
Tenosynovial Giant Cell Tumor Treatment Market Size, Share, Trends and Forecast by Disease Type, Treatment Type, End-User, and Region, 2026-2034
Tenosynovial Giant Cell Tumor Treatment Market Size and Share:
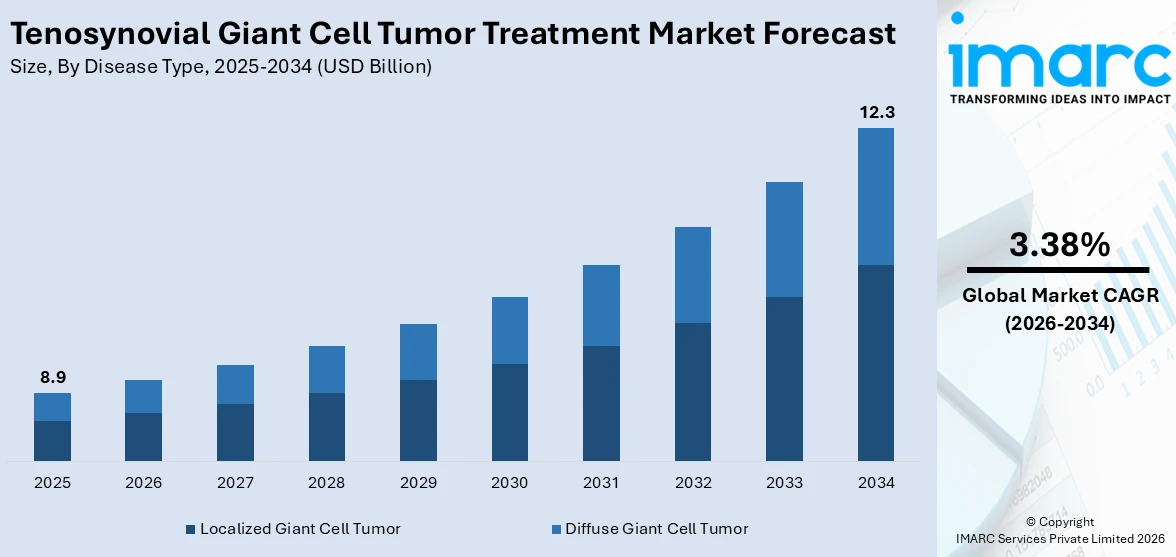
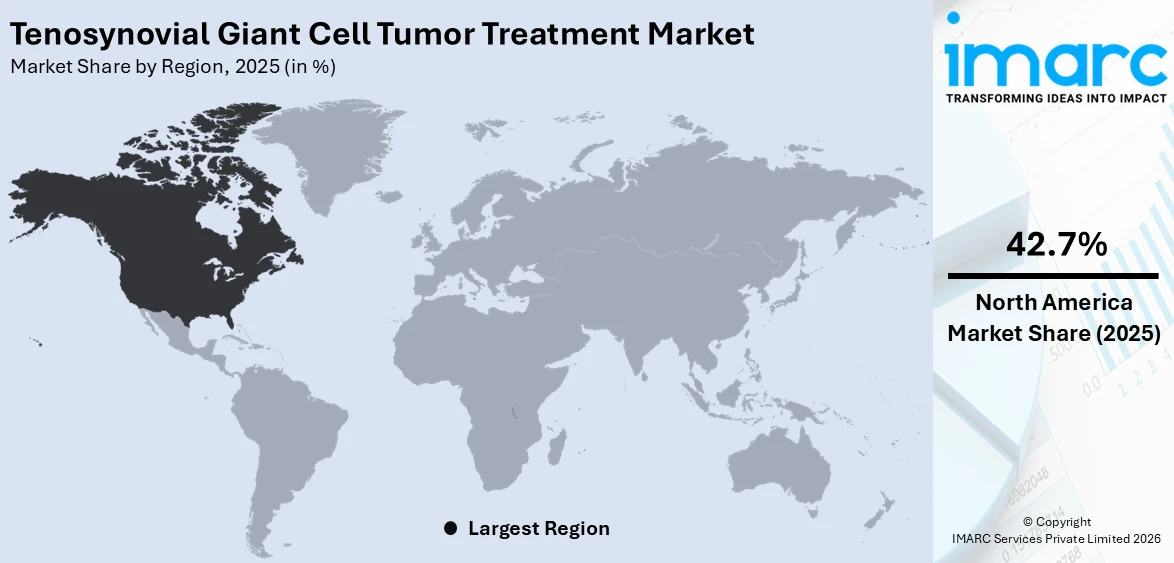
The global tenosynovial giant cell tumor treatment market size was valued at USD 8.9 Billion in 2025. Looking forward, IMARC Group estimates the market to reach USD 12.3 Billion by 2034, exhibiting a CAGR of 3.38% from 2026-2034. North America currently dominates the market, holding a market share of 42.7% in 2025. The region benefits from advanced healthcare infrastructure, well-established reimbursement frameworks, rising prevalence of musculoskeletal neoplasms, and growing adoption of targeted systemic therapies for rare conditions, all contributing to the tenosynovial giant cell tumor treatment market share.
The global tenosynovial giant cell tumor treatment market is experiencing steady expansion driven by multiple factors. Increasing clinical awareness of tenosynovial giant cell tumor among healthcare professionals is facilitating earlier and more accurate diagnoses, thereby expanding the addressable patient population. Moreover, significant advancements in understanding the molecular pathogenesis of TGCT, particularly the role of colony stimulating factor 1 receptor overexpression, have catalyzed the development of novel targeted systemic therapies that complement traditional surgical approaches. The rising demand for minimally invasive surgical procedures and arthroscopic interventions further supports market expansion by improving patient outcomes and reducing recovery periods. Additionally, growing healthcare expenditures across developed and developing economies are enhancing access to specialized oncology services. The tenosynovial giant cell tumor treatment market outlook remains favorable as expanding clinical trial pipelines, regulatory approvals for new pharmacological agents, and increasing collaborations between pharmaceutical companies and academic research institutions collectively drive sustained growth.
The United States has emerged as a major region in the tenosynovial giant cell tumor treatment market owing to many factors. The country possesses a highly developed healthcare system with specialized sarcoma and orthopedic oncology centers that offer comprehensive TGCT management. Robust insurance coverage and favorable reimbursement policies enable widespread adoption of both surgical interventions and novel pharmacotherapies. Additionally, the presence of leading pharmaceutical companies engaged in active research and development of colony stimulating factor 1 receptor inhibitors accelerates therapeutic innovation within the country. Furthermore, increasing patient advocacy efforts and growing awareness campaigns are promoting earlier detection and timely treatment initiation, strengthening the market growth across the country. For instance, in April 2025, Emactuzumab was granted Fast Track designation by the US FDA for the treatment of tenosynovial giant cell tumor (TGCT), supporting its accelerated development in an area with limited therapeutic options. The Phase 3 TANGENT study is assessing emactuzumab, a monoclonal antibody that targets and inhibits CSF-1R, in patients with TGCT.
To get more information on this market Request Sample
Tenosynovial Giant Cell Tumor Treatment Market Trends:
Growing Adoption of Multidisciplinary Care
The shift toward multidisciplinary team-based management of tenosynovial giant cell tumor is gaining significant momentum across healthcare systems worldwide. This collaborative approach integrates the expertise of orthopedic surgeons, medical oncologists, radiation oncologists, radiologists, and physical therapists to develop individualized treatment plans that optimize patient outcomes. Multidisciplinary tumor boards facilitate comprehensive case evaluation, enabling clinicians to determine the most appropriate combination of surgery, systemic therapy, radiation, and rehabilitative care based on disease subtype, tumor location, and recurrence risk. This approach is particularly valuable for patients with diffuse TGCT affecting complex anatomical locations where surgical outcomes may be suboptimal. The increasing emphasis on patient-reported outcomes and quality of life considerations within the tenosynovial giant cell tumor treatment market forecast further drives adoption of coordinated care models.
Rising Awareness and Improved Diagnosis
Increasing awareness of tenosynovial giant cell tumor among clinicians and patients is significantly improving diagnostic accuracy and reducing the time from symptom onset to definitive treatment. Enhanced understanding of TGCT clinical presentations, combined with advances in magnetic resonance imaging technology, enables more precise identification and characterization of both localized and diffuse tumor subtypes. Patient advocacy organizations and international research registries play a crucial role in disseminating educational resources and promoting referral to specialized sarcoma centers. For instance, a 2025 analysis of the TGCT Support Patient Registry encompassing 497 patients from 32 countries revealed that approximately 50 percent of patients were initially misdiagnosed prior to receiving their TGCT diagnosis. The tenosynovial giant cell tumor treatment market trends reflect growing investment in diagnostic infrastructure and clinician education programs aimed at reducing diagnostic delays. Additionally, expanding access to advanced imaging modalities across developing healthcare systems is broadening the identified patient population, creating new opportunities for timely intervention and improved long-term outcomes.
Advancement of CSF1R Targeted Therapies
The development and regulatory approval of colony stimulating factor 1 receptor inhibitors represent a transformative shift in the treatment landscape for tenosynovial giant cell tumor. These targeted systemic therapies address the underlying molecular driver of TGCT by blocking the CSF1/CSF1R signaling pathway responsible for abnormal inflammatory cell recruitment and tumor proliferation. The emergence of these pharmacological agents offers a crucial alternative for patients with inoperable, recurrent, or advanced disease who face limited surgical options. Clinical trials have demonstrated significant objective response rates and meaningful improvements in joint function, pain reduction, and overall quality of life. The growing pipeline of selective CSF1R inhibitors and ongoing long-term safety studies continue to expand treatment possibilities and reshape clinical practice guidelines for TGCT management globally.
Tenosynovial Giant Cell Tumor Treatment Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global tenosynovial giant cell tumor treatment market, along with forecast at the global, regional, and country levels from 2026-2034. The market has been categorized based on disease type, treatment type, and end-user.
Analysis by Disease Type:
- Localized Giant Cell Tumor
- Diffuse Giant Cell Tumor
Localized giant cell tumor holds 60.3% of the market share. Localized tenosynovial giant cell tumor, also known as nodular TGCT, is a well-circumscribed neoplasm that typically develops in smaller joints such as the fingers, hands, wrists, and toes. This subtype is characterized by slower growth patterns and lower recurrence rates compared with its diffuse counterpart, making complete surgical excision a highly effective treatment approach. The predominance of localized TGCT is attributed to its higher overall incidence rate, well-defined tumor margins, and favorable surgical outcomes that encourage timely clinical intervention. Additionally, the ease of detection through standard imaging techniques and routine orthopedic examinations contributes to a larger diagnosed patient population. For instance, a 2025 study in the Netherlands reported that the incidence rate for localized TGCT of extremities was 11 per million person-years compared with 5 to 8.4 per million for diffuse disease. Continued improvements in minimally invasive surgical approaches and the growing availability of arthroscopic procedures further reinforce the dominance of the localized segment.
Analysis by Treatment Type:
- Surgery
- Radiation Therapy
- Drug Therapy
Surgery leads the market with a share of 68.4%. Surgical intervention remains the primary and most widely employed treatment modality for tenosynovial giant cell tumors, encompassing procedures such as open synovectomy, arthroscopic resection, and total joint replacement in severe cases. The prevalence of surgery is led by the fact that it leads to instant tumor resection, quick alleviation of symptoms, and conclusive histologic diagnosis. Localized TGCT can be cured with high rates and relatively low morbidity with complete excision, and it is the standard of first-line therapy. Surgery tends to be the first line of therapy treatment, even in situations of diffuse disease, with subsequent introduction of systemic therapies to patients who have unresectable or recurrent tumors. Innovations in surgical and arthroscopic methods, as well as enhanced intra-surgery imaging-guided methods, will keep improving the accuracy of the procedures and decrease the postoperative complications, which will keep the segments on top.
Analysis by End-User:
Access the comprehensive market breakdown Request Sample
- Surgical Clinics
- Hospitals
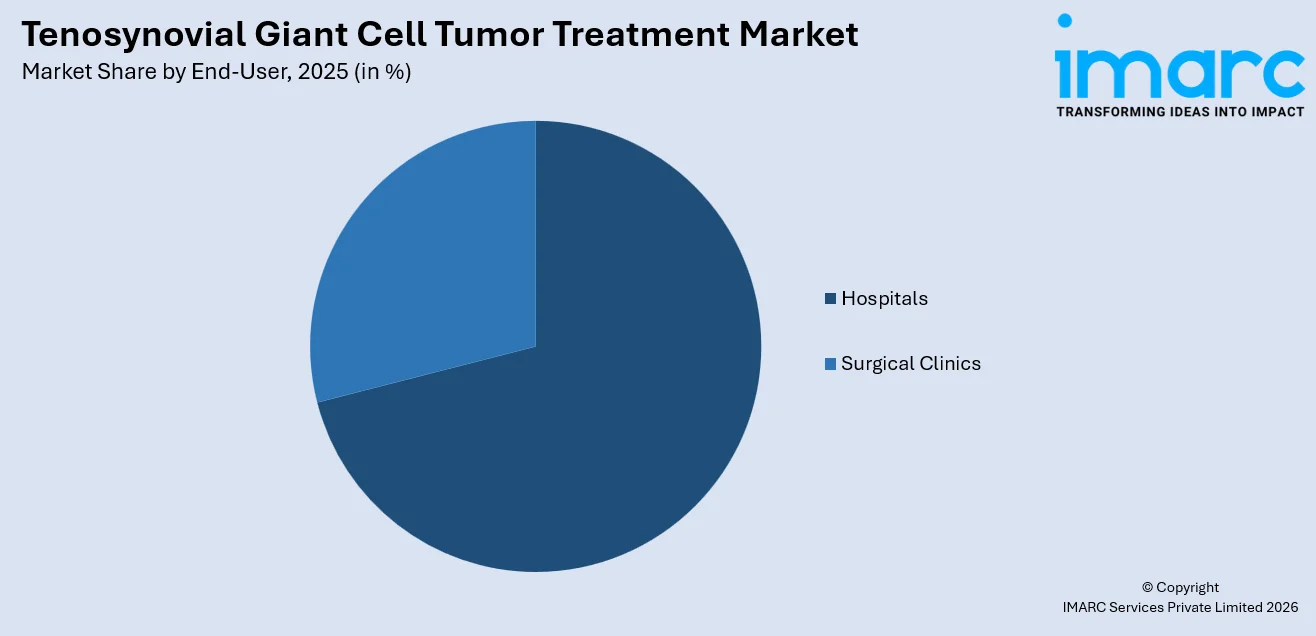
Hospitals dominate the market, with a share of 71.2%. Hospitals serve as the primary treatment setting for tenosynovial giant cell tumors due to their comprehensive infrastructure, multidisciplinary specialist teams, and access to advanced surgical facilities and diagnostic equipment. TGCT often necessitates complicated surgical operations, such as arthroscopic and total joint replacements, which demand specialized operating rooms, anesthesiology services, and postoperative monitoring, which are mostly available in hospitals. Moreover, hospitals have an oncology department that is integrated, and medical oncologists can administer systemic measures like colony-stimulating factor 1 receptor inhibitors under well-supervised protocols. The availability of advanced magnetic resonance imaging systems within hospital radiology departments enables precise preoperative planning and posttreatment surveillance, thereby driving the tenosynovial giant cell tumor treatment market growth. Additionally, the growing trend toward multidisciplinary tumor boards and specialized sarcoma centers within hospital networks further consolidates their leading position.
Regional Analysis:
To get more information on the regional analysis of this market Request Sample
- North America
- United States
- Canada
- Europe
- Germany
- France
- Spain
- United Kingdom
- Italy
- Netherlands
- Others
- Asia Pacific
- Japan
- India
- China
- Others
- Latin America
- Mexico
- Brazil
- Others
- Middle East and Africa
- South Africa
- Saudi Arabia
- Others
North America, accounting for 42.7% of the share, enjoys the leading position in the market. The region benefits from a well-established healthcare infrastructure, high rates of diagnostic awareness among clinicians, and early adoption of novel therapeutic modalities including targeted systemic therapies. North America serves as the primary market for approved colony stimulating factor 1 receptor inhibitors, with both pexidartinib and vimseltinib commercially available for patients with symptomatic TGCT not amenable to surgery. The presence of leading academic medical centers and specialized sarcoma referral networks ensures comprehensive patient evaluation and access to cutting-edge treatment options. For instance, in November 2024, the American Joint Replacement Registry analyzed 4.3 million hip and knee arthroplasties highlighting the extensive orthopedic surgical capacity available within the region. Robust insurance coverage, supportive reimbursement frameworks for orphan drugs, and active clinical trial participation further strengthen the regional market position. Growing investment in rare disease research and the expanding pipeline of investigational therapies continue to reinforce North America as the dominant market.
Key Regional Takeaways:
United States Tenosynovial Giant Cell Tumor Treatment Market Analysis
The United States represents the largest individual market for tenosynovial giant cell tumor treatment globally, supported by its advanced healthcare system, extensive network of specialized orthopedic oncology centers, and strong pharmaceutical innovation ecosystem. Two approved colony-stimulating factor 1 receptor inhibitors are now available, making clinicians have various systemic treatment options to use with patients with inoperable or recurrent disease. The mature reimbursement environment of both commercial coverage and government initiatives enables wide access for patients to expensive targeted treatments and surgical procedures. Moreover, the United States is also a center of clinical research with several trials currently underway to test next-generation CSF1R inhibitors and combination regimens. For instance, in November 2024, Daiichi Sankyo presented data from seven abstracts highlighting new clinical findings and real-world evidence on TURALIO (pexidartinib) at the 2024 Annual Meeting of the Connective Tissue Oncology Society (#CTOS2024). The presentations reflected ongoing research efforts and provided deeper insights into the therapy’s application in clinical practice. The strong presence of patient advocacy organizations actively promotes early detection and appropriate referral pathways. Additionally, increasing healthcare expenditure and growing focus on precision medicine approaches for rare musculoskeletal conditions continue to drive sustained demand for advanced TGCT treatment options across the country.
Europe Tenosynovial Giant Cell Tumor Treatment Market Analysis
Europe represents a significant market for tenosynovial giant cell tumor treatment, driven by well-developed healthcare systems, specialized sarcoma treatment networks, and strong regulatory support for orphan medicinal products. Pharmaceutical development of therapeutics against rare diseases is encouraged by the European Medicines Agency giving a simplified centralized approval procedure and a maximum of 10 years market exclusivity of the designated orphan drug. Multiple European nations have specific networks of sarcoma referral and multidisciplinary tumor boards that guarantee uniform and elaborate TGCT management. For instance, in July 2025, the Committee for Medicinal Products for Human Use of the European Medicines Agency issued a positive opinion recommending the approval of vimseltinib for adult patients with symptomatic TGCT. The growing clinical trial network in Germany, France, the United Kingdom, and the Netherlands also contributes to the development of the research environment in the region. The rising interest in the TGCT among orthopedic surgeons and oncologists in Europe, along with the rising use of advanced magnetic resonance imaging to assess diagnostic evaluation, both serve as contributors to the enlargement of the identified population of patients and facilitate the expansion of the market.
Asia-Pacific Tenosynovial Giant Cell Tumor Treatment Market Analysis
The Asia-Pacific region represents a rapidly growing market for tenosynovial giant cell tumor treatment, supported by expanding healthcare infrastructure, increasing government investments in oncology services, and rising clinical awareness of rare musculoskeletal conditions. Countries such as Japan, China, and India are experiencing growing demand for advanced surgical procedures and targeted pharmacotherapies as healthcare systems modernize and specialist capabilities expand. For instance, in January 2026, Abbisko Therapeutics Co., Ltd. announced that the US Food and Drug Administration (FDA) had formally accepted its New Drug Application for pimicotinib (ABSK021). Pimicotinib is an orally administered, highly selective and potent small-molecule inhibitor targeting the colony-stimulating factor 1 receptor (CSF-1R), developed for the systemic treatment of patients with tenosynovial giant cell tumor (TGCT). The growing participation of Asian clinical trial sites in global TGCT studies is enhancing regional access to investigational therapies. Additionally, increasing medical tourism flows and the establishment of specialized orthopedic oncology departments within major hospital networks are broadening treatment availability across the region.
Latin America Tenosynovial Giant Cell Tumor Treatment Market Analysis
Latin America is witnessing a gradual expansion in the tenosynovial giant cell tumor treatment market as healthcare systems across the region continue to develop specialized oncology capabilities. Countries such as Brazil and Mexico are investing in the modernization of hospital infrastructure and expanding access to advanced surgical and pharmaceutical interventions. Governments are strengthening rare disease programs to enhance diagnostic capabilities, streamline treatment pathways, and improve access to specialized care for complex conditions, including musculoskeletal neoplasms. Growing collaboration between regional medical institutions and international sarcoma research networks is enhancing clinician education and promoting adherence to evidence-based TGCT management protocols across the region.
Middle East and Africa Tenosynovial Giant Cell Tumor Treatment Market Analysis
The Middle East and Africa region is experiencing emerging growth in the tenosynovial giant cell tumor treatment market, primarily driven by ongoing healthcare modernization initiatives and increasing investment in specialized medical services. Countries such as Saudi Arabia and South Africa are developing advanced orthopedic and oncology treatment facilities that enhance local capacity for managing complex musculoskeletal conditions. For instance, in 2025, Saudi Arabia continued expanding its healthcare infrastructure under Vision 2030, investing in specialized medical centers equipped with advanced surgical and diagnostic technologies. The growing engagement of regional healthcare providers with international clinical guidelines and the expanding availability of imported pharmaceutical products are gradually improving treatment options for TGCT patients.
Competitive Landscape:
The competitive landscape of the tenosynovial giant cell tumor treatment market is defined by active research and development efforts, strategic collaborations, and ongoing regulatory progress among leading pharmaceutical and biopharmaceutical companies. Market participants are concentrating on the development and commercialization of novel colony stimulating factor 1 receptor inhibitors designed to deliver enhanced efficacy and improved safety profiles compared with existing therapies. The introduction of systemic treatment options has transformed the therapeutic approach to symptomatic TGCT and broadened pharmacological alternatives. Companies are further investing in long-term safety evaluations and real-world evidence generation to strengthen clinical confidence and adoption. Meanwhile, several investigational agents continue to advance through late-stage development, intensifying competition and driving innovation within the market.
The report provides a comprehensive analysis of the competitive landscape in the tenosynovial giant cell tumor treatment market with detailed profiles of all major companies, including:
- Daiichi Sankyo Company Limited
- Deciphera Pharmaceuticals Inc.
- Five Prime Therapeutics Inc.
- Novartis AG
Latest News and Developments:
- In January 2026, Merck announced that the US Food and Drug Administration (FDA) had accepted its New Drug Application for pimicotinib as a systemic therapy for individuals with tenosynovial giant cell tumor (TGCT). The submission is supported by primary findings and extended follow-up data from the global Phase 3 MANEUVER trial, which showed significant and sustained tumor reduction along with notable improvements in patient clinical outcomes.
- In February 2025, Ono Pharmaceutical Co., Ltd. received U.S. FDA approval for vimseltinib, a treatment for tenosynovial giant cell tumor (TGCT), which will be commercialized under the brand name ROMVIMZA. With this authorization, Ono enters direct competition in the TGCT segment with fellow Japanese drugmaker Daiichi Sankyo, intensifying rivalry within this therapeutic market.
Tenosynovial Giant Cell Tumor Treatment Market Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Hstorical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Disease Types Covered | Localized Giant Cell Tumor, Diffuse Giant Cell Tumor |
| Treatment Types Covered | Surgery, Radiation Therapy, Drug Therapy |
| End-Users Covered | Surgical Clinics, Hospitals |
| Region Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, Spain, United Kingdom, Italy, Netherlands, Japan, India, China, Mexico, Brazil, South Africa, Saudi Arabia |
| Companies Covered | Daiichi Sankyo Company Limited, Deciphera Pharmaceuticals Inc., Five Prime Therapeutics Inc., Novartis AG, etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC's report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the tenosynovial giant cell tumor treatment market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global tenosynovial giant cell tumor treatment market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's Five Forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the tenosynovial giant cell tumor treatment industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Frequently Asked Questions About the Tenosynovial Giant Cell Tumor Treatment Market Report
The tenosynovial giant cell tumor treatment market was valued at USD 8.9 Billion in 2025.
The tenosynovial giant cell tumor treatment market is projected to exhibit a CAGR of 3.38% during 2026-2034, reaching a value of USD 12.3 Billion by 2034.
The key factors driving the market include increasing prevalence of tenosynovial giant cell tumor, rising clinical awareness and improved diagnostic capabilities, development and regulatory approval of novel colony stimulating factor 1 receptor inhibitors, growing adoption of multidisciplinary treatment approaches, expanding healthcare infrastructure, and increasing investment in rare disease research and therapeutic innovation.
North America currently dominates the tenosynovial giant cell tumor treatment market, accounting for a share of 42.7%. The region benefits from advanced healthcare infrastructure, strong pharmaceutical innovation, early adoption of targeted therapies, and comprehensive reimbursement frameworks.
Some of the major players in the tenosynovial giant cell tumor treatment market include Daiichi Sankyo Company Limited, Deciphera Pharmaceuticals Inc., Five Prime Therapeutics Inc., and Novartis AG, etc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












