
Small Molecule API Market Report by Type (Synthetic, Biotech), Manufacturing Method (In-House, Outsourced), Application (Ophthalmology, Cardiovascular, Metabolic, Dermatology, Respiratory, Urology, and Others), End User (Hospitals, Ambulatory Surgical Centers, and Others), and Region 2026-2034
Market Overview:
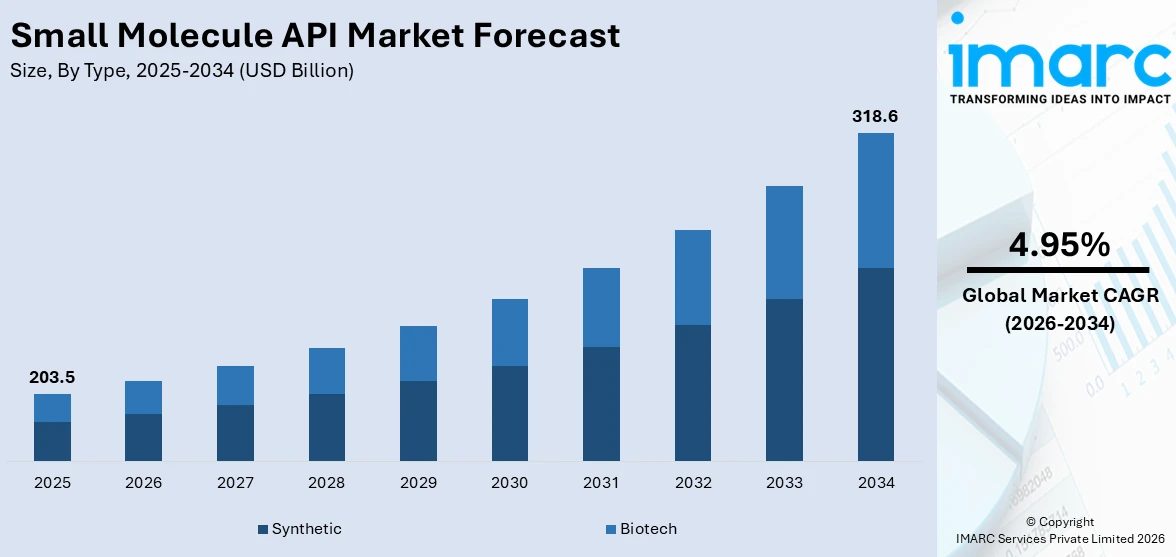
The global small molecule API market size reached USD 203.5 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 318.6 Billion by 2034, exhibiting a growth rate (CAGR) of 4.95% during 2026-2034. The increasing demand for small molecule drugs among the masses, the growing trend of outsourcing on the global level, and the widespread adoption of novel technologies represent some of the key factors driving the market.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025 |
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
| Market Size in 2025 | USD 203.5 Billion |
| Market Forecast in 2034 | USD 318.6 Billion |
| Market Growth Rate (2026-2034) | 4.95% |
A small molecule API, also known as a small molecule active pharmaceutical ingredient, refers to a chemically synthesized compound that serves as the primary active ingredient in a pharmaceutical drug. It is characterized by its relatively low molecular weight and ability to interact with specific biological targets to elicit a therapeutic effect. Due to their advantageous drug-like characteristics, including oral bioavailability, chemical stability, and effective production procedures, they are frequently employed in the pharmaceutical sector. These APIs are often developed through rational drug design or high-throughput screening methods, where scientists aim to identify compounds that can selectively bind to a target protein or receptor associated with a particular disease or medical condition. Additionally, it is designed to interact with the target in a specific manner, modulating its activity and thereby influencing the underlying disease processes. The chemical structure of a small molecule API plays a crucial role in determining its pharmacokinetic properties, including absorption, distribution, metabolism, and excretion.
To get more information on this market Request Sample
Significant growth in the pharmaceutical sector majorly drives the global market. Coupled with the rising prevalence of chronic diseases and the subsequent demand for effective treatments, this is providing an impetus to the demand. Also, with the number of individuals requiring medical intervention rises, there is a significant increase in the need for accessible and affordable medications, which is positively influencing the market. In addition, the regulatory landscape plays a vital role in shaping the small molecule API market. Stringent quality standards and regulatory requirements set by health authorities across the globe in order to ensure the safety and efficacy of pharmaceutical products, is driving the demand for small molecule API further. Some of the other factors driving the market include continual improvements in the healthcare sector and extensive research and development activities conducted by key players.
Small Molecule API Market Trends/Drivers:
Increasing Geriatric Population
The growing incidence of chronic diseases among geriatric population, such as cardiovascular disorders, cancer, diabetes, and respiratory diseases is driving the demand for small molecule APIs. These APIs are used as the active ingredients in pharmaceutical formulations to treat and manage these conditions, leading to a continuous need for their production. The global population is aging rapidly, resulting in a higher demand for healthcare services and medications. In line with this, older adults often require long-term medication therapy as they are more prone to medical ailments, which is significantly supporting the demand for small-molecule APIs as they are widely used in the production of medications to address age-related ailments.
Patent Expirations and Regulatory Support
With numerous drugs approaching patent expiry, this is opening up opportunities for the development and production of generic medications. Small molecule APIs play a crucial role in the production of generic drugs, as they offer cost-effective alternatives to brand-name medications. The expiration of patents encourages the entry of multiple manufacturers, leading to an increasing adoption of small molecule APIs. Also, regulatory bodies and other national regulatory agencies have implemented strict quality standards for drug manufacturing, which is impelling numerous key players to focus on compliance and quality assurance, thus creating a consistent demand for small molecule APIs across the globe.
Technological Advancements in API Synthesis
Continuous technological advancements in synthetic chemistry, process optimization, and manufacturing methods have significantly improved the efficiency and scalability of small molecule API synthesis. These advancements allow for streamlined manufacturing processes, reduced production costs, and increased yields. The availability of advanced synthesis techniques and technologies are supporting the growth of the small molecule API market. Additionally, continual advancements in drug delivery systems, such as oral solid dosage forms, inhalation devices, transdermal patches, and targeted drug delivery, are driving the demand for small molecule APIs further as these systems enhance patient compliance, improve drug efficacy, and reduce side effects. Also, small molecule APIs are essential for formulating these advanced drug delivery systems, which is propelling their demand on the global level.
Small Molecule API Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global small molecule API market report, along with forecasts at the global, regional, and country levels for 2026-2034. Our report has categorized the market based on type, manufacturing method, application, and end user.
Breakup by Type:
- Synthetic
- Biotech
Synthetic type represents the leading segment
The report has provided a detailed breakup and analysis of the small molecule API market based on the type. This includes synthetic and biotech. According to the report, synthetic type represented the largest segment.
Synthetic small molecule APIs offer advantages, such as consistent quality, scalability, and cost-effectiveness, making them highly desirable for pharmaceutical companies. With the increasing prevalence of chronic diseases and the expanding global population, there has been a significant rise in the demand for pharmaceutical products, thereby influencing the market for synthetic small molecule API. Additionally, the growing emphasis on personalized medicine and targeted therapies has necessitated the development of novel APIs that can be synthesized to meet specific therapeutic requirements.
Additionally, the biotech type is gaining immense traction across the globe due to the advancements in technology and scientific research, which enable the discovery and development of new small molecule APIs with improved efficacy, safety, and manufacturing processes. Furthermore, regulatory reforms and intellectual property protections play a crucial role by fostering innovation and providing market opportunities for biotech segment in the small molecule API industry.
Breakup by Manufacturing Method:
- In-house
- Outsourced
In-house dominates the small molecule API market
The report has provided a detailed breakup and analysis of the small molecule API market based on the manufacturing method. This includes in-house and outsourced. According to the report, in-house represented the largest segment.
The increasing regulatory scrutiny and the need for tighter quality control have compelled pharmaceutical companies to bring the manufacturing process in-house. By having direct control over production, companies can ensure adherence to strict quality standards and mitigate the risk of supply chain disruptions. Additionally, the rising demand for personalized medicine and niche therapeutic products requires flexible manufacturing capabilities that can be efficiently achieved through in-house operations, which is significantly supporting the in-house segment.
The small molecule active pharmaceutical ingredient (API) industry has witnessed a growing trend towards outsourced manufacturing methods, which is driven by cost reduction. This method allows companies to leverage lower labor and production costs in emerging markets, thereby influencing the market. Additionally, outsourcing provides access to specialized expertise and advanced technologies, enabling companies to tap into a broader pool of resources and knowledge. This allows for enhanced efficiency, quicker turnaround times, and improved quality control.
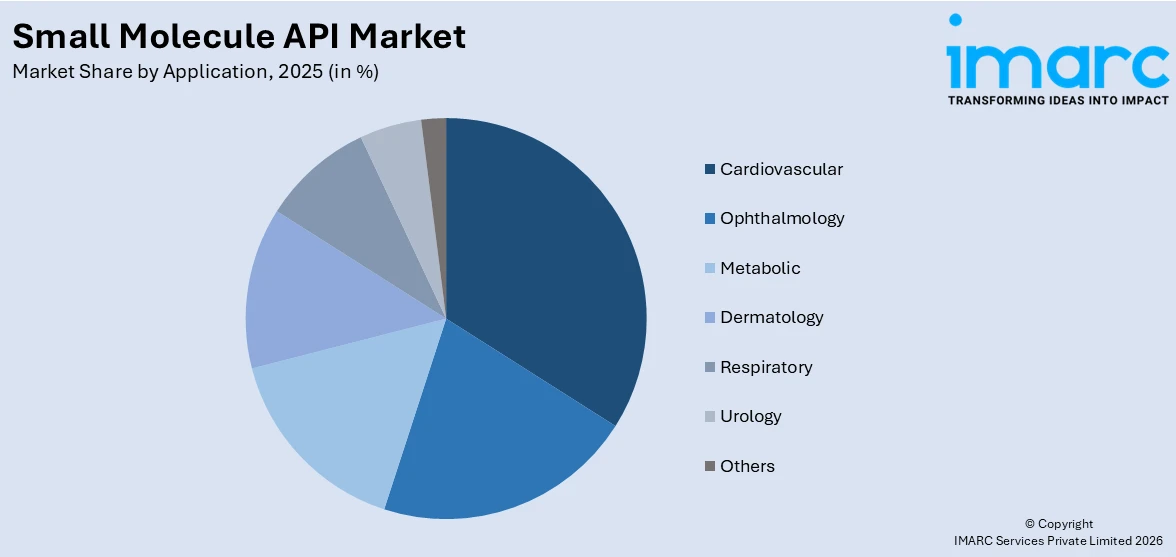
Breakup by Application:
Access the comprehensive market breakdown Request Sample
- Ophthalmology
- Cardiovascular
- Metabolic
- Dermatology
- Respiratory
- Urology
- Others
Cardiovascular accounts for the majority of the market share
The report has provided a detailed breakup and analysis of the small molecule API market based on the application. This includes ophthalmology, cardiovascular, metabolic, dermatology, respiratory, urology, and others. According to the report, cardiovascular represented the largest segment.
The small molecule active pharmaceutical ingredient (API) industry is experiencing significant growth due to the growing prevalence of cardiovascular diseases, including conditions, such as hypertension, hyperlipidemia, and heart failure. This is increasing the demand for effective medications to treat and manage these conditions, thus creating a positive market outlook for the market.
Technological advancements in drug delivery systems, such as improved ocular formulations and sustained-release mechanisms, have expanded the possibilities for small molecule API utilization in ophthalmology. Additionally, the aging population and increasing awareness of eye health have contributed to a higher patient pool seeking ophthalmic treatments, further propelling the demand for small molecule APIs.
In addition, the rising focus on personalized medicine and the need for tailored therapies for individual patients is further propelling the demand for metabolic applications in the small molecule API industry. Furthermore, the potential for substantial cost savings and improved treatment outcomes using small molecule APIs in metabolic applications is also a significant driving factor.
Breakup by End User:
- Hospitals
- Ambulatory Surgical Centers
- Others
Hospitals hold the largest market share
The report has provided a detailed breakup and analysis of the small molecule API market based on the end user. This includes hospitals, ambulatory surgical centers, and others. According to the report, hospitals represented the largest segment.
The stringent regulatory requirements and quality standards imposed on pharmaceutical products necessitate hospitals to source APIs from reliable and compliant manufacturers, which is driving the market. Also, the availability of specialized formulations and tailored drug development services for hospital settings is fueling the adoption of small molecule APIs in this segment.
The ambulatory surgical centers (ASCs) end-user segment plays a crucial role in driving the demand for small molecule active pharmaceutical ingredients (APIs). Continual advancements in medical technology and surgical techniques enable a broader range of procedures to be performed in ASCs, attracting patients seeking convenient and cost-effective healthcare solutions. In addition, favorable reimbursement policies and regulatory frameworks further supporting the segment.
Breakup by Region:
- North America
- United States
- Canada
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Others
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America exhibits a clear dominance, accounting for the largest small molecule API market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (Germany, France, the United Kingdom, Italy, Spain, and others); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa.
The small molecule API industry in North America is driven by the growing demand for effective pharmaceutical treatments. Small molecule APIs, with their ability to target specific disease pathways, offer significant therapeutic potential in addressing these conditions. Additionally, the region's well-established pharmaceutical infrastructure, including advanced research facilities, regulatory frameworks, and a skilled workforce, is contributing to the growth of the industry. Moreover, the rising investment in research and development activities by pharmaceutical companies and the focus on innovative drug discovery is propelling the demand for small molecule APIs.
The presence of a skilled workforce, cost-effective manufacturing capabilities, and favorable regulatory policies in countries, including India and China has attracted multinational pharmaceutical companies to establish production facilities in the Asia Pacific region, further boosting the small molecule API market. Furthermore, increasing investment in research and development activities, technological advancements, and a strong focus on quality standards are driving the innovation and production of small molecule APIs in the Asia Pacific.
Competitive Landscape:
The global small molecule API market is experiencing growth across the globe with the ongoing development of personalized medicine and targeted therapies necessitating specialized small molecule APIs that can address specific disease mechanisms or genetic profiles. This presents an opportunity for top companies to leverage their expertise in custom synthesis and manufacturing capabilities to cater to niche therapeutic areas and develop tailored solutions for patients. Additionally, the rising focus on generic drugs and the expiration of numerous blockbuster drug patents offer companies the chance to capitalize on the development and production of cost-effective generic small molecule APIs. Furthermore, the growing emphasis on contract manufacturing and outsourcing in the pharmaceutical industry provides a favorable environment for API manufacturers, as pharmaceutical companies seek to streamline their operations, reduce costs, and focus on their core competencies of drug formulation and marketing.
The report has provided a comprehensive analysis of the competitive landscape in the global small molecule API market. Detailed profiles of all major companies have also been provided. Some of the key players in the market include:
- AstraZeneca plc
- Baxter International Inc.
- Cambrex Corporation
- Dr. Reddy’s Laboratories Ltd.
- Lonza Group AG
- Pfizer Inc.
- Sun Pharmaceutical Industries Limited
- Teva Pharmaceutical Industries Ltd.
Recent Developments:
- In October 2022, Pfizer Inc. announced news regarding the completion of its acquisition of Biohaven Pharmaceutical Holding Company Ltd. With the acquisition, Pfizer gained access to a promising portfolio of CGRP receptor antagonists.
- In June 2022, Dr Reddy's Laboratories announced its subsidiary has partnered with US-based Olema Pharmaceuticals Inc to research, develop and commercialize novel small molecule inhibitors of a target.
- In December 2022, Lonza Group AG announced the beginning of new mid-scale manufacturing facilities at its Nansha, China, active pharmaceutical ingredients (API) manufacturing facility.
Small Molecule API Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical and Forecast Trends, Industry Catalysts and Challenges, Segment-Wise Historical and Predictive Market Assessment:
|
| Types Covered | Synthetic, Biotech |
| Manufacturing Methods Covered | In-House, Outsourced |
| Applications Covered | Ophthalmology, Cardiovascular, Metabolic, Dermatology, Respiratory, Urology, Others |
| End Users Covered | Hospitals, Ambulatory Surgical Centers, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | AstraZeneca plc, Baxter International Inc., Cambrex Corporation, Dr. Reddy’s Laboratories Ltd., Lonza Group AG, Pfizer Inc., Sun Pharmaceutical Industries Limited, Teva Pharmaceutical Industries Ltd., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report:
- How has the global small molecule API market performed so far, and how will it perform in the coming years?
- What are the drivers, restraints, and opportunities in the global small molecule API market?
- What is the impact of each driver, restraint, and opportunity on the global small molecule API market?
- What are the key regional markets?
- Which countries represent the most attractive small molecule API market?
- What is the breakup of the market based on the type?
- Which is the most attractive type in the small molecule API market?
- What is the breakup of the market based on the manufacturing method?
- Which is the most attractive manufacturing method in the small molecule API market?
- What is the breakup of the market based on the application?
- Which is the most attractive application in the small molecule API market?
- What is the breakup of the market based on the end user?
- Which is the most attractive end user in the small molecule API market?
- What is the competitive structure of the global small molecule API market?
- Who are the key players/companies in the global small molecule API market?
Key Benefits for Stakeholders:
- IMARC’s report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the small molecule API market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global small molecule API market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assist stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the small molecule API industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












