
Recombinant Protein Market Size, Share, Trends and Forecast by Product, Application, End User, and Region, 2026-2034
Recombinant Protein Market Size and Share:
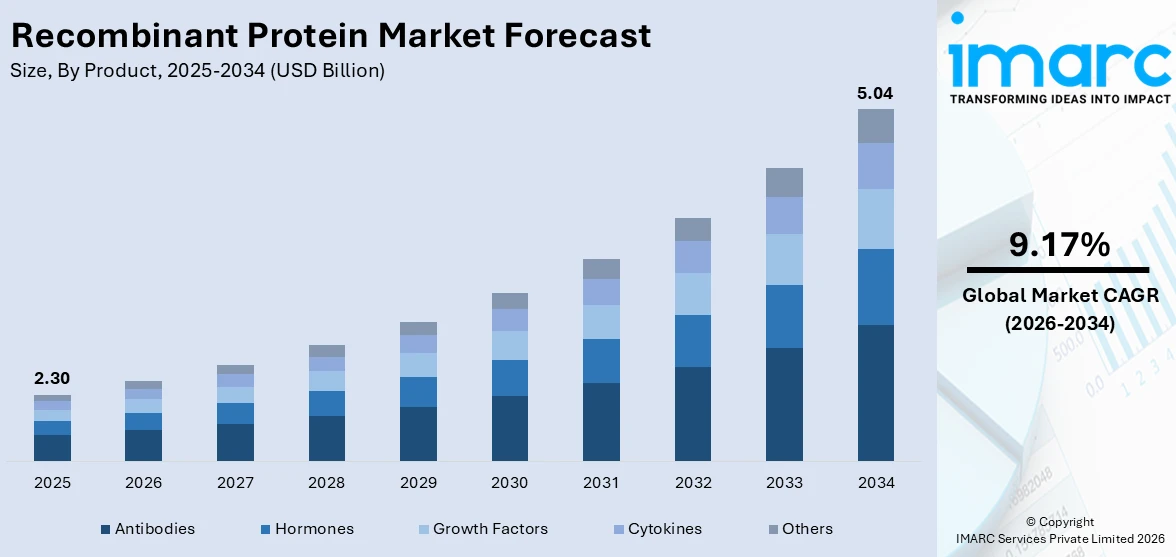
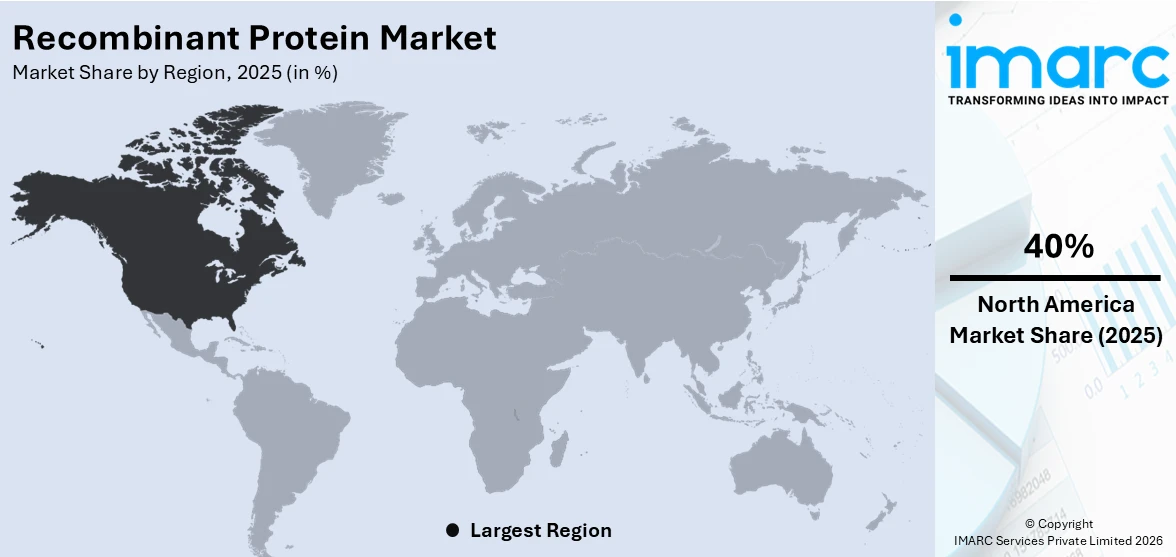
The global recombinant protein market size was valued at USD 2.3 Billion in 2025. Looking forward, IMARC Group estimates the market to reach USD 5.04 Billion by 2034, exhibiting a CAGR of 9.17% from 2026-2034. North America currently dominates the market, holding a market share of 40% in 2025. The region benefits from its robust biopharmaceutical infrastructure, high concentration of leading academic research centers, significant government funding directed toward life sciences, and strong adoption of biologics in clinical and therapeutic settings, all collectively expanding the recombinant protein market share.
The market is being mainly driven by the increasing global incidence of chronic and rare diseases, thus increasing the demand for advanced therapeutic solutions. The increasing trend of pharmaceutical and biopharmaceutical companies shifting their focus from small molecule therapies to biologics, such as recombinant proteins, has opened up immense opportunities for growth in various therapeutic areas. The increasing investment by government organizations across the globe in proteomics and genomics research is helping in the development of innovative recombinant protein-based solutions for diagnosis and treatment. The development of advanced recombinant protein expression platforms has helped in accelerating the development of high-quality autoantigens, which are largely used in immunotherapy procedures. In addition, the increasing use of recombinant proteins as research reagents in molecular biology, proteomics, and cell biology is increasing the applications of recombinant proteins. The increasing interest in the development of biosimilars, especially for off-patent biologic therapeutics, is helping in creating a favorable market outlook for innovation and large-scale manufacturing of recombinant proteins in the global life sciences industry.
The United States has emerged as a major region in the Recombinant Protein market owing to many factors. The country is home to one of the world’s most advanced biopharmaceutical ecosystems, supported by a dense network of research universities, specialized biotechnology clusters, and robust federal funding from agencies such as the National Institutes of Health. A mature venture capital environment enables continuous financing of early-stage recombinant protein therapeutics, accelerating the pipeline from laboratory research to commercial production. The regulatory environment in the United States provides structured pathways for the approval of biologics and biosimilars, supporting expedited market entry for innovative recombinant protein products, thereby supporting the recombinant protein market growth. According to data from the U.S. Food and Drug Administration, 50 new therapeutics were approved in 2024, with 16 being protein-based therapies, representing 32% of all approvals. This strong regulatory output reflects the country’s leadership in biopharmaceutical innovation and the sustained demand for recombinant protein market forecast solutions across multiple therapeutic areas.
To get more information on this market Request Sample
Recombinant Protein Market Trends:
Expanding Antibody-Based Therapeutic Pipelines
The development of antibody-based therapeutics has emerged as one of the most transformative trends shaping the global recombinant protein market. Recombinant antibodies, expressed through sophisticated mammalian and microbial cell systems, have become the cornerstone of modern oncology, immunology, and rare disease therapies due to their exceptional target specificity, potency, and well-characterized safety profiles. The expanding pipeline of monoclonal antibodies, bispecific antibodies, and antibody-drug conjugates has generated sustained demand for high-quality recombinant antibody proteins across both research and commercial manufacturing settings. Pharmaceutical and biopharmaceutical organizations are channeling considerable investments into next-generation antibody formats, leveraging protein engineering technologies to enhance binding affinity, reduce immunogenicity, and extend therapeutic half-lives. Recombinant antibody production is also being increasingly integrated into early-stage drug target validation, enabling precise identification of disease-associated biomarkers. In 2025, Spain's Grifols, a healthcare firm that produces plasma-derived medicines, revealed a partnership with the Netherlands' FcR Therapeutics, a biotech startup, to create recombinant nanobodies for autoimmune disease treatments.
AI Integration in Protein Engineering and Expression
The convergence of artificial intelligence with protein engineering is fundamentally reshaping recombinant protein production workflows across the biopharmaceutical and research sectors. Advanced computational tools, including machine learning algorithms and deep learning-based protein structure prediction models, are enabling researchers to design novel protein variants with enhanced stability, bioactivity, and manufacturability in considerably shorter development timelines. These innovations are directly reducing the time-to-clinic for recombinant therapeutics, improving production yields in microbial and mammalian expression systems, and enabling the development of previously structurally complex protein targets. Simultaneously, the integration of automation and high-throughput screening into protein production platforms is allowing more efficient identification of optimal expression conditions, contributing to scalable and cost-effective manufacturing. Regulatory agencies and government bodies are recognizing the importance of these technologies, with the United States Department of Defense revealed 12 more grants to bioindustrial companies through the Distributed Bioindustrial Manufacturing Program (DBIMP). This announcement elevates the DBIMP to 25 awards thus far, amounting to $42 million, anticipated to accelerate further adoption of AI-enabled platforms across the recombinant protein market outlook.
Rising Government Investments in Biopharmaceutical R&D
Heightened government investment in biopharmaceutical research and development is emerging as a defining driver of recombinant protein market forecast expansion across global markets. National governments and public funding bodies are increasingly recognizing recombinant proteins as strategically important assets for public health, economic competitiveness, and biosecurity, leading to the establishment of dedicated programs to advance their development and production. Funding initiatives spanning multiple geographies are supporting a range of activities, from early-stage academic research in protein science to commercial-scale biopharmaceutical manufacturing infrastructure development. These investments are catalyzing collaborations between government agencies, academic institutions, and private-sector biotechnology firms, creating innovation ecosystems that accelerate the pace of therapeutic protein discovery. Programs designed to expand domestic biologics production capacities are also reducing supply chain vulnerabilities identified during recent global health disruptions. India, for example, announced a USD 1.1 billion investment initiative to boost the development of biologics and biosimilars, explicitly including recombinant protein drugs among the prioritized modalities, reinforcing the significant role of public funding in driving recombinant protein market trends globally.
Recombinant Protein Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global recombinant protein market, along with forecast at the global, regional, and country levels from 2026-2034. The market has been categorized based on product, application, and end user.
Analysis by Product:
- Antibodies
- Hormones
- Growth Factors
- Cytokines
- Others
Antibodies hold 26% of the market share. Recombinant antibodies represent the most widely utilized product category within the recombinant protein market, reflecting their central role in therapeutic development, diagnostic assay design, and biomedical research. These proteins are produced using recombinant DNA technology to achieve high specificity, reproducibility, and batch-to-batch consistency, which are critical attributes for both clinical and laboratory applications. Recombinant antibodies are deployed extensively in immunotherapy regimens targeting cancer, autoimmune disorders, and inflammatory conditions, making them indispensable components of the modern biopharmaceutical pipeline. The segment benefits from rapid advancement of antibody engineering platforms, which have enabled the development of novel antibody formats including bispecific antibodies and antibody-drug conjugates that offer improved therapeutic precision. Moreover, governments are approving new therapeutics, demonstrating significant regulatory momentum behind antibody-based recombinant proteins and their expanding presence in global drug approval pipelines.
Analysis by Application:
Access the comprehensive market breakdown Request Sample
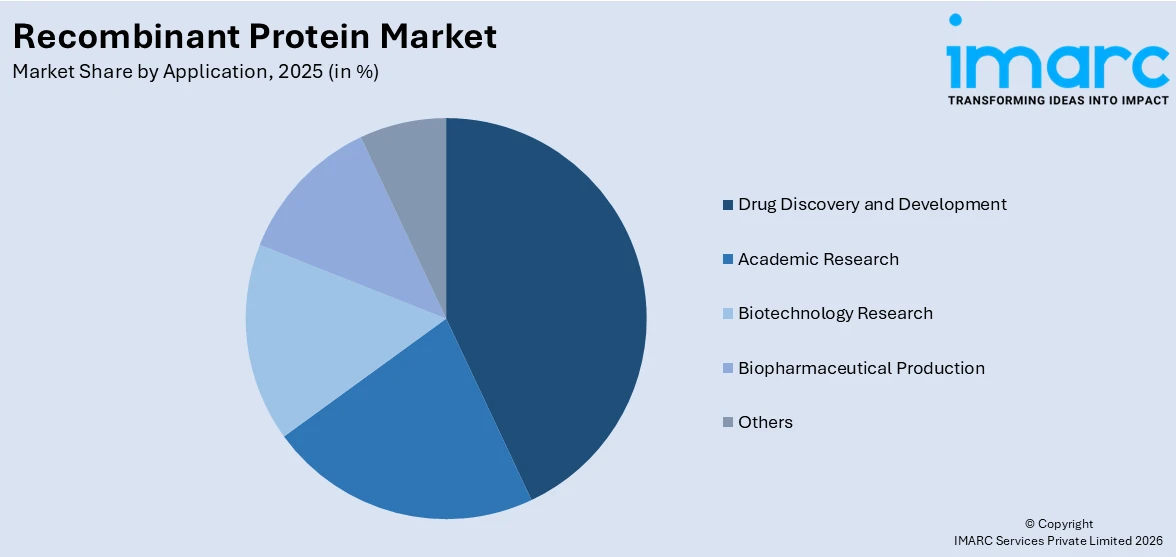
- Drug Discovery and Development
- Academic Research
- Biotechnology Research
- Biopharmaceutical Production
- Others
Drug discovery and development lead the market with a share of 50%. This segment’s commanding position reflects the indispensable role of recombinant proteins in identifying new therapeutic targets, validating drug candidates, and supporting the transition from preclinical models to clinical evaluation. Recombinant proteins serve as essential reagents and biological tools across multiple stages of the drug discovery process, including target identification, lead compound screening, structural biology studies, and preclinical efficacy assessment. The growing pipeline of biologic drugs, driven by the rising prevalence of cancer, diabetes, autoimmune disorders, and rare diseases, has significantly expanded the demand for highly specialized recombinant proteins in drug development workflows. Research organizations and biopharmaceutical companies are heavily investing in protein-based screening platforms that enable rapid and cost-effective evaluation of therapeutic candidates.
Analysis by End User:
- Pharmaceutical and Biopharmaceutical Companies
- Biotechnology Companies
- Academic and Research Institutes
- Contract Research Organization
- Others
Pharmaceutical and biopharmaceutical companies dominate the market, with a share of 61%. This segment’s leading position stems from the large-scale and multifaceted use of recombinant proteins throughout the drug discovery, development, and commercial manufacturing continuum. Pharmaceutical and biopharmaceutical organizations rely on recombinant proteins as critical research tools for biomarker identification, target validation, and lead optimization, as well as key inputs in the production of biologics including monoclonal antibodies, therapeutic enzymes, and hormones. The ongoing transition from traditional small-molecule drugs to biologic therapies has intensified demand for recombinant protein products, encouraging organizations to expand internal research capabilities and establish partnerships with contract research and manufacturing organizations. Increasing regulatory approvals for novel biologics across major markets are expected to further consolidate the dominance of this segment globally.
Regional Analysis:
To get more information on the regional analysis of this market Request Sample
- North America
- United States
- Canada
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America, accounting for 40% of the share, enjoys the leading position in the market. The region’s dominance is attributable to its highly developed biopharmaceutical industry ecosystem, which encompasses a dense concentration of leading research universities, technology transfer offices, and specialized biotechnology firms engaged in recombinant protein development and manufacturing. Government agencies in North America have consistently allocated substantial funding to life sciences research, creating a stable and innovation-supportive environment for the advancement of recombinant protein technologies. The region also benefits from a mature venture capital ecosystem that enables continuous financing of early-stage therapeutic protein programs, accelerating the journey from discovery to market entry. Well-established regulatory frameworks provide structured pathways for biologics and biosimilar approvals, enabling consistent throughput of recombinant protein-derived products.
Key Regional Takeaways:
United States Recombinant Protein Market Analysis
The United States has established itself as the primary growth engine of the global recombinant protein market, reflecting a powerful convergence of advanced biotechnology infrastructure, robust research and development investments, and a highly favorable regulatory environment. The country hosts a substantial concentration of pharmaceutical and biopharmaceutical companies, academic medical centers, and contract research organizations, each contributing to the continuous expansion of the recombinant protein development pipeline. Federal agencies including the National Institutes of Health and the Department of Defense have maintained substantial funding commitments to biomedical research and biomanufacturing capacity, creating a sustained innovation ecosystem that supports both foundational science and commercial development. The adoption of personalized medicine principles, alongside the rapid expansion of the biosimilar market following multiple biologic patent expirations, is driving additional demand for recombinant protein products. The country’s large and aging population, coupled with increasing incidence rates of chronic conditions such as cancer, diabetes, and autoimmune disorders, further strengthens the long-term demand environment. In 2025, Novavax, Inc. announced that the U.S. Food and Drug Administration has granted approval for the Nuvaxovid™ 2025-2026 Formula to prevent COVID-19 in individuals aged 65 and older, or those between 12 and 64 with at least one underlying condition that increases their risk for severe COVID-19 outcomes.
Europe Recombinant Protein Market Analysis
Europe represents the second-largest regional market for recombinant proteins, underpinned by a robust biopharmaceutical ecosystem characterized by strong academic-industry collaborations and a steadily expanding base of biosimilar development activities. Countries such as Germany, France, and the United Kingdom host world-class research institutions and pharmaceutical manufacturers that generate consistent demand for high-purity recombinant proteins across therapeutic research and commercial production stages. The European Medicines Agency provides a well-structured regulatory framework that facilitates the approval of biologics, creating transparent and efficient market entry pathways for recombinant protein-based products. The aging population across key European nations is driving elevated demand for protein-based therapeutics addressing oncology, metabolic diseases, and immunological conditions. Supportive regulatory frameworks are being actively reinforced by new policy initiatives like the European Commission adopted a landmark proposal on December 16, 2025 for the European Biotech Act, a regulatory framework specifically designed to strengthen the Union’s biotechnology and biomanufacturing sectors and accelerate the development and commercialization of biologics, including recombinant proteins, across member states.
Asia-Pacific Recombinant Protein Market Analysis
Asia-Pacific is emerging as one of the fastest-growing regions in the global recombinant protein market, driven by expanding biopharmaceutical manufacturing capacities, rising incidence of chronic diseases, and supportive government initiatives aimed at building domestic biotechnology capabilities. Countries including China, Japan, India, and South Korea are making significant strides in recombinant protein research, production, and commercialization, fueled by government funding programs and increasing foreign direct investments in biotech infrastructure. The region benefits from growing demand for biosimilars and biologics from rapidly expanding healthcare systems across diverse economies. India demonstrated particularly strong policy commitment to this sector, with the government announcing investment program to boost the development of biologics and biosimilars, explicitly including recombinant protein drugs among prioritized product categories, underscoring its strategic focus on building a globally competitive biopharmaceutical manufacturing base.
Latin America Recombinant Protein Market Analysis
Latin America is demonstrating increasing potential as a growing market for recombinant proteins, driven by rising healthcare expenditures, expanding biopharmaceutical production capabilities, and the growing burden of chronic diseases such as cancer, diabetes, and cardiovascular conditions. Brazil and Mexico represent the region’s largest markets, leveraging investments in healthcare infrastructure and growing biotechnology sectors to expand the domestic production of biologics. Government initiatives across the region are supporting the adoption of biosimilars and recombinant protein-based therapeutics to improve treatment access and reduce healthcare costs.
Middle East and Africa Recombinant Protein Market Analysis
The Middle East and Africa region represents an emerging market for recombinant proteins, characterized by increasing government investments in healthcare infrastructure, growing pharmaceutical manufacturing capabilities, and the rising prevalence of chronic diseases including diabetes, cancer, and cardiovascular conditions. Gulf Cooperation Council countries, particularly Saudi Arabia and the United Arab Emirates, have been at the forefront of healthcare modernization initiatives, creating expanding opportunities for biopharmaceutical products including recombinant proteins. Saudi Arabia’s Vision 2030 initiative has catalyzed substantial investment in healthcare and biotechnology infrastructure, with the kingdom directing considerable resources toward life sciences research and biomanufacturing capabilities.
Competitive Landscape:
The global recombinant protein market is characterized by a moderately fragmented competitive landscape, with established multinationals and specialized niche providers actively competing across research-grade, clinical-grade, and commercial-grade recombinant protein segments. Market leaders are deploying diverse growth strategies, including product portfolio expansion, strategic licensing agreements, academic-industry collaborations, and investments in advanced expression system technologies, to maintain and strengthen their competitive positions. The integration of artificial intelligence into protein design and bioprocess optimization is increasingly being leveraged by leading players to differentiate their offerings and deliver faster innovation cycles. Contract development and manufacturing organizations are playing an increasingly important role in the ecosystem, enabling smaller biopharmaceutical companies to access recombinant protein production capabilities without significant capital investment. Mergers, acquisitions, and strategic partnerships between reagent providers, contract manufacturers, and biopharmaceutical developers are reshaping industry boundaries, while regulatory compliance and quality assurance remain critical differentiators as demand from clinical-stage drug developers and commercial manufacturers continues to intensify across major global markets.
The report provides a comprehensive analysis of the competitive landscape in the recombinant protein market with detailed profiles of all major companies, including:
- Abcam Limited
- ACROBiosystems
- Bio-RAD Laboratories Inc.
- Bio-Techne
- Bps Bioscience Inc.
- Enzo Biochem Inc.
- Genscript
- Prospec-Tany Technogene Ltd
- R&D Systems, Inc.
- RayBiotech, Inc.
- Sino Biological, Inc
- Thermo Fisher Scientific Inc
Latest News and Developments:
- March 2025: WuXi Biologics, a China-based contract research, development, and manufacturing organization (CRDMO), has unveiled its latest platform, EffiX. This exclusive technology employs an E. coli expression system aimed at maximizing the yield and stability of recombinant proteins and plasmid DNA production.
Recombinant Protein Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Products Covered | Antibodies, Hormones, Growth Factors, Cytokines, Others |
| Applications Covered | Drug Discovery and Development, Academic Research, Biotechnology Research, Biopharmaceutical Production, Others |
| End Users Covered | Pharmaceutical and Biopharmaceutical Companies, Biotechnology Companies, Academic and Research Institutes, Contract Research Organization, Others |
| Region Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Abcam Limited, ACROBiosystems, Bio-RAD Laboratories Inc., Bio-Techne, Bps Bioscience Inc., Enzo Biochem Inc., Genscript, Prospec-Tany Technogene Ltd, R&D Systems, Inc., RayBiotech, Inc., Sino Biological, Inc, Thermo Fisher Scientific Inc, etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the recombinant protein market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global recombinant protein market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's Five Forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the recombinant protein industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Key Questions Answered in This Report
The recombinant protein market was valued at USD 2.3 Billion in 2025.
The recombinant protein market is projected to exhibit a CAGR of 9.17% during 2026-2034, reaching a value of USD 5.04 Billion by 2034.
The rising prevalence of chronic diseases including cancer, diabetes, and autoimmune disorders is driving demand for recombinant protein-based biologics. Government investments in proteomics and genomics research, the growing shift toward biologic and biosimilar therapies, and advancements in protein engineering and expression technologies are collectively accelerating innovation and expanding the commercial applications of recombinant proteins globally.
North America currently dominates the recombinant protein market, accounting for a share of 40%. The region benefits from advanced biopharmaceutical infrastructure, substantial government funding in life sciences, a mature venture capital ecosystem, and strong regulatory frameworks that support the approval and commercialization of recombinant protein-based therapeutics.
Some of the major players in the recombinant protein market include Abcam Limited, ACROBiosystems, Bio-RAD Laboratories Inc., Bio-Techne, Bps Bioscience Inc., Enzo Biochem Inc., Genscript, Prospec-Tany Technogene Ltd, R&D Systems, Inc., RayBiotech, Inc., Sino Biological, Inc, Thermo Fisher Scientific Inc, etc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












