
Neuroendovascular Coil Market Report by Product Type (Guglielmi Detachable Coils (GDCs), Matrix Coils), Indication (Ruptured Aneurysm, Unruptured Aneurysm), Application (Tumor Surgery, Medical Teaching, and Others), End User (Hospitals, Ambulatory Surgical Centers (ASCs), Stroke Centers, and Others), and Region 2026-2034
Market Overview:
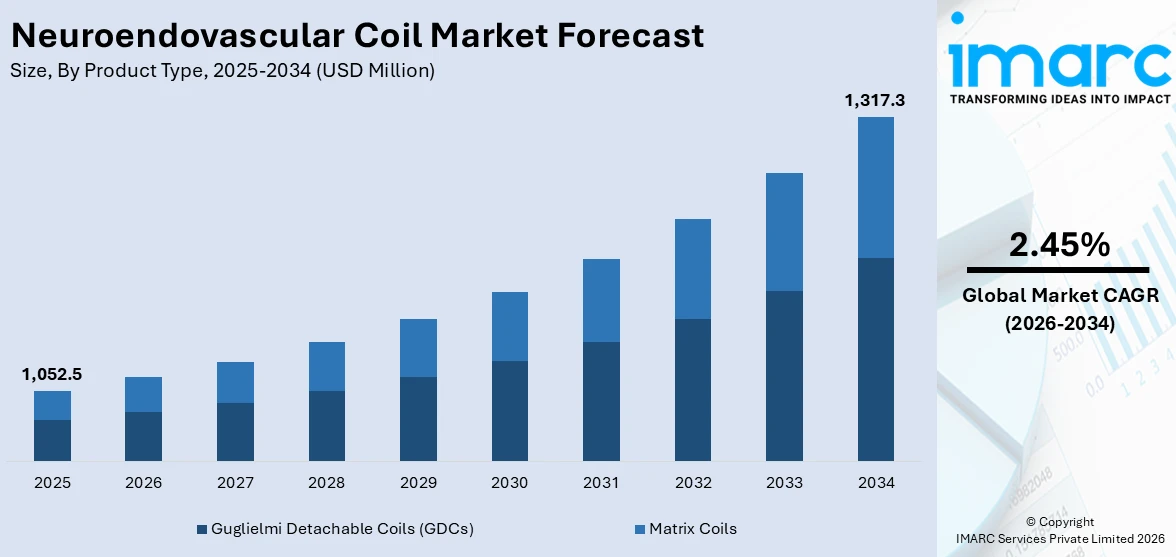
The global neuroendovascular coil market size reached USD 1,052.5 Million in 2025. Looking forward, IMARC Group expects the market to reach USD 1,317.3 Million by 2034, exhibiting a growth rate (CAGR) of 2.45% during 2026-2034.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 1,052.5 Million |
|
Market Forecast in 2034
|
USD 1,317.3 Million |
| Market Growth Rate (2026-2034) | 2.45% |
Neuroendovascular coiling is a minimally invasive endovascular treatment which is performed to block the blood flow in a blood vessel in the brain. It is essential to treat an aneurysm, a bulge caused by weakness in the wall of a blood vessel, which can rupture due to rising pressure and bleed into the brain. The symptoms of a ruptured aneurysm generally include sudden onset of a severe headache, nausea, vomiting, blurred or double vision, and seizure. In neuroendovascular coiling, a surgeon tightly packs the aneurysm with detachable platinum coils that promote blood clotting and reduce pressure on the outer wall. Neuroendovascular coiling is becoming popular across the globe as it incurs less cost and lowers the risk for the patients.
To get more information on this market Request Sample
Several advances have been made in the neuroendovascular coil technology in terms of shape, length and bioactivity. Besides this, significant progress has been made in intracranial stents to improve their deployment and delivery. Surgeons have started integrating intracranial stents with the coil technology to treat aneurysms and reduce issues related to recurrence. Moreover, endovascular coiling is being considered a better surgical technique than clipping regarding postoperative complications, favorable outcomes and rehabilitation. Other than this, various market players are investing in the introduction of technologically advanced products. For instance, in 2018, Cerenovus, a division of the Johnson & Johnson Medical Devices Companies, launched the Galaxy G3 Mini embolic coil which is designed to have an irregular shape to enable the device to fill open spaces in cerebral aneurysms.
IMARC Group’s latest report provides a deep insight into the global neuroendovascular coil market covering all its essential aspects. This ranges from macro overview of the market to micro details of the industry performance, recent trends, key market drivers and challenges, SWOT analysis, Porter’s five forces analysis, value chain analysis, etc. This report is a must-read for entrepreneurs, investors, researchers, consultants, business strategists, and all those who have any kind of stake or are planning to foray into the neuroendovascular coil market in any manner.
Key Market Segmentation:
IMARC Group provides an analysis of the key trends in each sub-segment of the global neuroendovascular coil market report, along with forecasts at the global and regional level from 2026-2034. Our report has categorized the market based on product type, indication, application, and end user.
Breakup by Product Type:
- Guglielmi Detachable Coils (GDCs)
- Matrix Coils
Breakup by Indication:
- Ruptured Aneurysm
- Unruptured Aneurysm
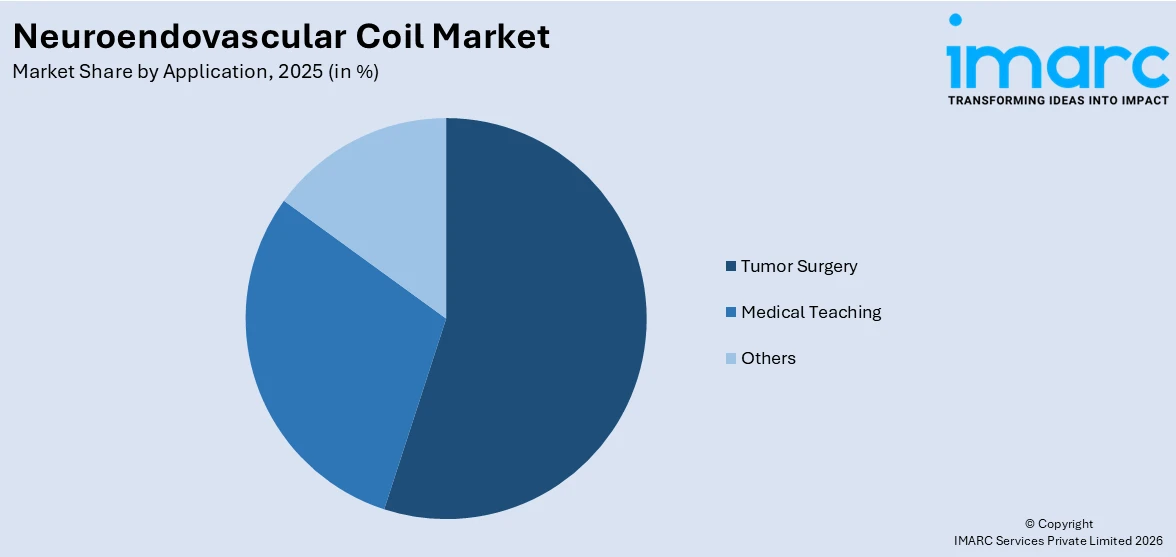
Breakup by Application:
Access the comprehensive market breakdown Request Sample
- Tumor Surgery
- Medical Teaching
- Others
Breakup by End User:
- Hospitals
- Ambulatory Surgical Centers (ASCs)
- Stroke Centers
- Others
Breakup by Region:
- North America
- Europe
- Asia Pacific
- Middle East and Africa
- Latin America
Competitive Landscape:
The report has also analysed the competitive landscape of the market with some of the key players being Medtronic, Stryker Corporation, DePuy Synthes, Penumbra, Johnson & Johnson Services Inc., Terumo Corporation, Cardinal Health, Boston Scientific Corporation, Cook Medical Incorporated, Balt Extrusion, EndoShape, etc.
Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Million USD |
| Segment Coverage | Product Type, Indication, Application, End User, Region |
| Region Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Companies Covered | Medtronic, Stryker Corporation, DePuy Synthes, Penumbra, Johnson & Johnson Services Inc., Terumo Corporation, Cardinal Health, Boston Scientific Corporation, Cook Medical Incorporated, Balt Extrusion and EndoShape |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report:
- How has the global neuroendovascular coil market performed so far and how will it perform in the coming years?
- What are the key regional markets in the global neuroendovascular coil industry?
What has been the impact of COVID-19 on the global neuroendovascular coil industry? - What is the breakup of the market based on the product type?
- What is the breakup of the market based on the indication?
- What is the breakup of the market based on the application?
- What is the breakup of the market based on the end user?
- What are the various stages in the value chain of the global neuroendovascular coil industry?
- What are the key driving factors and challenges in the global neuroendovascular coil industry?
- What is the structure of the global neuroendovascular coil industry and who are the key players?
- What is the degree of competition in the neuroendovascular coil industry?
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












