
Molecular Quality Controls Market Report by Product Type (Independent Control, Instrument Specific-Control), Analyte Type (Single-Analyte Controls, Multi-Analyte Controls), Application (Oncology, Genetic Testing, Infectious Diseases, and Others), End User (Hospitals, Clinical Laboratories, IVD Manufacturers and CROs, and Others), and Region 2026-2034
Market Overview:
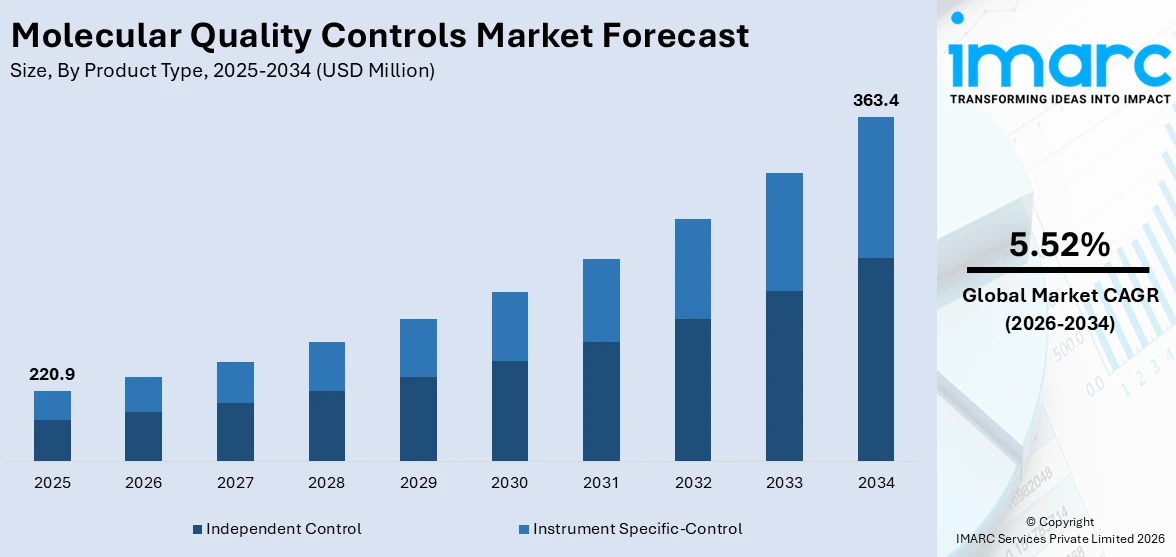
The global molecular quality controls market size reached USD 220.9 Million in 2025. Looking forward, IMARC Group expects the market to reach USD 363.4 Million by 2034, exhibiting a growth rate (CAGR) of 5.52% during 2026-2034. The increasing adoption of molecular diagnostics in healthcare, rising prevalence of infectious diseases and chronic conditions, continuous advancements in technology and automation, surging demand for accurate and reliable quality control solutions, and the growing needs of laboratories and healthcare providers are some of the key factors supporting the market growth.
|
Report Attribute
|
Key Statistics |
|---|---|
|
Base Year
|
2025 |
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
| Market Size in 2025 | USD 220.9 Million |
| Market Forecast in 2034 | USD 363.4 Million |
| Market Growth Rate (2026-2034) | 5.52% |
Molecular quality controls are standardized substances used to evaluate the performance and accuracy of molecular diagnostic tests, such as those for detecting specific genes, mutations, or pathogens. These controls help ensure that the test procedures are working correctly by providing known reference materials to compare against the patient's sample. Consequently, they are widely used in the fields of research and diagnostics, contributing to the accuracy and reliability of experimental outcomes. The production of molecular quality controls involves stringent processes that adhere to industry standards and guidelines. Expert teams meticulously select and validate appropriate analytes and concentrations, ensuring stability and reproducibility. These controls are then manufactured in large batches to minimize batch-to-batch variability, using state-of-the-art technology and rigorous quality assurance procedures. One of the primary advantages of molecular quality controls lies in their ability to assess the entire testing process, from sample preparation to data analysis. They enable laboratories to identify and correct any inconsistencies, inaccuracies, or technical errors that may occur during testing, thereby improving the reliability and comparability of results. There are various types of molecular quality controls tailored to different applications, including positive controls, negative controls, and full-process controls.
To get more information on this market Request Sample
In the global molecular quality controls market, several drivers have emerged that are shaping its growth and adoption, including the increasing adoption of molecular diagnostics in healthcare settings. Moreover, the rising prevalence of infectious diseases and chronic conditions have fueled the need for effective quality control solutions to maintain the accuracy of diagnostic tests, which, in turn, is supporting the market growth. Apart from this, advancements in technology and automation facilitate the development of innovative molecular quality control products, improving overall testing efficiency and creating a positive outlook for the market. Additionally, stringent regulatory requirements imposed by healthcare authorities worldwide propel the market as laboratories seek compliant quality control measures. Furthermore, the growing focus on personalized medicine and precision healthcare and the expanding research and development (R&D) activities in genomics and proteomics are providing an impetus to the market growth.
Molecular Quality Controls Market Trends/Drivers:
Increasing adoption of molecular diagnostics
The rapid adoption of molecular diagnostics in healthcare settings is a primary driver for the demand for molecular quality controls. Molecular diagnostics involves the detection and analysis of genetic material (DNA or RNA) to identify diseases, pathogens, or genetic variations. These tests play a crucial role in diagnosing infectious diseases, genetic disorders, and various types of cancers. As the importance of molecular diagnostics grows, healthcare providers and laboratories place a significant emphasis on ensuring the accuracy and reliability of test results. High-quality molecular quality controls are essential to verify the performance of diagnostic assays, monitor the consistency of results, and detect any potential errors or deviations in the testing process. Consequently, the increasing adoption of molecular diagnostics fuels the demand for effective quality control solutions to maintain the precision and credibility of these tests.
Rising prevalence of infectious diseases and chronic conditions
The global prevalence of infectious diseases and chronic conditions, such as diabetes, cardiovascular diseases, and cancer, continues to rise. This escalating health burden highlights the need for accurate and timely diagnostic solutions. Molecular diagnostics have proven to be valuable tools in the early detection and monitoring of infectious diseases and chronic conditions. To ensure reliable test outcomes, it becomes imperative to use high-quality molecular quality controls to validate the accuracy and consistency of diagnostic tests. In the face of such health challenges, healthcare providers and clinical laboratories seek robust quality control measures to minimize the risk of false-negative or false-positive results and to provide patients with the most accurate diagnoses, enabling appropriate treatment decisions.
Advancements in technology and automation
Technological advancements and the integration of automation have revolutionized the field of molecular diagnostics. Innovative technologies have led to the development of more sophisticated and sensitive diagnostic assays, enhancing the accuracy and efficiency of testing procedures. Automation streamlines laboratory workflows, reduces human errors, and increases the throughput of tests. However, the implementation of these advanced technologies requires rigorous quality assurance to ensure their proper functioning and reliability. Molecular quality controls are vital in assessing the performance of automated diagnostic systems and validating their accuracy. As the industry continues to embrace cutting-edge technologies and automation, the demand for effective molecular quality controls rises to ensure that these advancements deliver precise and dependable results, thereby augmenting the market growth.
Molecular Quality Controls Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global molecular quality controls market report, along with forecasts at the global, regional, and country levels for 2026-2034. Our report has categorized the market based on product type, analyte type, application, and end user.
Breakup by Product Type:
- Independent Control
- Instrument Specific-Control
Independent control dominates the market
The report has provided a detailed breakup and analysis of the market based on the product type. This includes independent control and instrument specific-control. According to the report, independent control represented the largest segment.
Independent and instrument-specific controls are vital components of the global molecular quality controls market. Independent controls, compatible with various molecular diagnostic instruments and assays, provide versatility. They offer a wide range of analytes and cross-platform utility, ensuring flexibility and cost-effectiveness. Their standalone nature allows for unbiased and accurate assessment of the entire testing process, making them a preferred choice in laboratories. Instrument-specific controls, on the other hand, are tailored to match specific diagnostic instruments and testing methodologies. They are designed to mimic the matrix of patient samples, ensuring precise evaluation of a particular instrument or assay's performance. Their use minimizes potential discrepancies between control results and patient samples, enhancing the accuracy and reliability of test outcomes. This specificity makes them a significant driver in the molecular quality controls market.
Breakup by Analyte Type:
- Single-Analyte Controls
- Multi-Analyte Controls
Single-analyte controls dominate the market
The report has provided a detailed breakup and analysis of the market based on the analyte type. This includes single-analyte and multi-analyte controls. According to the report, single-analyte controls represented the largest segment.
Single-analyte and multi-analyte controls are two essential elements in the molecular diagnostics market. Single-analyte controls, with their specific and focused nature, are designed to validate the performance of individual molecular tests. They cater to the rising demand for targeted diagnostics, bolstered by the prominence of molecular diagnostics in precision medicine and disease management. These controls offer tailored solutions, ensuring the precision and credibility of single-target assays. On the other hand, multi-analyte controls are marked by their versatility and efficiency. As laboratories increasingly adopt comprehensive testing panels for various diseases, these controls become indispensable. They allow simultaneous validation of multiple targets within a single assay, streamlining the testing process while saving time and resources. As the demand for cost-effective and time-efficient diagnostics rises, multi-analyte controls continue to solidify their essential role in the molecular diagnostics landscape.
Breakup by Application:
Access the comprehensive market breakdown Request Sample
- Oncology
- Genetic Testing
- Infectious Diseases
- Others
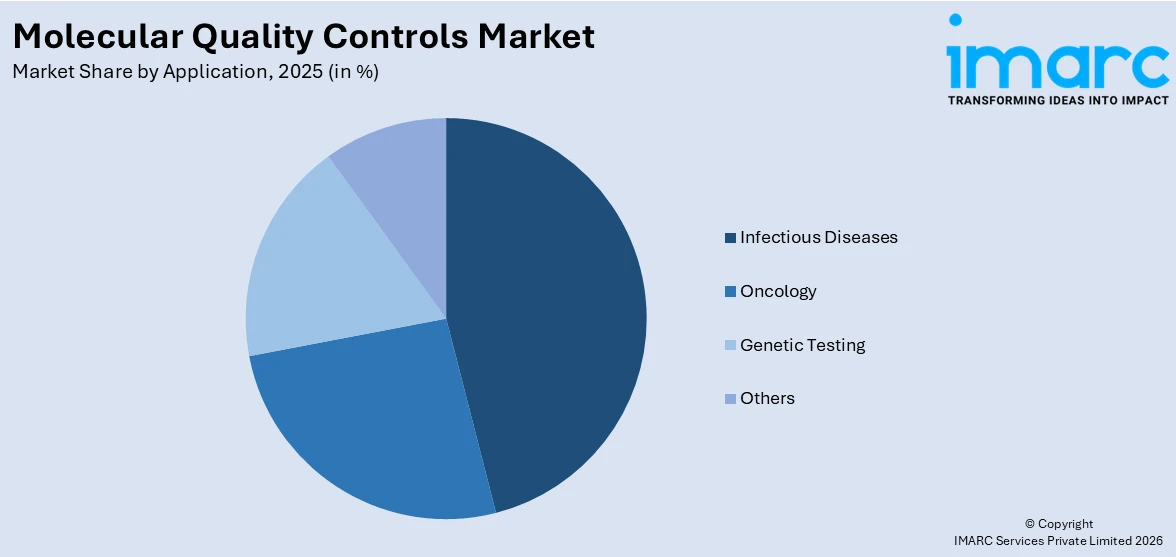
Infectious diseases dominate the market
The report has provided a detailed breakup and analysis of the market based on the application. This includes oncology, genetic testing, infectious diseases, and others. According to the report, infectious diseases represented the largest segment.
The oncology segment is driven by the escalating global cancer burden, with molecular diagnostics crucial for identifying specific genetic mutations and biomarkers associated with different cancers, thus driving demand for accurate quality controls. Genetic testing, enriched by the growing interest in personalized medicine and the identification of genetic predispositions to diseases, holds a prominent position in the market. The infectious diseases segment is driven by the need for accurate and rapid diagnostics due to the increasing prevalence of infectious pathogens. The 'others' segment, which includes applications like cardiovascular diseases, neurology, and reproductive health, is propelled by the expansion of molecular diagnostics into diverse medical specialties, all of which underscore the importance of reliable quality control measures in molecular diagnostics.
Breakup by End User:
- Hospitals
- Clinical Laboratories
- IVD Manufacturers and CROs
- Others
Clinical laboratories dominate the market
The report has provided a detailed breakup and analysis of the market based on the end user. This includes hospitals, clinical laboratories, IVD manufacturers and CROs, and others. According to the report, clinical laboratories represented the largest segment.
Hospitals, dealing with a high volume of patients, require reliable quality control solutions to ensure precise molecular testing results, critical for effective treatment decisions. Clinical laboratories, key players in conducting molecular diagnostic tests, prioritize quality control solutions to maintain high standards of accuracy and reliability. IVD manufacturers and CROs, involved in the development and commercialization of molecular diagnostic assays, mainly incorporate quality controls into their product design and production. The 'others' segment, encompassing diverse end users, also integrates molecular diagnostics into their operations, highlighting the broad applicability of molecular quality controls across healthcare and research settings.
Breakup by Region:
- North America
- United States
- Canada
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Others
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America exhibits a clear dominance, accounting for the largest molecular quality controls market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (Germany, France, the United Kingdom, Italy, Spain, and others); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America accounted for the largest market share.
North America's advanced healthcare infrastructure and strong focus on research and development have fostered the growth of the molecular quality controls market. The well-established healthcare systems, coupled with high healthcare expenditure, enable the adoption of molecular diagnostics and quality control solutions. The rising prevalence of chronic diseases further stimulates the market. Europe's well-developed healthcare framework and extensive clinical laboratory network also contribute to the market growth. The region's ageing population and increasing chronic diseases have elevated the demand for advanced molecular diagnostics, necessitating effective quality control measures.
In Asia Pacific, the swiftly expanding healthcare infrastructure, increasing awareness about personalized medicine, and a large patient population support the market's growth. The escalating prevalence of infectious diseases and chronic conditions boosts the adoption of molecular diagnostics and quality control solutions. In Latin America and the Middle East and Africa, improvements in healthcare infrastructure, rising awareness about molecular diagnostics, increased investments in healthcare, and a growing emphasis on disease management and prevention drive the market growth.
Competitive Landscape:
The competitive landscape of the global molecular quality controls market is characterized by intense competition and a diverse array of players vying for market share. Established multinational companies, regional players, and niche-focused firms all contribute to the market's dynamism. These companies compete based on product quality, technological advancements, regulatory compliance, pricing, and distribution networks. As the market expands, partnerships, collaborations, and mergers and acquisitions are becoming prevalent strategies to bolster market presence and expand product portfolios.
Additionally, players are investing significantly in research and development to introduce innovative molecular quality control solutions, catering to evolving customer needs and regulatory requirements. Furthermore, market leaders emphasize building strong brand equity and establishing themselves as trusted experts in the domain of molecular diagnostics. As healthcare providers and laboratories prioritize accuracy and reliability in diagnostic testing, competition among players in the molecular quality controls market is expected to remain fierce and result in continuous advancements and improved solutions.
The report has provided a comprehensive analysis of the competitive landscape in the market. Detailed profiles of all major companies have also been provided. Some of the key players in the market include:
- Bio-Rad Laboratories Inc.
- F. Hoffmann-La Roche AG
- Maine Molecular Quality Controls Inc.
- Microbiologics
- Qnostics Ltd.
- Quidel Corporation
- SeraCare Life Sciences Inc. (LGC Clinical Diagnostics)
- Thermo Fisher Scientific Inc.
- ZeptoMetrix Corporation (Antylia Scientific)
Molecular Quality Controls Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Million USD |
| Scope of the Report | Exploration of Historical and Forecast Trends, Industry Catalysts and Challenges, Segment-Wise Historical and Predictive Market Assessment:
|
| Product Types Covered | Independent Control, Instrument Specific-Control |
| Analyte Types Covered | Single-Analyte Controls, Multi-Analyte Controls |
| Applications Covered | Oncology, Genetic Testing, Infectious Diseases, Others |
| End Users Covered | Hospitals, Clinical Laboratories, IVD Manufacturers and CROs, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Bio-Rad Laboratories Inc., F. Hoffmann-La Roche AG, Maine Molecular Quality Controls Inc., Microbiologics, Qnostics Ltd., Quidel Corporation, SeraCare Life Sciences Inc. (LGC Clinical Diagnostics), Thermo Fisher Scientific Inc., ZeptoMetrix Corporation (Antylia Scientific), etc |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report:
- How has the global molecular quality controls market performed so far, and how will it perform in the coming years?
- What are the drivers, restraints, and opportunities in the global molecular quality controls market?
- What is the impact of each driver, restraint, and opportunity on the global molecular quality controls market?
- What are the key regional markets?
- Which countries represent the most attractive molecular quality controls market?
- What is the breakup of the market based on the product type?
- Which is the most attractive product type in the molecular quality controls market?
- What is the breakup of the market based on the analyte type?
- Which is the most attractive analyte type in the molecular quality controls market?
- What is the breakup of the market based on the application?
- Which is the most attractive application in the molecular quality controls market?
- What is the breakup of the market based on the end user?
- Which is the most attractive end user in the molecular quality controls market?
- What is the competitive structure of the global molecular quality controls market?
- Who are the key players/companies in the global molecular quality controls market?
Key Benefits for Stakeholders:
- IMARC’s report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the molecular quality controls market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global molecular quality controls market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assist stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the molecular quality controls industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.

 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












