
Vaccine Contract Manufacturing Market Size, Share, Trends and Forecast by Vaccine Type, Workflow, Application, and Region, 2026-2034
Vaccine Contract Manufacturing Market Size and Share:
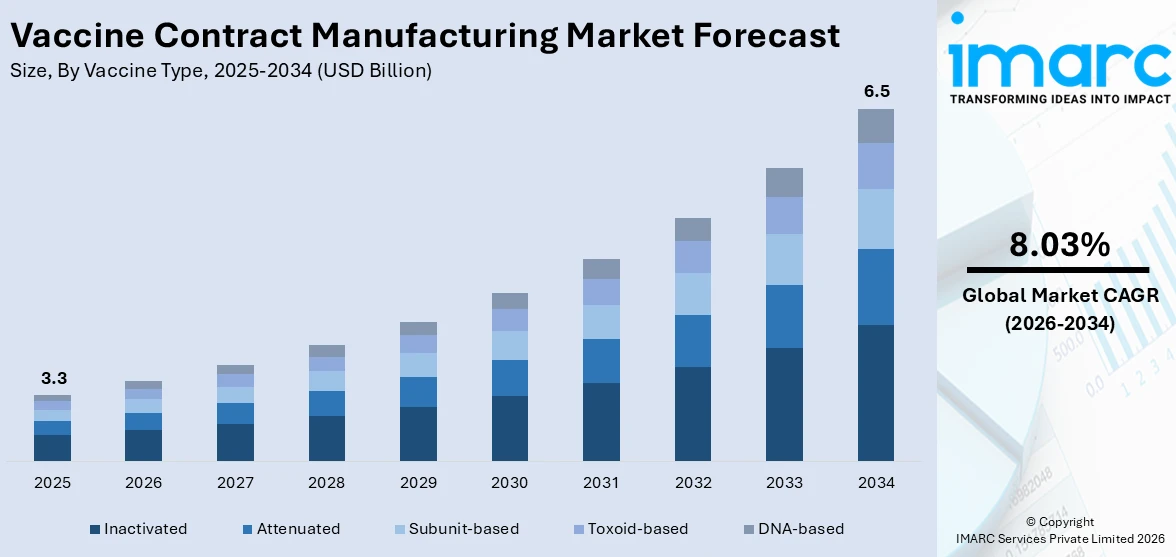
The global vaccine contract manufacturing market size was valued at USD 3.3 Billion in 2025. The market is projected to reach USD 6.5 Billion by 2034, exhibiting a CAGR of 8.03% from 2026-2034. North America currently dominates the market, holding a market share of 36% in 2025. Governments worldwide are prioritizing immunization as a key public health strategy, offering grants, subsidies, and procurement commitments to ensure timely access to vaccines. Besides this, the expansion of contract manufacturing organizations (CMOs) is propelling the vaccine contract manufacturing market share.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 3.3 Billion |
|
Market Forecast in 2034
|
USD 6.5 Billion |
| Market Growth Rate (2026-2034) | 8.03% |
The market is being propelled by rising global demand for vaccines, driven by increasing infectious disease prevalence, greater focus on preventive healthcare, and preparedness for future pandemics. High research and production costs often discourage pharmaceutical and biotech firms from investing in large-scale facilities, leading them to outsource to contract manufacturers with established expertise and regulatory compliance capabilities. The expanding pipeline of advanced vaccines, including messenger ribonucleic acid (mRNA) and viral vector platforms, requires specialized technologies that CMOs are better positioned to provide. Additionally, the need for flexible and scalable production capacity, combined with the pressure to reduce time-to-market, is making outsourcing a strategic choice.
To get more information on this market Request Sample
The United States has emerged as a major region in the vaccine contract manufacturing market owing to many factors. Rising focus on public health, immunization programs, and preparedness against emerging infectious diseases is propelling the vaccine contract manufacturing market growth. High research and development (R&D) and production costs, along with stringent regulatory requirements, are encouraging pharmaceutical and biotech firms to collaborate with experienced contract manufacturers that offer compliance expertise, advanced facilities, and cost efficiency. The country’s growing pipeline of biologics, including mRNA and viral vector vaccines, requires specialized technologies and flexible manufacturing capacities that many firms prefer to outsource. As per the IMARC Group, the United States biologics market is set to attain USD 371.0 Billion by 2033, exhibiting a CAGR of 7.4% from 2025-2033.
Vaccine Contract Manufacturing Market Trends:
Increasing awareness about vaccination
The growing awareness about vaccination is offering a favorable vaccine contract manufacturing market outlook. As individuals, communities, and governments are recognizing the critical role of vaccines in preventing infectious diseases and reducing healthcare burdens, immunization programs are being expanded globally. Vaccination plays a pivotal role in improving human as well as veterinary health. It reduces the need for antibiotics and prevents ailments. According to the WHO, as of August 2024, vaccination averted 3.5 Million to 5 Million fatalities annually from illnesses, such as diphtheria, tetanus, pertussis (whooping cough), influenza, and measles. This heightened consciousness is not only boosting vaccination rates but also encouraging pharmaceutical and biotech companies to scale up production quickly, often through outsourcing to contract manufacturers. CMOs provide the expertise, infrastructure, and capacity needed to meet this increasing demand efficiently while maintaining quality and compliance standards.
Rising government support
Rising government support is among the major vaccine contract manufacturing market trends. Governments worldwide are prioritizing immunization as a key public health strategy, offering grants, subsidies, and procurement commitments to ensure timely access to vaccines. This backing reduces financial risks for pharmaceutical and biotech companies, enabling them to outsource manufacturing to specialized CMOs with advanced infrastructure and regulatory expertise. Additionally, public-private partnerships are fostering innovations, improving supply chain resilience, and expanding global vaccine access. Governments are also streamlining regulatory pathways during emergencies, accelerating approval and production timelines. Such proactive support is not only strengthening confidence in contract manufacturing but also ensuring CMOs play a vital role in meeting both domestic and international vaccination needs efficiently. A UNICEF campaign effectiveness survey conducted in March 2025 found that 94% of parents and caregivers expressed increased interest in vaccination, after the Rota Vaccine media campaign by the Ministry of Health and UNICEF, and 78% reported they understood the vaccine ‘clearly’ or ‘very clearly.’
Increasing SARS-CoV-2 activity
Rising SARS-CoV-2 activity is catalyzing the demand for vaccines, booster shots, and next-generation formulations designed to address emerging variants. According to WHO data, worldwide SARS-CoV-2 activity has been increasing since mid-February 2025, with the test positivity rate attaining 11%, the highest recorded since July 2024. Continued viral circulation highlights the need for large-scale, flexible production capacity that pharmaceutical firms often achieve through outsourcing to CMOs. Contract manufacturers provide the expertise and scalability to rapidly adapt production processes for updated vaccines, ensuring timely global supply. The unpredictability of variant evolution and recurring infection waves is reinforcing the importance of maintaining strong vaccine pipelines, which is further increasing reliance on CMOs. Additionally, SARS-CoV-2 has accelerated awareness about vaccine preparedness and encouraged investments in advanced technologies like mRNA platforms, where CMOs play a pivotal role. As per the vaccine contract manufacturing market forecast, the ongoing viral activity will continue to fuel the market expansion.
Vaccine Contract Manufacturing Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global vaccine contract manufacturing market, along with forecast at the global, regional, and country levels from 2026-2034. The market has been categorized based on vaccine type, workflow, and application.
Analysis by Vaccine Type:
- Attenuated
- Inactivated
- Subunit-based
- Toxoid-based
- DNA-based
Inactivated holds 33.4% of the market share in 2025. These vaccines use killed or non-replicating pathogens, making them safer for individuals with weakened immune systems and suitable for mass immunization programs. Their production process is standardized and well-understood, allowing contract manufacturers to scale up with fewer risks compared to newer or more complex vaccine platforms. Additionally, regulatory bodies worldwide have long experience approving inactivated vaccines, which speeds up licensing and market access. These vaccines are extensively used against influenza, polio, hepatitis A, rabies, and several other infectious diseases, ensuring steady demand from both public and private healthcare providers. CMOs benefit from consistent demand, established production technologies, and the ability to serve large-scale vaccination campaigns, especially in developing regions. Furthermore, inactivated vaccines are often more stable and easier to store and transport compared to some modern alternatives, which supports global distribution.
Analysis by Workflow:
- Downstream
- Fill and Finish Operations
- Analytical and QC Studies
- Packaging
- Upstream
- Mammalian Expression Systems
- Bacterial Expression Systems
- Yeast Expression Systems
- Baculovirus/Insect Expression Systems
- Others
Downstream (fill and finish operations, analytical and QC studies, and packaging) accounts for 58% of the market share. Downstream processes are critical in ensuring the purity, safety, and effectiveness of vaccines before distribution. While upstream activities focus on cultivation and production, downstream operations, such as purification, filtration, separation, and formulation, ensure that vaccines meet stringent regulatory and quality standards. These processes are often more complex, resource-intensive, and cost-demanding, making them a major contributor to overall manufacturing expenditure. Contract manufacturers with advanced downstream capabilities add value by improving yield, maintaining consistency across large batches, and reducing contamination risks. With rising demand for sophisticated vaccines like mRNA and viral vector platforms, the downstream stage has gained greater importance in handling delicate biomolecules. Its pivotal role in product validation, compliance, and large-scale readiness is driving its dominance in the market, as no vaccine can be commercialized without rigorous downstream processing.
Analysis by Application:
Access the comprehensive market breakdown Request Sample
- Human Use
- Veterinary
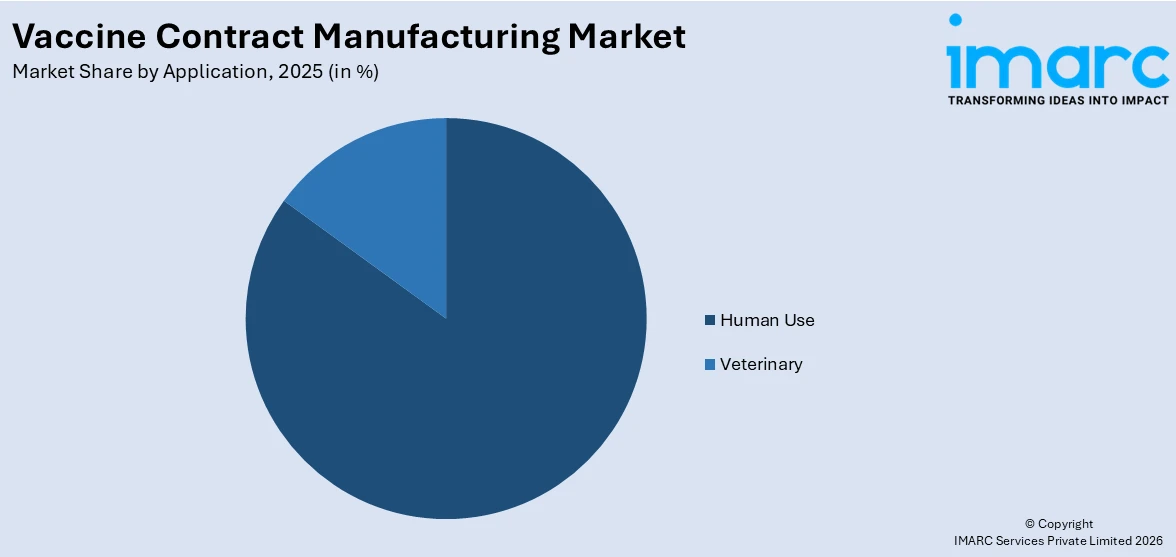
Human use holds 85% of the market share. Human use vaccines are prioritized globally because of their vital function in averting infectious diseases, lowering morbidity and mortality, and supporting public health initiatives. Governments, international health organizations, and private healthcare providers are investing in large-scale immunization programs, creating a continuous need for reliable vaccine supply. Pharmaceutical and biotech companies often rely on contract manufacturers to meet this demand efficiently, leveraging their expertise, advanced facilities, and compliance with stringent regulatory standards. The rising prevalence of infectious diseases, coupled with pandemic preparedness and emerging vaccine technologies, such as mRNA, viral vector, and recombinant platforms, is further intensifying production requirements. While veterinary vaccines are essential, the scale, urgency, and societal impact of human vaccination far exceed other applications, making human use the dominant driver of growth in the market worldwide.
Regional Analysis:
To get more information on the regional analysis of this market Request Sample
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
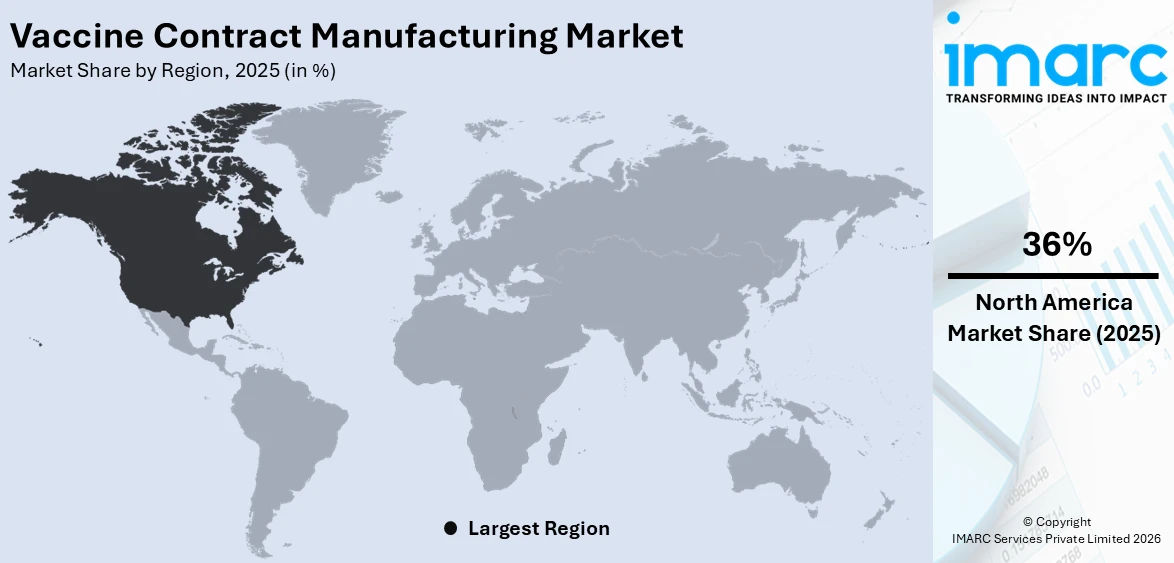
North America, accounting for a share of 36%, enjoys the leading position in the market. The region is noted for its strong biotechnology and pharmaceutical ecosystem, advanced infrastructure, and high investments in R&D activities. The region is home to leading CMOs and technology providers that offer cutting-edge capabilities in biologics, mRNA, and viral vector vaccine production, making it a hub for innovations and large-scale manufacturing. Stringent regulatory frameworks ensure high standards of quality and compliance, which enhances trust in vaccines manufactured in the region. Additionally, the presence of a well-established healthcare system, government funding for immunization programs, and strong academic-industry collaborations are accelerating vaccine development and outsourcing activities. In March 2025, Canada revealed a commitment of CAD 675 Million in fresh funding for Gavi, the Vaccine Alliance's upcoming strategic phase from 2026 to 2030, known as ‘Gavi 6.0.’ The commitment will aid Gavi's goal to safeguard a greater number of individuals from more diseases at an accelerated pace.
Key Regional Takeaways:
United States Vaccine Contract Manufacturing Market Analysis
The United States holds 88% of the market share in North America. The United States vaccine contract manufacturing market is primarily driven by the increasing prevalence of infectious diseases and the resurgence of certain vaccine-preventable illnesses. In line with this, the growing public and private investments in strengthening domestic manufacturing infrastructure and capacity are fueling the market expansion. As such, in April 2025, Merck initiated the development of a USD 1 Billion, 470,000-square-foot biologics facility in Wilmington, Delaware to manufacture KEYTRUDA and future therapies, resulting in over 500 permanent jobs and enhancing US manufacturing and innovation capabilities. Similarly, the broadening of novel vaccine platforms, including mRNA, recombinant, and vector-based technologies, is bolstering the market growth. The heightened need to meet stringent regulatory standards is also encouraging pharmaceutical companies to collaborate with experienced CMOs for compliance and quality assurance. Additionally, continual advancements in single-use bioprocessing technologies, improving production flexibility and reducing timelines, are propelling the market growth. The heightened focus on pandemic preparedness and vaccine stockpiling is driving long-term outsourcing contracts. Furthermore, the rise of personalized and therapeutic vaccines is diversifying service requirements, while the United States’ skilled biologics workforce continues to attract global manufacturing partnerships.
Europe Vaccine Contract Manufacturing Market Analysis
The European market is experiencing growth due to rising demand for pandemic preparedness measures, encouraging governments to establish large-scale production partnerships. In accordance with this, the increasing focus on regional self-sufficiency in vaccine supply is driving localized production agreements. The rising presence of strong biopharmaceutical hubs in Germany, Belgium, and the Netherlands, which are attracting significant investments in advanced facilities, is fostering the market expansion. Accordingly, in June 2025, Leyden Labs obtained EUR 20 Million from the European Investment Bank through HERA Invest to promote its pan-influenza nasal spray and pandemic readiness initiatives, aiding EU biodefense, antibiotic resistance, and respiratory virus safety projects. Moreover, ongoing advancements in bioprocessing technologies are improving scalability, efficiency, and quality standards. The heightened regulatory requirements are also motivating pharmaceutical companies to partner with experienced CMOs to ensure compliance. Furthermore, the rising prevalence of infectious diseases and the sustained demand for large-volume manufacturing are supporting the market growth. Besides this, the expansion of Europe’s vaccine pipeline is diversifying service needs, creating lucrative opportunities in the market.
Asia-Pacific Vaccine Contract Manufacturing Market Analysis
The Asia-Pacific market for vaccine contract manufacturing is largely influenced by the rapid broadening of national immunization programs. Similarly, the growing pipeline of next-generation vaccines, including mRNA, recombinant, and vector-based platforms, is fostering opportunities for specialized CMOs. The strategic collaborations between multinational pharmaceutical companies and regional manufacturers are accelerating technology transfer and ensuring compliance with global quality standards. Similarly, heightened focus on pandemic preparedness, prompting long-term manufacturing partnerships to secure supply resilience, is propelling the market growth. The region’s cost advantages in skilled labor, raw materials, and operational efficiency are also attracting global outsourcing projects. Moreover, Asia Pacific’s rising role as a competitive hub for vaccine production and export is reinforcing its prominence in the market. As such, in January 2025, Bharat Biotech allocated INR 1,500 Crore to establish a vaccine hub in Odisha that would produce eight Billion doses each year, generate 3,500 jobs, and manufacture vaccines for cholera, malaria, and polio, with plans for Chikungunya and Zika vaccines in the future.
Latin America Vaccine Contract Manufacturing Market Analysis
In Latin America, the market is advancing due to the expansion of government-led immunization programs aimed at achieving greater regional vaccine self-sufficiency. The Ministry of Health in Brazil announced that starting in 2026, 60 Million dengue vaccine doses will be manufactured annually. The initial phase will focus on people between the ages of 2 and 59, with an investment of BRL 68 Million allocated for the first production. Furthermore, rising investments in biopharmaceutical infrastructure, particularly in Brazil and Mexico, are strengthening large-scale production capacity. Additionally, strategic technology transfer agreements with multinational pharmaceutical companies are enabling the utilization of advanced manufacturing platforms and adherence to global quality standards.
Middle East and Africa Vaccine Contract Manufacturing Market Analysis
The Middle East and Africa market is accumulating traction due to government-led initiatives aimed at promoting regional vaccine production capacity and decreasing dependence on imports. In addition to this, numerous collaborations with global pharmaceutical companies, enabling access to advanced manufacturing technologies and regulatory expertise, are impelling the market growth. Similarly, the increasing prevalence of infectious diseases is driving the demand for locally produced vaccines to ensure a timely supply for national immunization programs. Apart from this, expanded public health funding is strengthening long-term vaccine manufacturing sustainability across the region. Accordingly, in June 2025, the Department of Health in Abu Dhabi sealed agreements with Sanofi, Boehringer Ingelheim, and Abbott to enhance vaccine production, promote R&D activities, localize drug manufacturing, and reinforce the Emirate’s international life sciences standing.
Competitive Landscape:
Key players are providing the technical expertise, infrastructure, and scalability needed to meet the growing global demand. These companies are investing heavily in advanced bioprocessing technologies, quality control systems, and regulatory compliance, enabling pharmaceutical and biotech firms to outsource vaccine production with confidence. By offering specialized capabilities, such as mRNA, viral vector, and recombinant vaccine manufacturing, key players are helping to accelerate development timelines and ensure consistent product quality. Their global presence also allows flexible capacity and localized production, improving supply chain resilience and accessibility. Moreover, strategic collaborations, joint ventures, and partnerships with innovators are strengthening the market by expanding service portfolios. In this way, key players act as enablers, ensuring efficiency, affordability, and speed in vaccine manufacturing. For instance, in October 2024, LenioBio collaborated with ReciBioPharm to broaden its CEPI-supported ALiCE cell-free protein expression technology for quicker vaccine manufacturing. The partnership involved transferring technology to Portugal for manufacturing at a 10-liter scale, aiding the 100 Days Mission to expedite the development of vaccines and preparedness for new health challenges.
The report provides a comprehensive analysis of the competitive landscape in the vaccine contract manufacturing market with detailed profiles of all major companies, including:
- Ajinomoto Co. Inc.
- Albany Molecular Research Inc
- Catalent Inc
- Cobra Biologics Limited (Charles River Laboratories International Inc.)
- Cytovance Biologics Inc. (Hepalink USA Inc.)
- Fujifilm Holdings Corporation
- ICON plc
- IDT Biologika GmbH
- Lonza Group AG
- Merck KGaA
- Pharmaceutical Product Development LLC
- PRA Health Sciences Inc.
Latest News and Developments:
- June 2025: Boehringer Ingelheim introduced a new single-dose vaccine for poultry in India, offering protection against Bursal, Newcastle, and Marek’s diseases. Created for broilers, layers, and breeders, it provided early, enduring immunity, decreased labor, lessened bird stress, and improved productivity while promoting sustainable, antibiotic-reducing agricultural methods.
- May 2025: TPG consented to purchase a 35% interest in SCHOTT Poonawalla from Serum Institute of India, while SCHOTT Pharma held 50%. The partnership, focused on injectables and vaccine contract production, would utilize TPG’s healthcare knowledge to enhance worldwide presence and innovation.
- May 2025: Sanofi and Vietnam Vaccine Company (VNVC) initiated a USD 77.2 Million collaboration to locally produce vaccines in Vietnam via technology transfer, training, and a new facility in Long An, targeting an annual production of 100 Million doses and enhancing regional public health readiness.
- January 2025: GeoVax revealed its enhanced MVA vaccine production method utilizing an avian suspension cell line to boost yield, reduce expenses, and enhance scalability. The innovation facilitated multi-antigen vaccines, improved distribution in resource-constrained areas, and bolstered global vaccine accessibility, including its COVID-19 candidate GEO-CM04S1.
- December 2024: Bavarian Nordic collaborated with the Serum Institute of India to manufacture Mpox vaccines for India and enhance global supply through contract production. The pact encompassed technology sharing, tackling increasing incidents, and boosting epidemic readiness for at-risk groups globally.
Vaccine Contract Manufacturing Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Vaccine Types Covered | Attenuated, Inactivated, Subunit-based, Toxoid-based, DNA-based |
| Workflows Covered |
|
| Applications Covered | Human Use, Veterinary |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Ajinomoto Co. Inc., Albany Molecular Research Inc, Catalent Inc, Cobra Biologics Limited (Charles River Laboratories International Inc.), Cytovance Biologics Inc. (Hepalink USA Inc.), Fujifilm Holdings Corporation, ICON plc, IDT Biologika GmbH, Lonza Group AG, Merck KGaA, Pharmaceutical Product Development LLC and PRA Health Sciences Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the vaccine contract manufacturing market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global vaccine contract manufacturing market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's Five Forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the vaccine contract manufacturing industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Frequently Asked Questions About the Vaccine Contract Manufacturing Market Size Report
The vaccine contract manufacturing market was valued at USD 3.3 Billion in 2025.
The vaccine contract manufacturing market is projected to exhibit a CAGR of 8.03% during 2026-2034, reaching a value of USD 6.5 Billion by 2034.
High development and manufacturing costs often discourage smaller biotech and pharmaceutical firms from setting up in-house facilities, leading them to partner with experienced contract manufacturers who offer cost efficiency and regulatory compliance expertise. Additionally, the growing pipeline of biologics, including complex mRNA and viral vector vaccines, requires specialized capabilities that contract manufacturers are well equipped to provide. Technological advancements in bioprocessing, coupled with the need for flexible, scalable production capacity, are further strengthening outsourcing trends.
North America currently dominates the vaccine contract manufacturing market, accounting for a share of 36% in 2025, due to its advanced biopharma infrastructure, strong R&D investments, skilled workforce, and the presence of leading CMOs. Stringent regulatory standards, government support, and cutting-edge technologies are further strengthening its dominance in vaccine production.
Some of the major players in the vaccine contract manufacturing market include Ajinomoto Co. Inc., Albany Molecular Research Inc, Catalent Inc, Cobra Biologics Limited (Charles River Laboratories International Inc.), Cytovance Biologics Inc. (Hepalink USA Inc.), Fujifilm Holdings Corporation, ICON plc, IDT Biologika GmbH, Lonza Group AG, Merck KGaA, Pharmaceutical Product Development LLC, PRA Health Sciences Inc., etc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












