
United States Shingles Vaccine Market Size, Share, Trends and Forecast by Product, Vaccine Type, End Use, and Region, 2026-2034
United States Shingles Vaccine Market Summary:
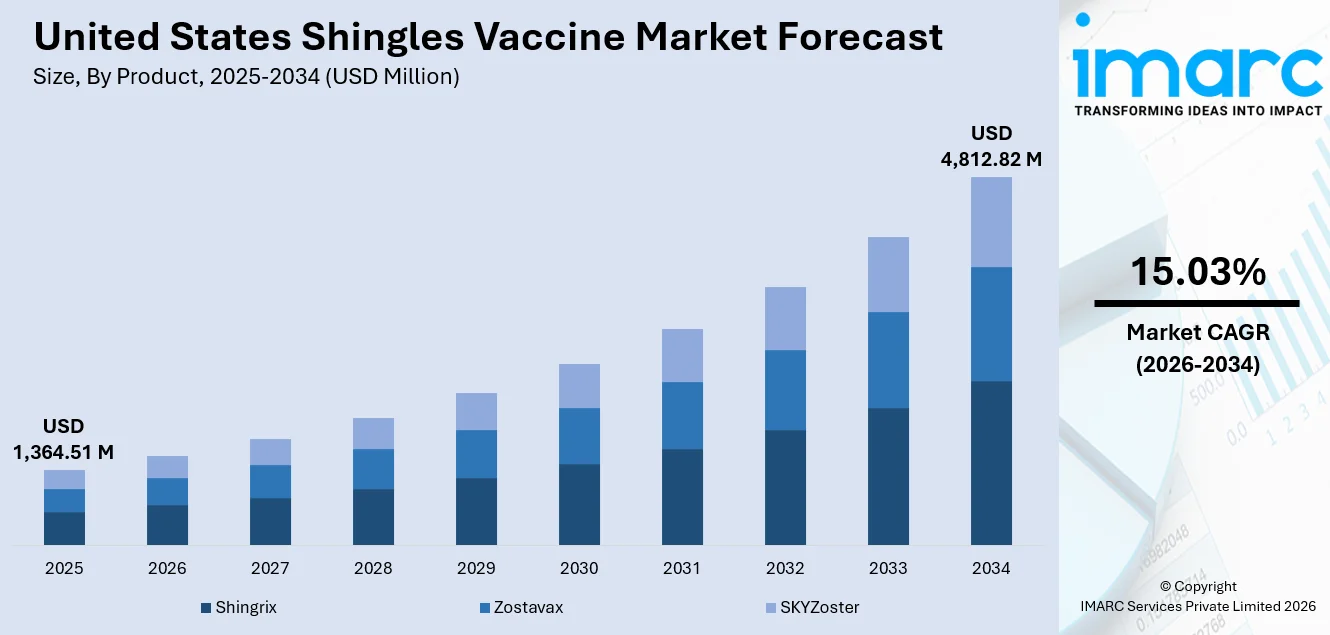
The United States shingles vaccine market size was valued at USD 1,364.51 Million in 2025 and is projected to reach USD 4,812.82 Million by 2034, growing at a compound annual growth rate of 15.03% from 2026-2034.
The United States shingles vaccine market is experiencing robust expansion driven by the rapidly aging population, strengthened federal immunization recommendations, and transformative legislative reforms that have eliminated cost barriers for Medicare beneficiaries. Increasing recognition of shingles-related complications among immunocompromised adults, growing adoption of recombinant vaccine technologies, and expanding clinical evidence linking shingles vaccination to broader neuroprotective benefits are collectively reshaping preventive healthcare paradigms and strengthening the United States shingles vaccine market share.
Key Takeaways and Insights:
- By Product: Shingrix dominates the market with a share of 79.4% in 2025, owing to its superior efficacy as a recombinant adjuvanted formulation that delivers sustained protection against herpes zoster and postherpetic neuralgia across adult age groups.
- By Vaccine Type: Recombinant vaccine leads the market with a share of 82.1% in 2025. This dominance is driven by the demonstrated immunological advantages of non-live subunit vaccine technology, which provides high seroconversion rates and broader eligibility including immunocompromised individuals.
- By End Use: Private healthcare settings represent the largest segment with a market share of 55.8% in 2025, reflecting strong consumer preference for convenient vaccination access through retail pharmacies, specialty clinics, and corporate wellness programs that offer streamlined scheduling and reduced wait times.
- By Region: South comprises the largest region with 34.5% share in 2025, driven by the concentration of retiree populations in metropolitan areas across Florida, Texas, and the Carolinas, combined with expanding pharmacy-based vaccination networks serving aging communities.
- Key Players: The market is highly concentrated, dominated by a leading recombinant vaccine with strong physician uptake and broad pharmacy distribution. Competition centers on pricing, payer coverage, supply reliability, and adult immunization outreach. New entrants must prove durable efficacy, safety in older adults, and convenient dosing.
To get more information on this market Request Sample
The United States shingles vaccine market is advancing as federal policy reforms, expanded clinical recommendations, and growing awareness of herpes zoster complications converge to accelerate vaccination uptake across eligible adult populations. The Inflation Reduction Act, which took effect in January 2023, eliminated out-of-pocket costs for all Advisory Committee on Immunization Practices-recommended vaccines under Medicare Part D, directly removing financial barriers for the largest at-risk demographic. Following this reform, shingles vaccine administration among Medicare Part D beneficiaries increased by of 26.3 additional vaccinations per 10,000 Medicare Part D enrollees. Median out-of-pocket costs dropped from USD 45.00 before the change to USD 0 afterward. Moreover, ongoing investments in manufacturing infrastructure, emerging clinical evidence suggesting neuroprotective benefits of shingles vaccination, and the development of next-generation candidates with improved tolerability profiles are collectively positioning the market for sustained long-term growth across urban and rural healthcare delivery settings throughout the country.
United States Shingles Vaccine Market Trends:
Expanding immunization coverage
The United States shingles vaccine market is undergoing a major shift following the implementation of the Inflation Reduction Act, which removed out-of-pocket costs for ACIP-recommended vaccines under Medicare Part D. This policy change has significantly reduced financial barriers for older adults, who represent the primary target population for shingles immunization. By improving affordability and access, the reform has encouraged higher vaccination rates, particularly among previously under-immunized groups. Increased uptake is strengthening market penetration and supporting sustained growth across pharmacies, clinics, and healthcare systems nationwide.
Emerging therapeutic applications beyond herpes zoster prevention
Emerging research suggests that shingles vaccination may offer benefits beyond preventing herpes zoster and its complications. Studies are exploring potential links between vaccination and reduced risks of certain cardiovascular events, stroke, and other inflammatory conditions, highlighting broader protective effects. A study found shingles vaccination reduced overall cardiac events by 23% and major cardiac events by 26%, with protective effects lasting up to eight years post-immunization. These findings are strengthening the clinical and economic value proposition of shingles immunization programs. As evidence continues to build, healthcare providers and policymakers may view shingles vaccination not only as infection prevention, but also as part of a wider preventive health strategy.
Next-generation vaccine development with improved tolerability profiles
The competitive landscape is evolving as pharmaceutical companies advance novel shingles vaccine candidates designed to address the reactogenicity limitations of current market offerings. As such, in August 2025, Dynavax Technologies announced positive Phase 1/2 Part 1 results for its Z-1018 shingles vaccine candidate, which demonstrated comparable immunogenicity to Shingrix while achieving markedly lower rates of adverse events, with only 12.5% local grade 2/3 reactions compared to 52.6% for the current market leader. These developments signal a shift toward better-tolerated formulations.
Market Outlook 2026-2034:
The United States shingles vaccine market is positioned for sustained expansion over the forecast period, underpinned by favorable demographic shifts, strengthened federal immunization policies, and an increasingly competitive product pipeline. The U.S. population aged 65 and older is projected to reach 82 million by 2050, representing approximately 23% of the total population, which will significantly expand the addressable patient pool. Continued elimination of cost-sharing under the Inflation Reduction Act is expected to drive higher vaccination rates among Medicare beneficiaries, while expanded ACIP recommendations for immunocompromised adults aged 19 and above are broadening eligibility criteria. Advancing clinical trials for next-generation candidates with improved tolerability, coupled with ongoing research into potential neuroprotective benefits, are anticipated to further strengthen the case for widespread shingles vaccination and accelerate market growth. The market generated a revenue of USD 1,364.51 Million in 2025 and is projected to reach a revenue of USD 4,812.82 Million by 2034, growing at a compound annual growth rate of 15.03% from 2026-2034.
United States Shingles Vaccine Market Report Segmentation:
|
Segment Category |
Leading Segment |
Market Share |
|
Product |
Shingrix |
79.4% |
|
Vaccine Type |
Recombinant Vaccine |
82.1% |
|
End Use |
Private Healthcare Settings |
55.8% |
|
Region |
South |
34.5% |
Product Insights:
- Shingrix
- Zostavax
- SKYZoster
Shingrix dominates with a market share of 79.4% of the total United States shingles vaccine market in 2025.
Shingrix has established itself as the definitive standard of care for shingles prevention in the United States, commanding the largest revenue share through its clinically superior recombinant adjuvanted formulation that delivers high efficacy across diverse adult populations. The vaccine's demonstrated ability to prevent herpes zoster and postherpetic neuralgia has earned broad endorsement from the Centers for Disease Control and Prevention and the Advisory Committee on Immunization Practices for adults aged 50 and older. Accordingly, in 2024, ACIP reaffirmed its recommendation of Shingrix as a two-dose regimen for adults aged 50 and above while extending the recommendation to immunocompromised adults aged 19 and older, further solidifying its clinical authority and market positioning.
The commercial strength of Shingrix is further reinforced by its expanding global footprint and consistent revenue generation for its manufacturer. GSK reported Shingrix sales of GBP 945 Million in the first quarter of 2024 alone, representing an 18% year-over-year increase that underscores the sustained demand trajectory across both domestic and international markets. The vaccine's inclusion in Medicare Part D coverage without cost-sharing following the Inflation Reduction Act has significantly enhanced accessibility for the largest at-risk demographic, while its favorable risk-benefit profile compared to the discontinued live attenuated Zostavax continues to drive physician preference and patient acceptance across private and public healthcare delivery channels.
Vaccine Type Insights:
- Recombinant Vaccine
- Live Attenuated Vaccine
Recombinant vaccine leads with a share of 82.1% of the total United States shingles vaccine market in 2025.
The United States shingles vaccine market now relies on recombinant vaccine technology because it provides better immunological protection than live attenuated vaccine options. The recombinant subunit vaccines for vaccines use specific viral glycoprotein antigens together with their proprietary adjuvant systems to generate strong humoral and cell-mediated immune responses, which do not require live viral replication, thus eliminating health risks for people who have weak immune systems.
The recombinant shingles vaccine segment is gaining further momentum as manufacturers advance next-generation candidates built on non-live vaccine platforms. The technologies developed through these systems achieve their goal of maintaining effective immunogenicity while developing better safety and tolerability results which protect elderly people and patients with weakened immune systems. The research team conducts their work through ongoing research projects and their clinical development initiatives which aim to improve both adjuvant system performance and accompanying dosing methods that help patients accept their treatment. The recombinant platform will maintain its importance to drive market growth and create unique product advantages because innovation will continue to develop new solutions.
End Use Insights:
Access the comprehensive market breakdown Request Sample
- Private Healthcare Settings
- Government Healthcare Settings
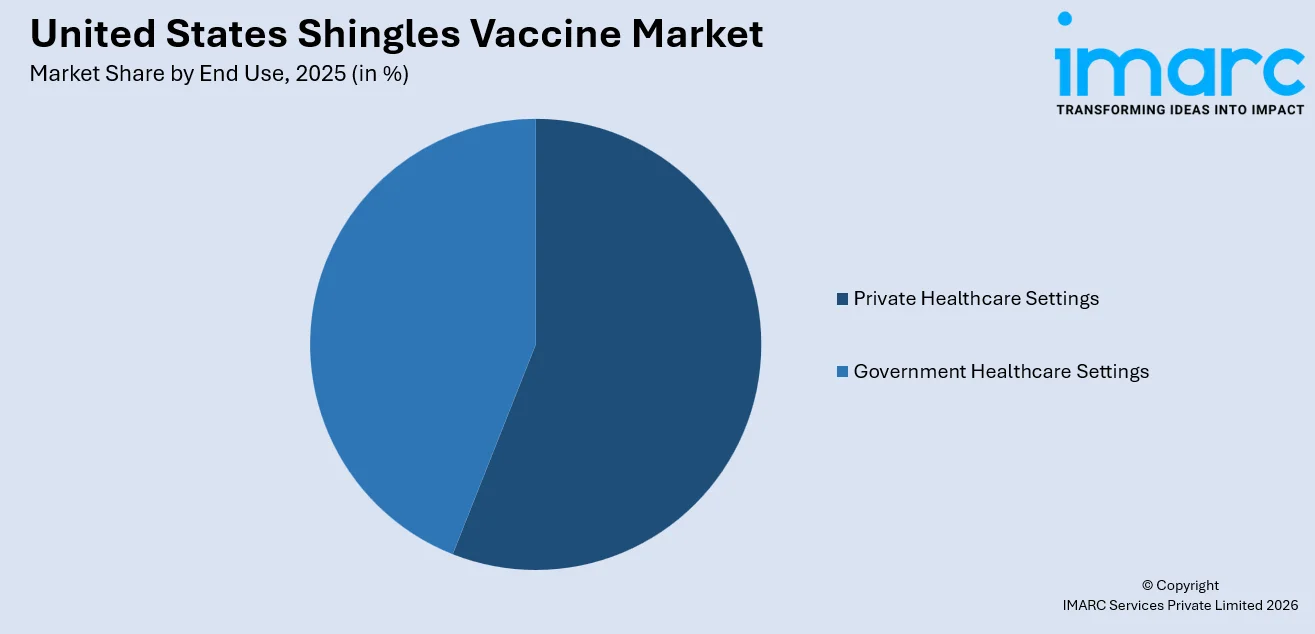
The private healthcare settings segment exhibits a clear dominance with a 55.8% share of the total United States shingles vaccine market in 2025.
Private healthcare settings have emerged as the primary channel for shingles vaccine administration in the United States, driven by the convenience and accessibility offered through retail pharmacies, specialty clinics, and physician offices that facilitate streamlined vaccination workflows. The segment benefits from shorter wait times, extended operating hours, and the growing integration of immunization services into routine pharmacy visits, which align with consumer preferences for efficient healthcare interactions. Approximately 95% of Medicare Part D beneficiaries receive their shingles vaccinations at pharmacy settings, underscoring the critical role of private retail infrastructure in driving vaccination uptake across the eligible adult population.
The segment's continued strength is reinforced by expanding corporate wellness programs, employer-sponsored immunization campaigns, and the growing role of private health insurance networks in promoting preventive care among working-age adults. The elimination of Medicare Part D cost-sharing under the Inflation Reduction Act reduced the average out-of-pocket cost from approximately USD 77 per vaccination to zero for covered beneficiaries, removing a significant financial deterrent that previously suppressed demand in private pharmacy channels.
Regional Insights:
- Northeast
- Midwest
- South
- West
South represents the leading segment with a 34.5% share of the total United States shingles vaccine market in 2025.
The United States shingles vaccine market reaches its highest total in the South region because this area contains multiple retirement communities and fast-growing urban centers which have high numbers of senior citizens. Florida, which reported approximately 22% of its population aged 65 and older in 2024, serves as a primary demand center within the region, while Texas and the Carolinas contribute through expanding healthcare infrastructure and growing retiree migration. The region benefits from extensive pharmacy-based vaccination networks, with major retail chains operating dense distribution footprints across suburban and urban communities.
The South experienced the highest population increase among all U.S. regions because multiple metropolitan areas showed substantial growth in their elderly population during recent times. The growing demographic expansion leads to more people who have increased susceptibility to shingles, which creates a larger group of people who need vaccination programs. The region's vaccine demand increases because older adults constitute the main demographic that medical professionals recommend for shingles vaccination.
Market Dynamics:
Growth Drivers:
Why is the United States Shingles Vaccine Market Growing?
Rapidly aging U.S. population expanding the addressable patient pool
The United States is undergoing a pronounced demographic shift as its elderly population continues to expand, directly strengthening demand for shingles vaccination. In 2024, adults aged 65 and older reached 61.2 million, accounting for 18% of the total population. This steady aging trend is enlarging the core immunization target group, particularly in states known for large retiree communities. As longevity improves and the share of older adults rises, healthcare systems are preparing for sustained growth in shingles vaccination uptake across pharmacies, primary care networks, and public health programs nationwide.
Rising Clinical Emphasis on Preventing Severe Complications
Healthcare providers are placing greater emphasis on shingles vaccination as awareness grows around the severity of its complications. Research found that most PCPs recognize shingles vaccination importance and age-related risk. About 82% identified key risk factors, 44% knew all approved vaccine indications, and only 29% reported full awareness of current ACIP recommendations. While shingles itself can be painful, its most concerning outcome is postherpetic neuralgia, a persistent nerve pain condition that can last months or even years and significantly reduce quality of life. Older adults and immunocompromised individuals face higher risks of prolonged discomfort and secondary health issues. As clinical guidelines continue to reinforce early vaccination, physicians and pharmacists are proactively recommending immunization during routine visits. This shift toward preventive care is driving stronger vaccine uptake and reinforcing long-term market growth.
Enhanced Medicare coverage under the Inflation Reduction Act
The implementation of the Inflation Reduction Act in January 2023 significantly improved shingles vaccine accessibility by eliminating out-of-pocket costs for ACIP-recommended adult vaccines under Medicare Part D. Before this reform, cost-sharing requirements often discouraged older adults on fixed incomes from receiving vaccination, despite strong clinical recommendations. By removing financial barriers, the policy has supported broader uptake across Medicare beneficiaries and expanded coverage among previously under-immunized groups. The resulting increase in vaccination rates highlights the strong relationship between affordability and preventive healthcare participation. This shift is strengthening market penetration, improving public health outcomes, and reinforcing sustained growth in shingles immunization services across pharmacies, clinics, and healthcare systems nationwide.
Market Restraints:
What Challenges the United States Shingles Vaccine Market is Facing?
Suboptimal Vaccination Rates Despite Strong Clinical Recommendations
Despite clear recommendations from national health authorities, shingles vaccination coverage in the United States remains below desired public health goals. Many eligible adults either delay initiation or fail to complete the required two-dose schedule. Gaps in patient awareness, inconsistent provider recommendations, and logistical challenges in returning for the second dose continue to limit full immunization. These barriers highlight the need for stronger education, reminder systems, and coordinated follow-up strategies to improve completion rates.
Geographic and Racial Disparities in Vaccine Access
The U.S. shingles vaccine market continues to face structural disparities that affect equitable coverage across regions and demographic groups. Vaccination rates vary widely between states and communities, reflecting uneven healthcare access and outreach. Racial and ethnic minorities often experience lower immunization uptake due to differences in healthcare access, trust, awareness, and provider engagement. Addressing these inequities requires targeted community education, improved access to vaccination sites, and culturally sensitive public health initiatives.
Reactogenicity Concerns Limiting Uptake
The leading shingles vaccine is known to cause temporary but noticeable side effects, which can discourage some patients from initiating or completing the two-dose series. Reports of injection-site pain, fatigue, and other systemic reactions contribute to hesitancy, particularly among older adults concerned about tolerability. While these reactions are generally short-lived and indicate immune response, they can influence patient perception and adherence. Improved counseling and future formulations with enhanced tolerability may help increase acceptance and completion rates.
Competitive Landscape:
The United States shingles vaccine market is characterized by a highly concentrated competitive structure with a single dominant player controlling the overwhelming majority of revenue share through its clinically superior recombinant formulation. Competition is evolving as emerging pharmaceutical companies advance novel vaccine candidates through clinical development, targeting improved tolerability and differentiated adjuvant systems to challenge established market positioning. Strategic investments in domestic manufacturing capacity, expanding distribution partnerships with pharmacy chains, and ongoing research into potential secondary health benefits are shaping competitive dynamics. The anticipated entry of next-generation recombinant candidates is expected to intensify competition and drive innovation in formulation science, pricing strategies, and patient access programs across the market.
Recent Developments:
- In January 2026, A USC observational study published suggests shingles vaccination may slow biological aging in adults over 70. Vaccinated individuals showed lower inflammation, slower epigenetic aging, and improved immune markers compared with unvaccinated peers. Effects were strongest within three years post-vaccination, supporting broader healthy-aging benefits beyond infection prevention.
- In July 2025, the U.S. FDA approved GSK’s Shingrix in a new prefilled syringe format, eliminating the need to reconstitute separate vials before administration. The updated presentation simplifies delivery for healthcare professionals while maintaining clinical comparability. Shingrix remains indicated for adults aged 50 and older and certain immunocompromised individuals.
United States Shingles Vaccine Market Report Coverage:
|
Report Features |
Details |
|
Base Year of the Analysis |
2025 |
|
Historical Period |
2020-2025 |
|
Forecast Period |
2026-2034 |
|
Units |
Million USD |
|
Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
|
Products Covered |
Shingrix, Zostavax, SKYZoster |
|
Vaccine Types Covered |
Recombinant Vaccine, Live Attenuated Vaccine |
|
End Uses Covered |
Private Healthcare Settings, Government Healthcare Settings |
|
Regions Covered |
Northeast, Midwest, South, West |
|
Customization Scope |
10% Free Customization |
|
Post-Sale Analyst Support |
10-12 Weeks |
|
Delivery Format |
PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Frequently Asked Questions About the United States Shingles Vaccine Market Report
The United States shingles vaccine market size was valued at USD 1,364.51 Million in 2025.
The United States shingles vaccine market is expected to grow at a compound annual growth rate of 15.03% from 2026-2034 to reach USD 4,812.82 Million by 2034.
Shingrix dominated the market with a share of 79.4%, driven by its superior recombinant adjuvanted formulation that delivers high efficacy and sustained protection endorsed for elderly population.
Key factors driving the United States shingles vaccine market include the rapidly aging population, elimination of Medicare Part D cost-sharing under the Inflation Reduction Act, expanded ACIP recommendations for immunocompromised adults, and growing awareness of shingles-related complications.
Major challenges include suboptimal vaccination rates despite strong clinical recommendations, significant geographic and racial disparities in vaccine access, high reactogenicity of the current market leader, and persistent gaps in two-dose series completion rates.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












