growth in the coming years.
Aging Population
The growing aging population is one of the primary demographic drivers for the interventional cardiology market. For instance, according to the World Health Organization (WHO), by 2030, one in every six persons in the world will be 60 or older. At this time, the proportion of the population aged 60 and up will rise from 1 billion in 2020 to 1.4 billion. By 2050, the global population of persons aged 60 and up will double (2.1 billion). Older individuals are more prone to developing cardiovascular diseases due to factors like reduced physical activity and comorbid conditions such as diabetes and hypertension. These factors further positively influence the interventional cardiology devices market size.
Product Innovations
Drug-eluting stents (DES) have been a major breakthrough, reducing the rate of restenosis compared to bare-metal stents. Newer DES devices, which are polymer-coated or have enhanced drug formulations, are driving growth. For instance, in May 2024, Abbott, a global healthcare business, launched XIENCE Sierra, an everolimus-eluting coronary stent system, in India. The stent is coated with the medicine everolimus and a polymer (PBMA and PVDF-HFP) coating that helps distribute the drug at a controlled rate, thereby boosting the interventional cardiology devices market share.
Global Interventional Cardiology Devices Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global interventional cardiology devices market report, along with forecasts at the global, regional, and country levels from 2026-2034. Our report has categorized the market based on product and end user.
Breakup by Product:
- Stents
- Drug Eluting Stents
- Bare Metal Stents
- Bio-Absorbable Stents
- Interventional Catheters
- IVUS Catheters
- Guiding Catheters
- Angiography Catheters
- PTCA Guidewires
- Balloon Inflation Device
- PTCA Balloons
- Cutting Balloons
- Scoring Balloons
- Drug Eluting Balloons
- Normal Balloons
Stents hold the majority of the total market share
The report has provided a detailed breakup and analysis of the interventional cardiology devices market based on the product. This includes stents (drug eluting stents, bare metal stents, and bio-absorbable stents), interventional catheters (IVUS catheters, guiding catheters, and angiography catheters), PTCA guidewires, balloon inflation device, and PTCA balloons (cutting balloons, scoring balloons, drug eluting balloons, and normal balloons). According to the report, stents hold the majority of the total market share.
According to the interventional cardiology devices market outlook, the global increase in cardiovascular diseases, particularly coronary artery disease (CAD) and peripheral artery disease (PAD), is a major driver of stent demand. As risk factors such as hypertension, diabetes, obesity, smoking, and sedentary lifestyles become more prevalent, so does the need for stenting procedures to restore blood flow and prevent heart attacks or strokes. Moreover, aging populations, particularly in developed countries, are a significant driver for stent demand. As people age, they are more likely to suffer from conditions like atherosclerosis, where stents are commonly used to open narrowed or blocked arteries. With the growing number of elderly individuals globally, the demand for stents is expected to rise substantially.
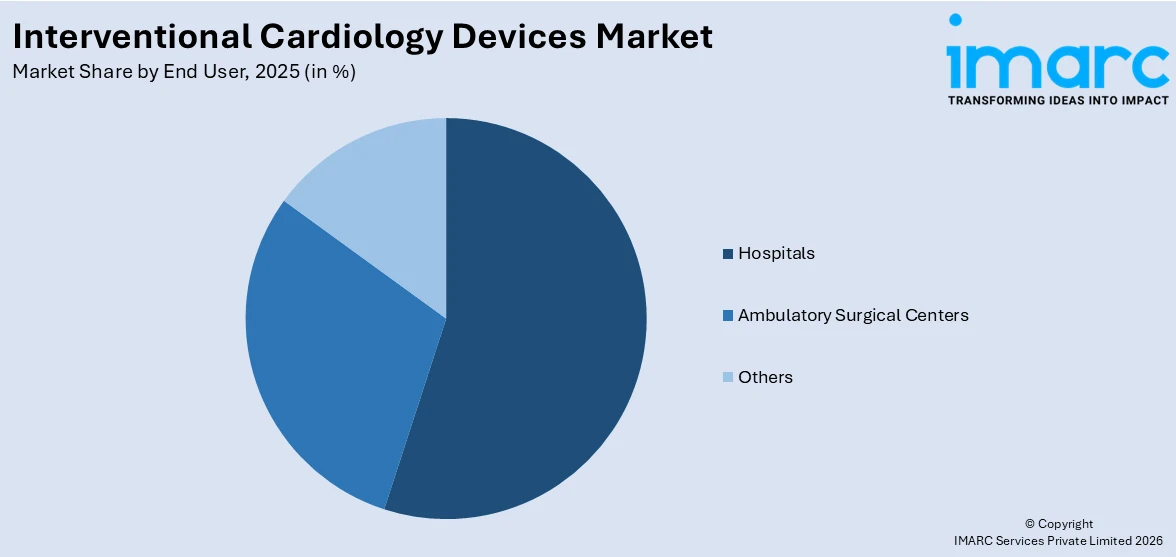
Breakup by End User:
Access the comprehensive market breakdown Request Sample
- Hospitals
- Ambulatory Surgical Centers
- Others
Hospitals exhibit a clear dominance in the market
The report has provided a detailed breakup and analysis of the interventional cardiology devices market based on the end user. This includes hospitals, ambulatory surgical centers, and others. According to the report, hospitals exhibit a clear dominance in the market.
According to the interventional cardiology devices market overview, hospitals are increasingly investing in hybrid operating rooms and advanced catheterization laboratories (cath labs) equipped with the latest interventional cardiology devices. Moreover, the global rise in the elderly population, who are more susceptible to cardiovascular diseases, is leading to increased hospital admissions for heart-related conditions. As older patients require more frequent interventional cardiology procedures, hospitals are seeing a growing demand for devices that support coronary interventions, such as stents, valves, and balloons.
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America currently dominates the global market
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America currently dominates the global market.
According to the interventional cardiology devices market statistics, cardiovascular diseases remain a leading cause of morbidity and mortality in North America, particularly in the United States and Canada. For instance, according to the American Heart Association, cardiovascular disease (CVD) is the most common cause of death in the United States, accounting for nearly 928,700 deaths in 2020. Factors such as sedentary lifestyles, high rates of obesity, hypertension, diabetes, and smoking contribute to the increasing incidence of coronary artery disease (CAD), heart attacks, and strokes. This rising burden of CVDs drives the demand for interventional cardiology devices, such as stents, angioplasty balloons, and atherectomy devices.
Competitive Landscape:
The market research report has provided a comprehensive analysis of the competitive landscape. Detailed profiles of all major market companies have also been provided. Some of the key players in the market include:
- Abbott Laboratories
- Alvimedica
- B. Braun Melsungen AG
- Biosensors International Group Ltd. (Bluesail Medical Co. Ltd.)
- BIOTRONIK Inc.
- Boston Scientific Corporation
- Cook Group Incorporated
- ENDOCOR GmbH
- Koninklijke Philips N.V.
- Medinol Ltd.
- Medtronic plc
- Terumo Corporation
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
Interventional Cardiology Devices Market Recent Developments:
- September 2024: Argon Medical Devices, a provider of medical device solutions for interventional radiology, vascular surgery, interventional cardiology, and oncology treatments, launched the CLEANER Vac™ Thrombectomy System for the removal of blood clots from the peripheral venous vasculature.
- June 2024: Columbia University Irving Medical Center's Division of Cardiology established the Mitral and Tricuspid Center. This advanced clinical research center aims to improve the diagnosis and treatment of tricuspid and mitral valve conditions by bringing together experts from various cardiovascular disciplines.
- May 2024: GE HealthCare and Medis Medical Imaging collaborated to improve non-invasive coronary assessments.
Interventional Cardiology Devices Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Products Covered |
|
| End Users Covered | Hospitals, Ambulatory Surgical Centers, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Abbott Laboratories, Alvimedica, B. Braun Melsungen AG, Biosensors International Group Ltd. (Bluesail Medical Co. Ltd.), BIOTRONIK Inc., Boston Scientific Corporation, Cook Group Incorporated, ENDOCOR GmbH, Koninklijke Philips N.V., Medinol Ltd., Medtronic plc, Terumo Corporation, etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC's report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the interventional cardiology devices market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global interventional cardiology devices market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the interventional cardiology devices industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Frequently Asked Questions About the Interventional Cardiology Devices Market Report
The global interventional cardiology devices market was valued at USD 26.1 Billion in 2025.
We expect the global interventional cardiology devices market to exhibit a CAGR of 6.02% during 2026-2034.
The high prevalence of cardiovascular disorders, coupled with the rising utilization of interventional cardiology for performing cardiovascular procedures, such as angioplasty and stenting, is primarily driving the global interventional cardiology devices market.
The sudden outbreak of the COVID-19 pandemic had led to the postponement of elective cardiovascular surgeries to reduce the risk of the coronavirus infections upon hospital visits and interaction with medical equipment, thereby limiting the demand for interventional cardiology devices.
Based on the product, the global interventional cardiology devices market has been segmented into stents, interventional catheters, PTCA guidewires, balloon inflation device, and PTCA balloons. Currently, stents hold the majority of the total market share.
Based on the end user, the global interventional cardiology devices market can be divided into hospitals, ambulatory surgical centers, and others. Among these, hospitals exhibit a clear dominance in the market.
On a regional level, the market has been classified into North America, Asia-Pacific, Europe, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global interventional cardiology devices market include Abbott Laboratories, Alvimedica, B. Braun Melsungen AG, Biosensors International Group Ltd. (Bluesail Medical Co. Ltd.), BIOTRONIK Inc., Boston Scientific Corporation, Cook Group Incorporated, ENDOCOR GmbH, Koninklijke Philips N.V., Medinol Ltd., Medtronic plc, and Terumo Corporation.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












