
Regulatory Affairs Outsourcing Market Report by Services (Regulatory Consulting, Legal Representation, Regulatory Writing and Publishing, Product Registration and Clinical Trial Applications, and Others), Company Size (Small, Medium, Large), Category (Generic Drugs, Innovator Drugs, Biologic Drugs, Biosimilars, Medical Devices, and Others), Indication (Oncology, Neurology, Cardiology, Immunology, and Others), Stage (Preclinical, Clinical, PMA (Post Market Authorization)), End User (Medical Device Companies, Pharmaceutical Companies, Biotechnology Companies), and Region 2026-2034
Market Overview:
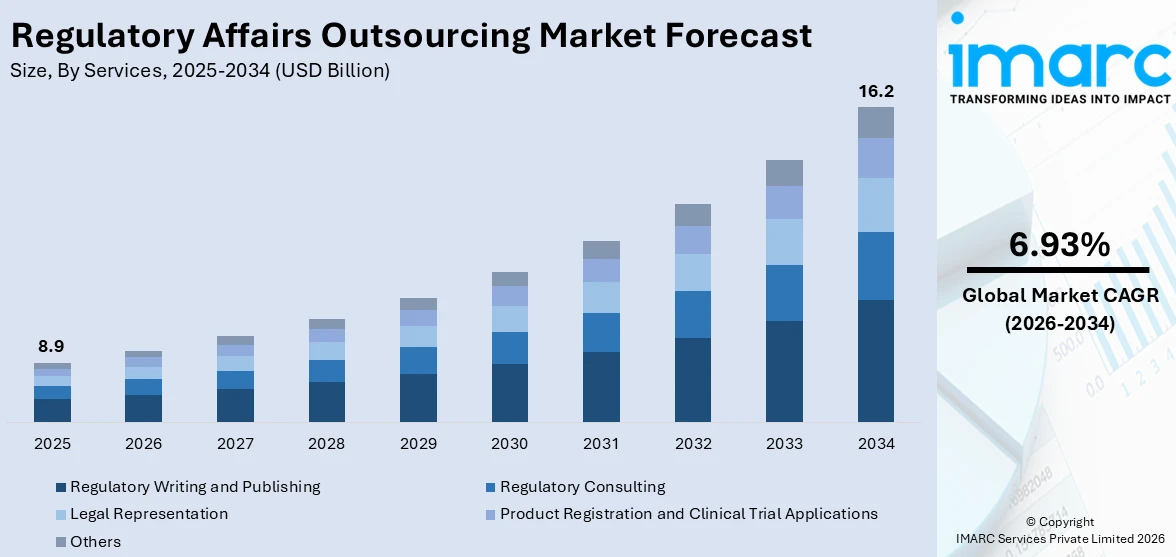
The global regulatory affairs outsourcing market size reached USD 8.9 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 16.2 Billion by 2034, exhibiting a growth rate (CAGR) of 6.93% during 2026-2034. The evolving and complex regulations, globalization of industries, resource optimization, ongoing technological advancements, specialized expertise requirements, and the need for rapid approvals are some of the major factors propelling the market.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 8.9 Billion |
|
Market Forecast in 2034
|
USD 16.2 Billion |
| Market Growth Rate (2026-2034) | 6.93% |
Regulatory affairs outsourcing is the practice of companies delegating their regulatory compliance and documentation tasks to external specialized service providers. In this arrangement, businesses, particularly those in highly regulated industries such as pharmaceuticals, biotechnology, and medical devices, hire third-party experts or firms to manage regulatory affairs on their behalf. This encompasses activities like product registration, quality assurance, compliance with government regulations, and submission of required documentation to regulatory authorities. Outsourcing these tasks allows companies to tap into the expertise of professionals well-versed in regulatory frameworks, streamline processes, reduce overhead costs, and ensure adherence to evolving regulatory standards, ultimately facilitating market entry and product approval.
To get more information on this market Request Sample
The global regulatory affairs outsourcing market is experiencing substantial growth primarily due to the increasingly complex and stringent regulatory requirements imposed by governments and regulatory bodies across various industries, including pharmaceuticals, medical devices, and food and beverages (F&B). Moreover, the expanding product adoption to enable companies to access a pool of professionals well-versed in the latest regulatory changes and compliance standards, thereby ensuring timely approvals and market access, is contributing to the market growth. Additionally, the globalization of markets has expanded the geographical reach of businesses, necessitating compliance with a multitude of international regulations, thereby bolstering the market growth. Furthermore, the growing need for outsourcing to provide a scalable solution to address diverse regulatory needs across regions is aiding in market expansion. Apart from this, the emerging trend toward cost-efficiency and operational flexibility drives companies to leverage external regulatory services, allowing them to focus on core competencies while mitigating overhead expenses, creating a positive outlook for market expansion.
Regulatory Affairs Outsourcing Market Trends/Drivers:
Increasing complexity in the regulatory landscape
One of the primary drivers of regulatory affairs outsourcing is the increasingly intricate and ever-evolving regulatory environment across various sectors. Industries such as pharmaceuticals, biotechnology, medical devices, and food and beverages (F&B) are subject to strict regulations to ensure product safety, efficacy, and compliance with quality standards. Keeping up with these regulations demands specialized knowledge and continuous monitoring. Outsourcing regulatory affairs allows companies to tap into the expertise of professionals who specialize in understanding the nuances of regional and international regulatory frameworks. Apart from this, these experts help businesses navigate the complex web of rules and ensure that their products meet compliance standards, ultimately facilitating market entry and product approval.
Expanding globalization of markets
Another pivotal factor fueling the growth of regulatory affairs outsourcing is the globalization of markets. As businesses expand their operations globally, they encounter a diverse array of regulatory requirements that vary by region and country. Complying with these regulations can be an intimidating task, especially for companies with limited in-house regulatory resources. In confluence with this, outsourcing provides a scalable solution to address these diverse regulatory needs efficiently. Regulatory affairs outsourcing firms often have a global presence and can assist businesses in adapting their products and processes to meet the specific requirements of different markets, thereby enabling market access and international growth.
Rising focus on cost-efficiency and operational productivity
Cost efficiency and the need for operational focus are driving companies to leverage external regulatory services. Maintaining an in-house regulatory affairs team involves substantial overhead costs, including salaries, training, and infrastructure. Outsourcing regulatory affairs allows organizations to reduce these fixed costs and gain flexibility in resource allocation. This approach enables companies to redirect resources towards core competencies such as research and development (R&D), marketing, and production. By entrusting regulatory activities to specialized outsourcing partners, businesses can streamline their operations, increase efficiency, and respond more effectively to market dynamics.
Regulatory Affairs Outsourcing Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global regulatory affairs outsourcing market report, along with forecasts at the global, regional and country levels from 2026-2034. Our report has categorized the market based on services, company size, category, indication, stage and end user.
Breakup by Services:
- Regulatory Consulting
- Legal Representation
- Regulatory Writing and Publishing
- Product Registration and Clinical Trial Applications
- Others
Regulatory writing and publishing dominate the market
The report has provided a detailed breakup and analysis of the market based on the services. This includes regulatory consulting, legal representation, regulatory writing and publishing, product registration and clinical trial applications, and others. According to the report, regulatory writing and publishing represented the largest segment.
The demand for regulatory writing and publishing outsourcing is driven by the growing complexity of regulatory documentation, which necessitates specialized expertise and resources. As regulatory requirements become more intricate and region-specific, businesses seek external partners with in-depth knowledge of document preparation and submission processes. Besides this, the need for rapid approvals and market access compels companies to outsource these critical tasks to ensure accurate, compliant, and timely submissions. Furthermore, the adoption of electronic submission systems and the digital transformation of regulatory processes create demand for outsourcing providers with the technological infrastructure and proficiency required for efficient electronic document preparation and publishing.
Breakup by Company Size:
- Small
- Medium
- Large
Medium holds the largest share of the market
A detailed breakup and analysis of the market based on the company size have also been provided in the report. This includes small, medium, and large. According to the report, medium accounted for the largest market share.
Another key factor driving the global regulatory affairs outsourcing market is the rising demand for specialized expertise and resource optimization in medium-sized companies. Medium-sized firms often face resource constraints and may lack an in-house regulatory affairs team with comprehensive knowledge of evolving regulations. Outsourcing enables them to access a pool of experienced regulatory professionals without the overhead costs of maintaining a full-time team. Additionally, as these companies expand into new markets, they require adaptable regulatory support to navigate diverse and complex regulatory landscapes, making outsourcing an efficient and strategic choice to ensure compliance, market entry, and growth.
Breakup by Category:
- Generic Drugs
- Innovator Drugs
- Biologic Drugs
- Biosimilars
- Medical Devices
- Therapeutic
- Diagnostic
- Others
Medical devices dominate the market
The report has provided a detailed breakup and analysis of the market based on the category. This includes generic drugs, innovator drugs, biologic drugs, biosimilars, medical devices (therapeutic and diagnostic), and others. According to the report, medical devices represented the largest segment.
The medical devices sector is subject to rigorous and evolving regulatory requirements globally, requiring specialized expertise to navigate. Outsourcing offers access to regulatory professionals who possess in-depth knowledge of medical device regulations, ensuring compliance and quicker approvals. Besides this, the constant innovation in medical device technology demands efficient and timely submissions to bring new products to market. Outsourcing streamlines regulatory processes, allowing companies to focus on R&D, thereby fueling the market's growth. Moreover, as the medical device market expands internationally, outsourced services provide support in adapting products to diverse regulatory environments, making outsourcing indispensable for market entry and sustained growth in this sector.
Breakup by Indication:
- Oncology
- Neurology
- Cardiology
- Immunology
- Others
Oncology holds the largest share of the market
A detailed breakup and analysis of the market based on the indication have also been provided in the report. This includes oncology, neurology, cardiology, immunology, and others. According to the report, oncology accounted for the largest market share.
The unique challenges and rapid advancements in this therapeutic area are impelling the demand for regulatory affairs outsourcing in the field of oncology. Oncological products often undergo accelerated development and require expedited regulatory pathways due to their potential life-saving nature. This demands specialized knowledge of oncology-specific regulations and fast-track approval processes, which outsourcing partners can provide. Furthermore, the increasing complexity of oncology clinical trials and the need for comprehensive data management and submissions necessitate outsourcing to experts who can navigate these intricate regulatory landscapes, ensuring timely approvals and market access for critical oncology treatments.
Breakup by Stage:
- Preclinical
- Clinical
- PMA (Post Market Authorization)
Clinical dominates the market
The report has provided a detailed breakup and analysis of the market based on the stage. This includes preclinical, clinical, and post market authorization (PMA). According to the report, clinical represented the largest segment.
The surging complexity and resource-intensive nature of clinical trials are contributing to the increasing demand for regulatory affairs outsourcing at the clinical stage. Companies engaging in clinical research must adhere to stringent regulatory requirements to ensure the safety, security, and efficacy of investigational products. Outsourcing offers access to regulatory professionals with specialized knowledge of clinical trial regulations and documentation, helping companies navigate this intricate landscape efficiently. Concurrently, the need for rapid study initiation and adherence to evolving regulatory standards drives the demand for outsourced support in protocol development, submissions, and compliance, ensuring that clinical trials progress smoothly and meet regulatory milestones effectively.
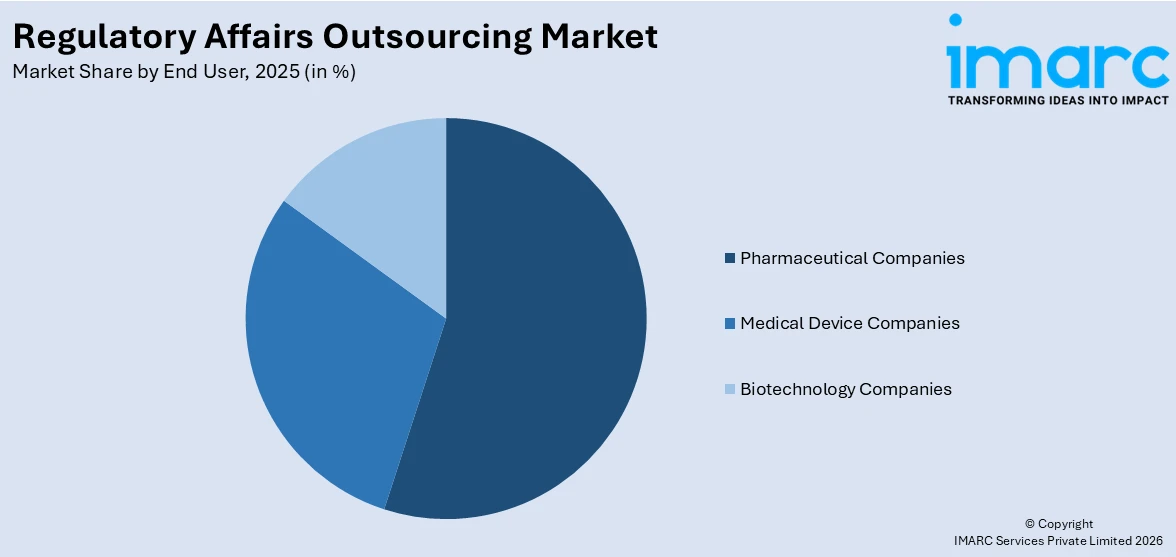
Breakup by End User:
Access the comprehensive market breakdown Request Sample
- Medical Device Companies
- Pharmaceutical Companies
- Biotechnology Companies
Pharmaceutical companies hold the largest share of the market
A detailed breakup and analysis of the market based on the end-user have also been provided in the report. This includes medical device, pharmaceutical, and biotechnology companies. According to the report, pharmaceutical companies accounted for the largest market share.
The pharmaceutical industry's evolving landscape represents the key factor aiding in market expansion. With an increasing emphasis on innovative drug development and stringent regulatory oversight, pharmaceutical firms require specialized expertise to navigate the complex regulatory pathways. Outsourcing provides access to regulatory professionals well-versed in pharmaceutical regulations, speeding up the approval process and ensuring compliance. Moreover, the demand for global market access and the need to adapt to regional regulatory variations make outsourcing an attractive option. It enables pharmaceutical companies to focus on core research and development activities while external partners handle regulatory affairs efficiently and effectively.
Breakup by Region:
- North America
- United States
- Canada
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
Asia-Pacific exhibits a clear dominance, accounting for the largest regulatory affairs outsourcing market share
The market research report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, Asia-Pacific accounted for the largest market share.
The increasing globalization of the pharmaceutical, healthcare, and life sciences sectors across the Asia Pacific has led to a surge in demand for regulatory services to navigate diverse regulatory landscapes across the region. Concurrent with this, the rising number of clinical trials and research activities in the region necessitates expertise in local regulatory requirements, making outsourcing an efficient solution for pharmaceutical and biotech companies looking to expand their presence. Furthermore, cost-efficiency plays a pivotal role as companies seek to optimize resources without compromising on compliance, making regulatory affairs outsourcing an attractive option in the Asia Pacific region.
Competitive Landscape:
The competitive landscape of the global regulatory affairs outsourcing market is characterized by a dynamic and diverse array of players vying for market share. Large multinational contract research organizations (CROs) hold prominent positions, offering comprehensive regulatory services across various industries and geographies. These industry giants leverage their extensive global networks, robust technology platforms, and deep regulatory expertise to cater to the diverse needs of clients. Additionally, niche and specialized regulatory affairs outsourcing firms focus on specific industries or regions, offering tailored solutions and in-depth regulatory knowledge. Emerging players, including startups and regional service providers, are also gaining traction by capitalizing on market niches and innovative service offerings. The competitive landscape is further shaped by strategic collaborations, mergers, and acquisitions as companies seek to expand their service portfolios and geographic reach.
The report has provided a comprehensive analysis of the competitive landscape in the market. Detailed profiles of all major companies have also been provided. Some of the key players in the market include:
- Charles River Laboratories International Inc.
- Covance Inc (Laboratory Corporation of America Holdings)
- Freyr
- ICON Plc
- Medpace Holdings Inc.
- Parexel International Corporation
- Pharmaceutical Product Development LLC
- Promedica International- A California Corporation
- Wuxi AppTec Co. Ltd.
Recent Developments:
- In February 2021, ICON plc announced the acquisition of PRA Health Sciences, Inc. in a cash and stock transaction valued at USD 12 billion approximately. This acquisition has strengthened the medical affairs service offering of the company.
Regulatory Affairs Outsourcing Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Predictive Market Assessment:
|
| Services Covered | Regulatory Consulting, Legal Representation, Regulatory Writing and Publishing, Product Registration and Clinical Trial Applications, Others |
| Company Sizes Covered | Small, Medium, Large |
| Categories Covered | Generic Drugs, Innovator Drugs, Biologic Drugs, Biosimilars, Medical Devices (Therapeutic and Diagnostic), Others |
| Indications Covered | Oncology, Neurology, Cardiology, Immunology, Others |
| Stages Covered | Preclinical, Clinical, PMA (Post Market Authorization) |
| End Users Covered | Medical Device Companies, Pharmaceutical Companies, Biotechnology Companies |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Charles River Laboratories International Inc., Covance Inc (Laboratory Corporation of America Holdings), Freyr, ICON Plc, Medpace Holdings Inc., Parexel International Corporation, Pharmaceutical Product Development LLC, Promedica International- A California Corporation, Wuxi AppTec Co. Ltd., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report:
- How has the global regulatory affairs outsourcing market performed so far and how will it perform in the coming years?
- What has been the impact of COVID-19 on the global regulatory affairs outsourcing market?
- What are the key regional markets?
- What is the breakup of the market based on the services?
- What is the breakup of the market based on the company size?
- What is the breakup of the market based on the category?
- What is the breakup of the market based on the indication?
- What is the breakup of the market based on the stage?
- What is the breakup of the market based on the end user?
- What are the various stages in the value chain of the industry?
- What are the key driving factors and challenges in the industry?
- What is the structure of the global regulatory affairs outsourcing market and who are the key players?
- What is the degree of competition in the industry?
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












