
Respiratory Syncytial Virus (RSV) Diagnostics Market Report by Product (Direct Fluorescent Antibody (DFA) Method, Rapid Antigen Diagnostic Test (RADTs), Molecular Diagnostics, Chromatographic Immunoassay, Diagnostic Imaging, Gel Microdroplets, Flow Cytometry, and Others), End Use (Hospitals and Clinics, Laboratory, and Others), and Region 2026-2034
Market Overview:
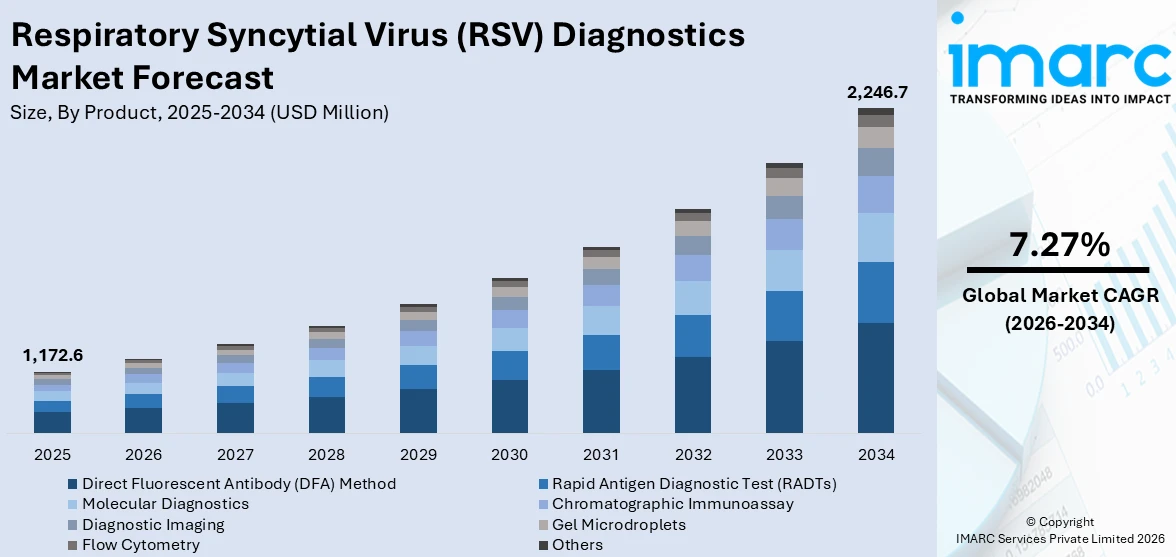
The global respiratory syncytial virus (RSV) diagnostics market size reached USD 1,172.6 Million in 2025. Looking forward, IMARC Group expects the market to reach USD 2,246.7 Million by 2034, exhibiting a growth rate (CAGR) of 7.27% during 2026-2034.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 1,172.6 Million |
|
Market Forecast in 2034
|
USD 2,246.7 Million |
| Market Growth Rate (2026-2034) | 7.27% |
Respiratory syncytial virus (RSV) is a viral infection of the lungs and respiratory tract, which can cause asthma, hospitalization, pneumonia, repeated infections, middle ear infection, and death. It can be diagnosed using blood tests, chest x-rays, a swab of secretions, and pulse oximetry by checking for signs like lung inflammation, white blood cell (WBC) count, and oxygen levels in the blood. Some of the commonly available treatment options for RSV are over-the-counter (OTC) medications, intravenous (IV) fluids, humidified oxygen, mechanical ventilation, and inhaled bronchodilators.
To get more information on this market Request Sample
Respiratory Syncytial Virus (RSV) Diagnostics Market Trends:
RSV infection can be more serious during the neonatal period as children under two years of age tend to develop severe respiratory symptoms. This, in confluence with the rising focus of parents on child medical care, represents one of the crucial factors fostering the growth of the market. Moreover, the high survival rates after early diagnosis and the easy availability of diagnostic modalities act as other growth-inducing factors. Apart from this, the improving healthcare infrastructure and favorable reimbursement policies for the treatment of hospitalized infants are also contributing to market growth. Furthermore, the advent of advanced techniques like bio- and nanotechnology is facilitating the development of modern detection systems for RSV, such as western blot, enzyme-linked immunosorbent assay (ELISA), direct immunofluorescence, and real-time polymerase chain reaction (PCR). Besides this, governing agencies of numerous countries are funding research and development (R&D) projects for the development of new therapeutics. Other factors projected to drive the market are the growing number of clinical vaccine trials, the escalating demand for in-vitro diagnostics, rising incidences of childhood pneumonia and the increasing adoption of molecular diagnostics.
Key Market Segmentation:
IMARC Group provides an analysis of the key trends in each sub-segment of the global respiratory syncytial virus (RSV) diagnostics market report, along with forecasts at the global, regional and country level from 2026-2034. Our report has categorized the market based on product and end use.
Breakup by Product:
- Direct Fluorescent Antibody (DFA) Method
- Rapid Antigen Diagnostic Test (RADTs)
- Molecular Diagnostics
- Chromatographic Immunoassay
- Diagnostic Imaging
- Gel Microdroplets
- Flow Cytometry
- Others
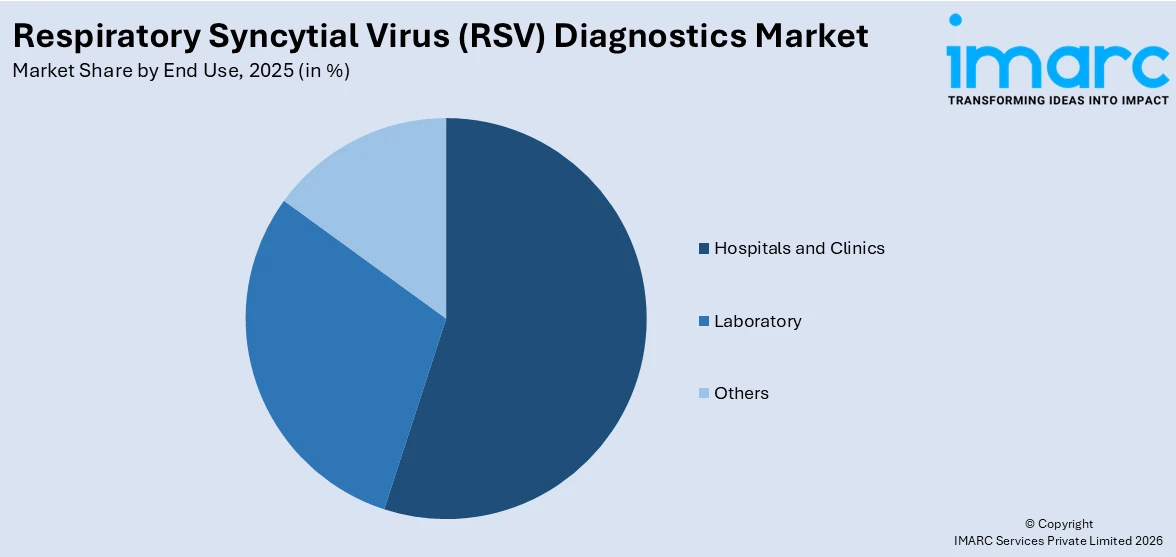
Breakup by End Use:
Access the comprehensive market breakdown Request Sample
- Hospitals and Clinics
- Laboratory
- Others
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
Competitive Landscape:
The competitive landscape of the industry has also been examined along with the profiles of the key players being Abbott Laboratories, Becton, Dickinson and Company, bioMérieux SA, Bio-Rad Laboratories Inc., Coris BioConcept, DiaSorin Molecular LLC (DiaSorin Inc), F. Hoffmann-La Roche AG, Merck KGaA, Quest Diagnostics Incorporated, Quidel Corporation and Thermo Fisher Scientific Inc.
Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Million USD |
| Segment Coverage | Product, End Use, Region |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Abbott Laboratories, Becton, Dickinson and Company, bioMérieux SA, Bio-Rad Laboratories Inc., Coris BioConcept, DiaSorin Molecular LLC (DiaSorin Inc), F. Hoffmann-La Roche AG, Merck KGaA, Quest Diagnostics Incorporated, Quidel Corporation and Thermo Fisher Scientific Inc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report:
- How has the global respiratory syncytial virus (RSV) diagnostics market performed so far and how will it perform in the coming years?
- What has been the impact of COVID-19 on the global respiratory syncytial virus (RSV) diagnostics market?
- What are the key regional markets?
- What is the breakup of the market based on the product?
- What is the breakup of the market based on the end use?
- What are the various stages in the value chain of the industry?
- What are the key driving factors and challenges in the industry?
- What is the structure of the global respiratory syncytial virus (RSV) diagnostics market and who are the key players?
- What is the degree of competition in the industry?
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












