
Oseltamivir (Tamiflu) Manufacturing Plant Project Report 2026: Industry Trends, Plant Setup, Machinery, Raw Materials, Investment Opportunities, Cost and Revenue
Report Overview:
IMARC Group’s report, titled “Oseltamivir (Tamiflu) Manufacturing Plant Project Report 2026: Industry Trends, Plant Setup, Machinery, Raw Materials, Investment Opportunities, Cost and Revenue” provides a complete roadmap for setting up an oseltamivir (Tamiflu) manufacturing plant. It covers a comprehensive market overview to micro-level information such as unit operations involved, raw material requirements, utility requirements, infrastructure requirements, machinery and technology requirements, manpower requirements, packaging requirements, transportation requirements, etc. The oseltamivir (Tamiflu) project report provides detailed insights into project economics, including capital investments, project funding, operating expenses, income and expenditure projections, fixed costs vs. variable costs, direct and indirect costs, expected ROI and net present value (NPV), profit and loss account, financial analysis, etc.
-manufacturing-plant-project-report--(1).webp)
Oseltamivir, commonly marketed as Tamiflu, is a medication that holds a crucial role in the realm of influenza treatment and prevention. It belongs to a class of antiviral drugs known as neuraminidase inhibitors and is highly effective in combatting the influenza virus, including both influenza A and B strains. Tamiflu works by inhibiting the virus's ability to replicate and spread within the body, reducing the severity and duration of flu symptoms. It plays a pivotal role during flu outbreaks and pandemics, offering a means to mitigate the spread of the virus and alleviate symptoms, particularly in high-risk populations. As a trusted antiviral medication, oseltamivir (Tamiflu) remains essential in the global effort to manage and control influenza infections.
The key advantage of oseltamivir lies in reducing the severity and duration of flu symptoms, helping patients recover more quickly and lowering the risk of complications, especially in vulnerable populations. Secondly, oseltamivir is effective in both the treatment and prophylaxis of influenza, making it suitable for managing outbreaks and preventing the virus's spread in high-risk environments, such as healthcare facilities and households. Moreover, its oral formulation and relatively mild side effects make it a convenient and well-tolerated option for a wide range of patients, including children and the elderly. Thus, it stands as a crucial medication for combating influenza, offering effectiveness, versatility, and accessibility in the fight against this contagious respiratory illness.
Several factors drive the oseltamivir market, contributing to its steady growth. The most significant driver is the recurring threat of seasonal influenza. Secondly, the increasing awareness of the importance of early intervention in influenza cases is a significant driver. Oseltamivir’s efficacy in reducing symptom severity and duration when administered promptly encourages its use as a first-line treatment option. Moreover, global travel and urbanization trends have increased the risk of influenza transmission, further boosting the demand for oseltamivir. Additionally, the availability of generic versions of oseltamivir enhances affordability and accessibility to the medication. Furthermore, research into new formulations, delivery methods, and antiviral agents for influenza treatment and prevention continues to drive innovation within the market. In conclusion, the market for oseltamivir is driven by factors, such as the recurring threat of influenza, early intervention emphasis, global travel patterns, accessibility, and ongoing research, making it a crucial component of influenza management strategies.
The following aspects have been covered in the oseltamivir (Tamiflu) manufacturing plant report:
- Market Analysis:
- Market Performance
- Market Breakup by Segment
- Market Breakup by Region
- Price Analysis
- Impact of COVID-19
- Market Outlook
The report provides insights into the landscape of the oseltamivir (Tamiflu) industry at the global level. The report also provides a segment-wise and region-wise breakup of the global oseltamivir (Tamiflu) industry. Additionally, it also provides the price analysis of feedstocks used in the manufacturing of oseltamivir (Tamiflu), along with the industry profit margins.
- Detailed Process Flow:
- Product Overview
- Unit Operations Involved
- Mass Balance and Raw Material Requirements
- Quality Assurance Criteria
- Technical Tests
The report also provides detailed information related to the process flow and various unit operations involved in an oseltamivir (Tamiflu) manufacturing plant. Furthermore, information related to mass balance and raw material requirements has also been provided in the report with a list of necessary quality assurance criteria and technical tests.
- Project Details, Requirements and Costs Involved:
- Land, Location and Site Development
- Plant Layout
- Machinery Requirements and Costs
- Raw Material Requirements and Costs
- Packaging Requirements and Costs
- Transportation Requirements and Costs
- Utility Requirements and Costs
- Human Resource Requirements and Costs
The report provides a detailed location analysis covering insights into the land location, selection criteria, location significance, environmental impact, and expenditure for setting up an oseltamivir (Tamiflu) manufacturing plant. Additionally, the report also provides information related to plant layout and factors influencing the same. Furthermore, other requirements and expenditures related to machinery, raw materials, packaging, transportation, utilities, and human resources have also been covered in the report.
Capital Expenditure (CapEx) and Operational Expenditure (OpEx) Analysis:
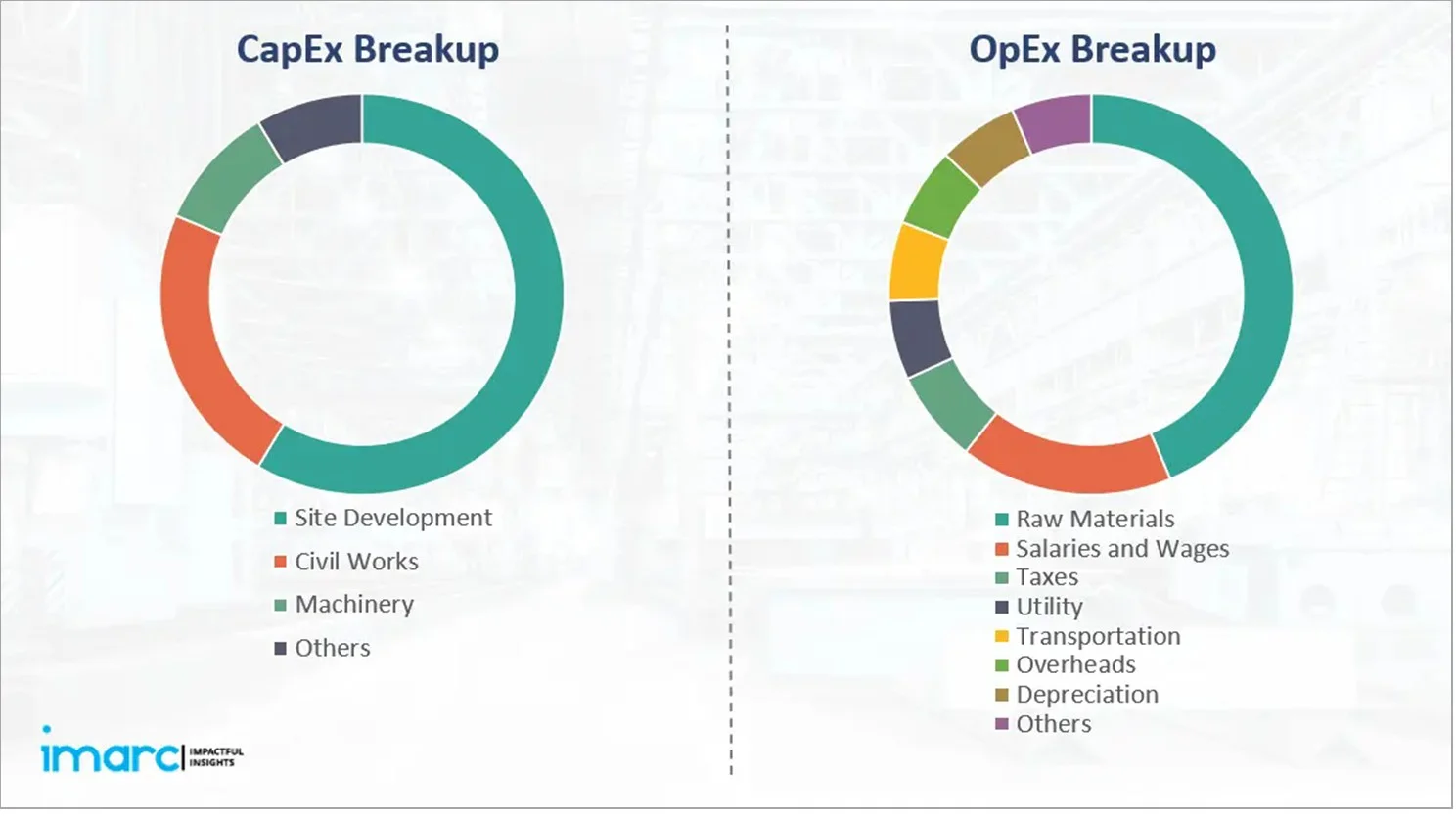
- Project Economics:
- Capital Investments
- Operating Costs
- Expenditure Projections
- Revenue Projections
- Taxation and Depreciation
- Profit Projections
- Financial Analysis
The report also covers a detailed analysis of the project economics for setting up an oseltamivir (Tamiflu) manufacturing plant. This includes the analysis and detailed understanding of capital expenditure (CapEx), operating expenditure (OpEx), income projections, taxation, depreciation, liquidity analysis, profitability analysis, payback period, NPV, uncertainty analysis, and sensitivity analysis. Furthermore, the report also provides a detailed analysis of the regulatory procedures and approvals, information related to financial assistance, along with a comprehensive list of certifications required for setting up an oseltamivir (Tamiflu) manufacturing plant.
Profitability Analysis:
| Particulars | Unit | Year 1 | Year 2 | Year 3 | Year 4 | Year 5 |
|---|---|---|---|---|---|---|
| Total Income | US$ | XX | XX | XX | XX | XX |
| Total Expenditure | US$ | XX | XX | XX | XX | XX |
| Gross Profit | US$ | XX | XX | XX | XX | XX |
| Gross Margin | % | XX | XX | XX | XX | XX |
| Net Profit | US$ | XX | XX | XX | XX | XX |
| Net Margin | % | XX | XX | XX | XX | XX |
Report Coverage:
| Report Features | Details |
|---|---|
| Product Name | Oseltamivir (Tamiflu) |
| Report Coverage | Detailed Process Flow: Unit Operations Involved, Quality Assurance Criteria, Technical Tests, Mass Balance, and Raw Material Requirements Land, Location and Site Development: Selection Criteria and Significance, Location Analysis, Project Planning and Phasing of Development, Environmental Impact, Land Requirement and Costs Plant Layout: Importance and Essentials, Layout, Factors Influencing Layout Plant Machinery: Machinery Requirements, Machinery Costs, Machinery Suppliers (Provided on Request) Raw Materials: Raw Material Requirements, Raw Material Details and Procurement, Raw Material Costs, Raw Material Suppliers (Provided on Request) Packaging: Packaging Requirements, Packaging Material Details and Procurement, Packaging Costs, Packaging Material Suppliers (Provided on Request) Other Requirements and Costs: Transportation Requirements and Costs, Utility Requirements and Costs, Energy Requirements and Costs, Water Requirements and Costs, Human Resource Requirements and Costs Project Economics: Capital Costs, Techno-Economic Parameters, Income Projections, Expenditure Projections, Product Pricing and Margins, Taxation, Depreciation Financial Analysis: Liquidity Analysis, Profitability Analysis, Payback Period, Net Present Value, Internal Rate of Return, Profit and Loss Account, Uncertainty Analysis, Sensitivity Analysis, Economic Analysis Other Analysis Covered in The Report: Market Trends and Analysis, Market Segmentation, Market Breakup by Region, Price Trends, Competitive Landscape, Regulatory Landscape, Strategic Recommendations, Case Study of a Successful Venture |
| Currency | US$ (Data can also be provided in the local currency) |
| Customization Scope | The report can also be customized based on the requirement of the customer |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report?
- How has the oseltamivir (Tamiflu) market performed so far and how will it perform in the coming years?
- What is the market segmentation of the global oseltamivir (Tamiflu) market?
- What is the regional breakup of the global oseltamivir (Tamiflu) market?
- What are the price trends of various feedstocks in the oseltamivir (Tamiflu) industry?
- What is the structure of the oseltamivir (Tamiflu) industry and who are the key players?
- What are the various unit operations involved in an oseltamivir (Tamiflu) manufacturing plant?
- What is the total size of land required for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What is the layout of an oseltamivir (Tamiflu) manufacturing plant?
- What are the machinery requirements for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What are the raw material requirements for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What are the packaging requirements for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What are the transportation requirements for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What are the utility requirements for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What are the human resource requirements for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What are the infrastructure costs for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What are the capital costs for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What are the operating costs for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What should be the pricing mechanism of the final product?
- What will be the income and expenditures for an oseltamivir (Tamiflu) manufacturing plant?
- What is the time required to break even?
- What are the profit projections for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What are the key success and risk factors in the oseltamivir (Tamiflu) industry?
- What are the key regulatory procedures and requirements for setting up an oseltamivir (Tamiflu) manufacturing plant?
- What are the key certifications required for setting up an oseltamivir (Tamiflu) manufacturing plant?
Report Customization
While we have aimed to create an all-encompassing oseltamivir (Tamiflu) plant project report, we acknowledge that individual stakeholders may have unique demands. Thus, we offer customized report options that cater to your specific requirements. Our consultants are available to discuss your business requirements, and we can tailor the report's scope accordingly. Some of the common customizations that we are frequently requested to make by our clients include:
- The report can be customized based on the location (country/region) of your plant.
- The plant’s capacity can be customized based on your requirements.
- Plant machinery and costs can be customized based on your requirements.
- Any additions to the current scope can also be provided based on your requirements.
Why Buy IMARC Reports?
- The insights provided in our reports enable stakeholders to make informed business decisions by assessing the feasibility of a business venture.
- Our extensive network of consultants, raw material suppliers, machinery suppliers and subject matter experts spans over 100+ countries across North America, Europe, Asia Pacific, South America, Africa, and the Middle East.
- Our cost modeling team can assist you in understanding the most complex materials. With domain experts across numerous categories, we can assist you in determining how sensitive each component of the cost model is and how it can affect the final cost and prices.
- We keep a constant track of land costs, construction costs, utility costs, and labor costs across 100+ countries and update them regularly.
- Our client base consists of over 3000 organizations, including prominent corporations, governments, and institutions, who rely on us as their trusted business partners. Our clientele varies from small and start-up businesses to Fortune 500 companies.
- Our strong in-house team of engineers, statisticians, modeling experts, chartered accountants, architects, etc. have played a crucial role in constructing, expanding, and optimizing sustainable manufacturing plants worldwide.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
Frequently Asked Questions
Our feasibility studies assess several key factors to provide a detailed evaluation of your project's potential. The study includes a pricing analysis of feedstocks, helping to understand industry profit margins and cost variations. Detailed insights into mass balance, unit operations, raw material requirements, and the manufacturing process flow are also provided to ensure a clear understanding of the production setup.
The study also covers critical elements such as location analysis, environmental impact, plant layout, and costs associated with land, machinery, raw materials, packaging, transportation, utilities, and human resources. The project economics section provides an in-depth analysis of capital expenditure (CapEx), operating expenditure (OpEx), income projections, taxation, depreciation, liquidity, profitability, payback period, net present value (NPV), uncertainty, and sensitivity analysis.
Additionally, IMARC Group's feasibility studies address regulatory procedures, financial assistance, and necessary certifications, ensuring all legal and compliance requirements are met. By covering these aspects, IMARC Group's feasibility studies support informed decision-making, risk reduction, and the overall viability of your business.
Yes, our site identification services include a thorough assessment of potential sites based on proximity to raw materials, ensuring cost-effective and timely supply chain operations. We also consider access to infrastructure, such as transportation networks, utilities, and technology, which are essential for smooth plant operations. Labor availability is another key factor we analyze to ensure that the site has access to a skilled workforce. Additionally, we evaluate the environmental impact to ensure compliance with regulations and sustainable practices. By integrating these factors, we provide a comprehensive site evaluation to identify the optimal location for your new plant, supporting your strategic goals and operational efficiency.
Yes, our regulatory approvals and licensing services are made specifically to support companies in effectively and efficiently navigating the ever-changing regulatory environment. We start by carefully evaluating your company's requirements as well as the unique regulatory framework that applies to your sector. After that, you are assisted by our team of professionals throughout the whole process of securing the necessary industrial permits, business licenses, and environmental clearances.
Additionally, we help with industry-specific regulatory licensing so that your company complies with all sector-specific regulations. To further safeguard your brand and innovations from the outset, we also offer support for intellectual property rights (IPR) registrations and licensing. We manage the required documentation and communicate with pertinent authorities on your behalf by utilizing our deep industry experience and painstaking attention to detail. This allows you to focus on building and growing your business while we take care of the regulatory complexities. Partnering with us ensures that your business is fully compliant and ready to thrive from the start.
Our skilled engineering staff specializes in designing efficient and effective plant layouts that meet your unique needs. We begin with a comprehensive process design to ensure the best possible workflow and use of resources.
Our services include careful equipment selection, ensuring that your plant design incorporates the most suitable and advanced machinery. We also focus on meticulous layout planning, strategically arranging workstations and equipment to optimize output and reduce operational bottlenecks. Furthermore, our facility design takes into account every important factor, such as regulatory compliance, scalability, and safety.
By leveraging our expertise, we ensure that your plant layout not only meets your current operational needs but is also adaptable to future growth and technological advancements. Our goal is to create a well-organized, efficient, and compliant facility that enhances your overall operational efficiency and supports your business objectives.
Yes, our raw material and machinery sourcing services are designed to identify and connect you with reliable and affordable providers. We leverage our extensive network and industry expertise to source high-quality raw materials and advanced machinery that meet your specific requirements.
We also conduct supplier audits and evaluations so that you get the best value for your investment. We evaluate potential suppliers on the basis of cost, quality, reliability, and delivery timelines. We also make sure that all the legal requirements are met, including customs, tax regulations, labor laws, and import/export laws, to ensure you are fully compliant.
When you choose to work with us, you gain access to a list of suppliers pre-screened for affordability and reliability, helping you optimize your procurement process and reduce overall operational costs. It is our goal to assist you in establishing a new plant effectively and economically, ensuring long-term success and sustainability.
IMARC Group can effectively manage the construction of your new facility through our comprehensive construction management services. Our dedicated construction management team oversees the entire construction phase, ensuring that all activities are executed according to plan and within the specified timeline. We manage contractors, coordinating their efforts to ensure smooth and efficient workflow on-site.
Furthermore, our team pays great attention to the progress of the construction work, and we conduct site reviews and quality control to ensure that the construction work is done to the highest standards. We resolve any problems as they arise, thus avoiding any form of delay and keeping the project on schedule. When you work with us, you can be assured that your new facility will be constructed to the highest standards that will enable you to concentrate on your core business activities.
Yes, our distributor identification services are designed to connect companies with trusted distributors who meet high standards of reliability, performance, and ethical practices. We leverage our vast network of vetted distributors across various industries and regions to find the best match for your business needs.
Our process begins with a careful analysis of your particular needs and business goals. We then carry out rigorous research and due diligence to identify the distributors that have market reach and the capability to manage your product lines effectively. Our thorough vetting process involves the distributors' background checks and performance history reviews to ensure that they meet industry standards and have a proven track record.
Also, we ensure compliance with the relevant legislation and regulations to avoid legal issues that may affect the functioning of the company. Once potential distributors are identified, we organize business meetings, as well as handle all logistics to facilitate productive discussions. We provide continuous support throughout the partnership, including performance monitoring and logistics coordination, to ensure the success and optimization of your distribution networks in the long run.
Yes, our factory audit services provide a systematic and thorough evaluation of your plant's performance across various critical parameters. Our experienced auditors utilize industry best practices to conduct detailed inspections, focusing on safety, quality, efficiency, compliance, and sustainability.
Our audits assess safety protocols to ensure a secure working environment, evaluate quality standards to enhance product satisfaction and optimize processes to increase productivity and reduce costs. Compliance checks ensure adherence to relevant regulations, mitigating legal risks, and our sustainability assessments recommend eco-friendly practices to minimize environmental impact.
Our holistic approach delivers actionable insights through detailed audit reports, offering clear recommendations for continuous improvement. By partnering with us, you gain valuable insights into your plant's operations, enabling you to drive enhancements, achieve operational excellence, and maintain a competitive edge.
Yes, our contract manufacturer identification services are designed to assist businesses in navigating the complex landscape of contract manufacturing. We start by defining the ideal contract manufacturer through in-depth discussions with our clients, followed by extensive research to identify potential candidates.
Once potential manufacturers are identified, we conduct a rigorous evaluation and vetting process to ensure they meet high standards of quality, reliability, and compliance with relevant regulations. Our team handles the pre-screening process, arranging meetings between our clients and interested candidates, and managing all logistics to facilitate productive discussions.
We have strict performance monitoring and quality control checks to ensure the manufacturers remain at par with your expectations and contribute positively to your business throughout the partnership lifecycle. Through our expertise and extensive network, we ensure that you partner with capable and credible contract manufacturers, who help you drive efficiency, quality, and growth in your production processes.
IMARC Group offers a comprehensive suite of marketing and sales services dedicated to helping businesses increase revenues and effectively sell products. Our performance marketing services focus on data-driven campaigns that maximize return on investment, utilizing programmatic advertising and retargeting techniques to reach the right audience at the right time. We enhance your online presence through SEO and SMO, driving organic traffic and improving your digital footprint.
The lead generation strategies we use target high-quality quality leads that convert into loyal clients, while our brand promotion strategies including influencer marketing and creative designing enhance the visibility and credibility of your brand. Our social media management services assist you in engaging with your audience, establishing relationships with them, and driving conversions through targeted campaigns.
By partnering with IMARC Group, you benefit from our approach that is tailored to your business needs and unique objectives. Our strategies cover all aspects of digital marketing so that you can reach your target audience consistently, along with our ongoing support and optimization to keep your campaigns effective and relevant. Our cutting edge and performance driven marketing services help you gain the competitive advantage and ensure long-term success.








 Inquire Before Buying
Inquire Before Buying
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Request Customization
Request Customization