
COVID-19 Diagnostics Market Report by Product Type (Reagents and Kits, Instruments, and Others), Sample Type (Nasopharyngeal Swabs, Oropharyngeal Swabs, Nasal Swabs, Blood, and Others), Test Type (Molecular (RT-PCR) Testing, Antigen-based Testing, Antibody (Serology) Testing, and Others), Mode (Point-of-Care (PoC), Non-Point-of-Care (Non-PoC)), End Use (Laboratories, Hospitals, Diagnostic Centers and Clinics, and Others), and Region 2026-2034
Market Overview:
The global COVID-19 diagnostics market size reached USD 54.1 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 28.1 Billion by 2034.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Market Size in 2025
|
USD 54.1 Billion |
|
Market Forecast in 2034
|
USD 28.1 Billion |
COVID-19 diagnostics refer to the clinical tests conducted to detect the presence of the viral infection among individuals. COVID-19 is an infectious disease caused by the newly discovered zoonotic coronavirus. Some common symptoms include fever, fatigue, dry cough, shortness of breath, sore throat and body aches. The viral infection is usually diagnosed through reverse transcriptase-polymerase chain reaction (RT- PCR), antigen-based and antibody testing. These tests use reagents and kits to obtain the sample from nasopharyngeal (NP), oropharyngeal (OP) and nasal swabs, human saliva or blood. They are widely used in hospitals, laboratories, clinics, research institutes and diagnostic centers.
The sudden outbreak of coronavirus disease and an alarming increase in the number of COVID-19 cases across the globe are among the key factors creating a positive outlook for the market growth. The continuous rise in the number of daily cases has led to the widespread adoption of diagnostic tests for rapid and comprehensive testing to provide early treatment to the patients. In line with this, the increasing demand for mass testing and molecular diagnostics is contributing to the market growth. Additionally, various product innovations, such as the launch of novel antibody and serology test kits for home-testing, are acting as other growth-inducing factors. In comparison to the traditionally used testing kits, these variants are less time-consuming and can be integrated with software-based programs for accurate results. Other factors, including the implementation of favorable government polices to prevent the further spread of COVID-19 infection, along with significant improvements in the healthcare infrastructure, especially in the developing economies, are anticipated to drive the market growth.
Key Market Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global COVID-19 diagnostics market report, along with forecasts at the global, regional and country levels from 2026-2034. Our report has categorized the market based on product type, sample type, test type, mode and end use.
Breakup by Product Type:
- Reagents and Kits
- Instruments
- Others
Breakup by Sample Type:
- Nasopharyngeal Swabs
- Oropharyngeal Swabs
- Nasal Swabs
- Blood
- Others
Breakup by Test Type:
- Molecular (RT-PCR) Testing
- Antigen-based Testing
- Antibody (Serology) Testing
- Others
Breakup by Mode:
- Point-of-Care (PoC)
- Non-Point-of-Care (Non-PoC)
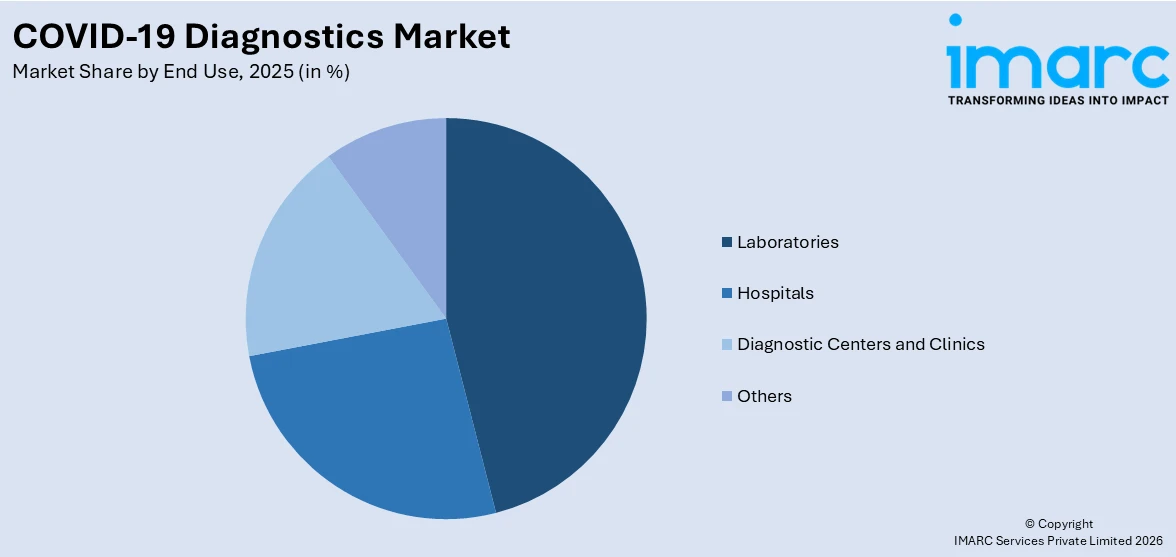
Breakup by End Use:
Access the comprehensive market breakdown Request Sample
- Laboratories
- Hospitals
- Diagnostic Centers and Clinics
- Others
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
Competitive Landscape:
The competitive landscape of the industry has also been examined along with the profiles of the key players being Abbott Laboratories, Becton Dickinson and Company, bioMérieux SA, Bio-Rad Laboratories, Danaher Corporation, F. Hoffman-La Roche Ltd. (Roche Holding AG), Luminex Corporation, PerkinElmer Inc., Quest Diagnostics Incorporated, Robert Bosch GmbH, Seegene Inc., Siemens Aktiengesellschaft and Thermo Fisher Scientific Inc.
Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Segment Coverage | Product Type, Sample Type, Test Type, Mode, End Use, Region |
| Region Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Abbott Laboratories, Becton Dickinson and Company, bioMérieux SA, Bio-Rad Laboratories, Danaher Corporation, F. Hoffman-La Roche Ltd. (Roche Holding AG), Luminex Corporation, PerkinElmer Inc., Quest Diagnostics Incorporated, Robert Bosch GmbH, Seegene Inc., Siemens Aktiengesellschaft, Thermo Fisher Scientific Inc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Frequently Asked Questions About the COVID-19 Diagnostics Market Report
The global COVID-19 diagnostics market was valued at USD 54.1 Billion in 2025.
Due to the sudden outbreak of the COVID-19 and the increasing number of cases, there has been a rising demand for COVID-19 diagnostics for rapid and comprehensive testing to provide early treatment to the coronavirus infected patients.
The rising demand for mass testing and molecular diagnostics, along with the advent of novel antibody and serology test kits for home-testing, is primarily driving the global COVID-19 diagnostics market.
Based on the product type, the global COVID-19 diagnostics market can be segmented into reagents and kits, instruments, and others. Currently, instruments hold the majority of the total market share.
Based on the sample type, the global COVID-19 diagnostics market has been divided into nasopharyngeal swabs, oropharyngeal swabs, nasal swabs, blood, and others. Among these, nasopharyngeal swabs currently exhibit a clear dominance in the market.
Based on the test type, the global COVID-19 diagnostics market can be categorized into molecular (RT-PCR) testing, antigen-based testing, antibody (serology) testing, and others. Currently, molecular (RT-PCR) testing accounts for the majority of the global market share.
Based on the mode, the global COVID-19 diagnostics market has been segregated into Point-of-Care (PoC) and Non-Point-of-Care (Non-PoC), where Non-Point-of-Care (Non-PoC) currently holds the largest market share.
Based on the end use, the global COVID-19 diagnostics market can be bifurcated into laboratories, hospitals, diagnostic centers and clinics, and others. Currently, laboratories exhibit a clear dominance in the market.
On a regional level, the market has been classified into North America, Asia-Pacific, Europe, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global COVID-19 diagnostics market include Abbott Laboratories, Becton Dickinson and Company, bioMérieux SA, Bio-Rad Laboratories, Danaher Corporation, F. Hoffman-La Roche Ltd. (Roche Holding AG), Luminex Corporation, PerkinElmer Inc., Quest Diagnostics Incorporated, Robert Bosch GmbH, Seegene Inc., Siemens Aktiengesellschaft, and Thermo Fisher Scientific Inc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












