
Real-World Evidence Solutions Market Report by Component (Clinical Settings Data, Claims Data, Patient-Powered Data, Pharmacy Data, and Others), Therapeutic Area (Oncology, Immunology, Neurology, Cardiovascular Disease, and Others), End User (Pharmaceutical and Medical Device Companies, Healthcare Payers, Healthcare Providers, and Others), and Region 2026-2034
Market Overview:
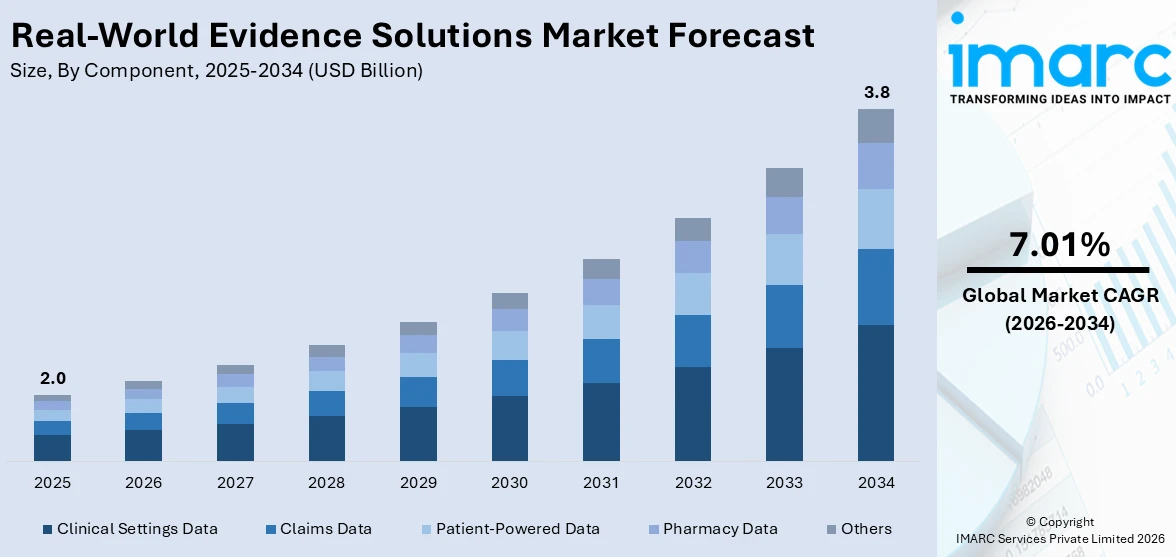
The global real-world evidence solutions market size reached USD 2.0 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 3.8 Billion by 2034, exhibiting a growth rate (CAGR) of 7.01% during 2026-2034.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
| Market Size in 2025 | USD 2.0 Billion |
| Market Forecast in 2034 | USD 3.8 Billion |
| Market Growth Rate (2026-2034) | 7.01% |
Real-world evidence (RWE) refers to clinical evidence about the use and potential benefits or risks of medical products via real-world data (RWD). It can be produced via different study designs or analyses, including randomized trials, big simple trials, pragmatic trials, and observational studies. It provides insight into real-world treatment patterns in patient groups, including dosing, compliance, adherence, off-label use, and the balance between efficacy and safety. As it can also assist researchers in discovering possible patients and developing correct inclusion criteria for clinical trials, which can help them design better pre-trial studies, there is a rise in the demand for RWE solutions around the world.
To get more information on this market Request Sample
Real-World Evidence Solutions Market Trends:
The growing demand for RWE solutions to get precise and clear information about the safety and efficacy of new products represents one of the key factors driving the market. Moreover, there is a rise in the demand for these solutions to detect varied reactions, product subpopulation effects, and product value when utilized by complex and comorbid patients. This, along with the burgeoning healthcare industry, is propelling the growth of the market. Additionally, the growing utilization of advanced analytics and technological solutions in RWE for clinical development, commercialization, speed innovation, and accelerating improvements in healthcare outcomes is positively influencing the market. Besides this, there are various initiatives undertaken by governments of several countries to promote the use of RWE solutions, which provide the efficacy of drugs and vaccines. This, coupled with a shift from volume to value-based care, is offering lucrative growth opportunities to industry investors. Apart from this, the growing adoption of digitalization and patient-centered virtual care is catalyzing the demand for RWE solutions. Furthermore, there is an increase in the prevalence of chronic diseases, such as cancer, obesity, and diabetes, across the globe. In line with this, the rising delays in drug development and the subsequent increase in development costs are bolstering the growth of the market.
Key Market Segmentation:
IMARC Group provides an analysis of the key trends in each sub-segment of the global real-world evidence solutions market report, along with forecasts at the global, regional and country levels from 2026-2034. Our report has categorized the market based on component, therapeutic area and end user.
Breakup by Component:
- Clinical Settings Data
- Claims Data
- Patient-Powered Data
- Pharmacy Data
- Others
Breakup by Therapeutic Area:
- Oncology
- Immunology
- Neurology
- Cardiovascular Disease
- Others
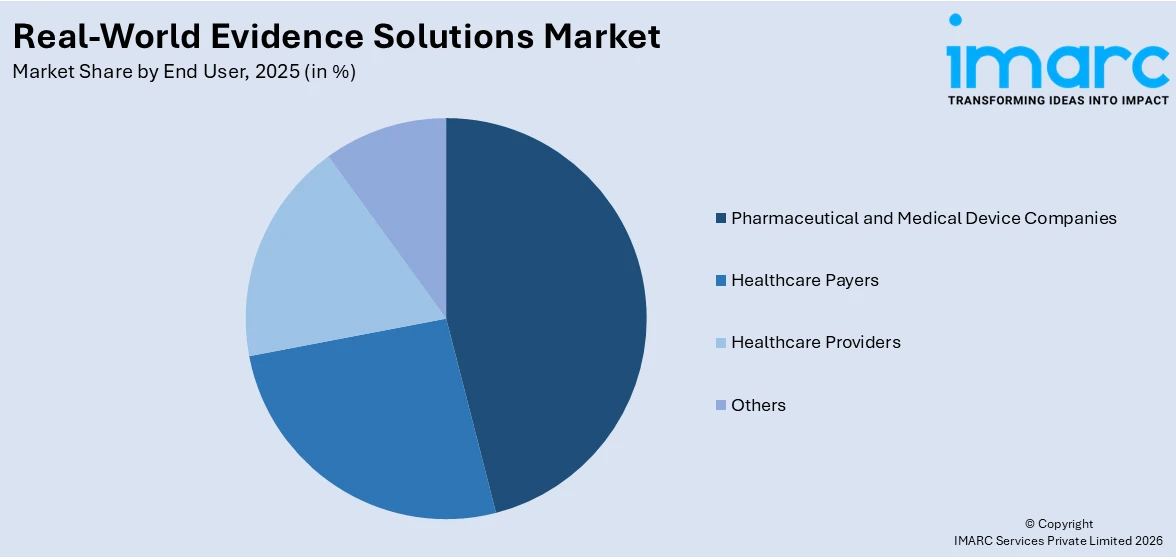
Breakup by End User:
Access the comprehensive market breakdown Request Sample
- Pharmaceutical and Medical Device Companies
- Healthcare Payers
- Healthcare Providers
- Others
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
Competitive Landscape:
The report provides a comprehensive analysis of the competitive landscape in the global real-world evidence solutions market with detailed profiles of all major companies, including:
- Aetion Inc.
- Clinigen Limited
- Flatiron Health
- ICON plc
- IQVIA Inc.
- Optum, Inc.
- Oracle Corporation
- Parexel International (MA) Corporation
- SAS Institute Inc.
- Syneos Health
- Thermo Fisher Scientific Inc.
Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Segment Coverage | Component, Therapeutic Area, End User, Region |
| Region Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Aetion Inc., Clinigen Limited, Flatiron Health, ICON plc, IQVIA Inc., Optum, Inc., Oracle Corporation, Parexel International (MA) Corporation, SAS Institute Inc., Syneos Health, Thermo Fisher Scientific Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report
The global real-world evidence solutions market was valued at USD 2.0 Billion in 2025.
We expect the global real-world evidence solutions market to exhibit a CAGR of 7.01% during 2026-2034.
The rising demand for real-world evidence solutions to detect varied reactions, product subpopulation effects, and its value when utilized by complex and comorbid patients is primarily driving the global real-world evidence solutions market.
The sudden outbreak of the COVID-19 pandemic has led to the growing adoption for real-world evidence solutions to monitor the efficacy and safety of drugs and vaccines against the coronavirus infection.
Based on the component, the global real-world evidence solutions market can be categorized into clinical settings data, claims data, patient-powered data, pharmacy data, and others. Currently, clinical settings data holds the majority of the total market share.
Based on the therapeutic area, the global real-world evidence solutions market has been segregated into oncology, immunology, neurology, cardiovascular disease, and others. Among these, oncology currently exhibits a clear dominance in the market.
Based on the end user, the global real-world evidence solutions market can be bifurcated into pharmaceutical and medical device companies, healthcare payers, healthcare providers, and others. Currently, pharmaceutical and medical device companies account for the largest market share.
On a regional level, the market has been classified into North America, Asia-Pacific, Europe, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global real-world evidence solutions market include Aetion Inc., Clinigen Limited, Flatiron Health, ICON plc, IQVIA Inc., Optum, Inc., Oracle Corporation, Parexel International (MA) Corporation, SAS Institute Inc., Syneos Health, and Thermo Fisher Scientific Inc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












