
Digital Patient Monitoring Devices Market Report by Monitoring Type (Wireless Sensor Technology, mHealth, Telehealth, Wearable Devices, Remote Patient Monitoring), Product (Diagnostic Monitoring Devices, Therapeutic Monitoring Devices), End User (Hospitals, Home Settings, Ambulatory Surgical Centers (ASC)), and Region 2026-2034
Global Digital Patient Monitoring Devices Market:
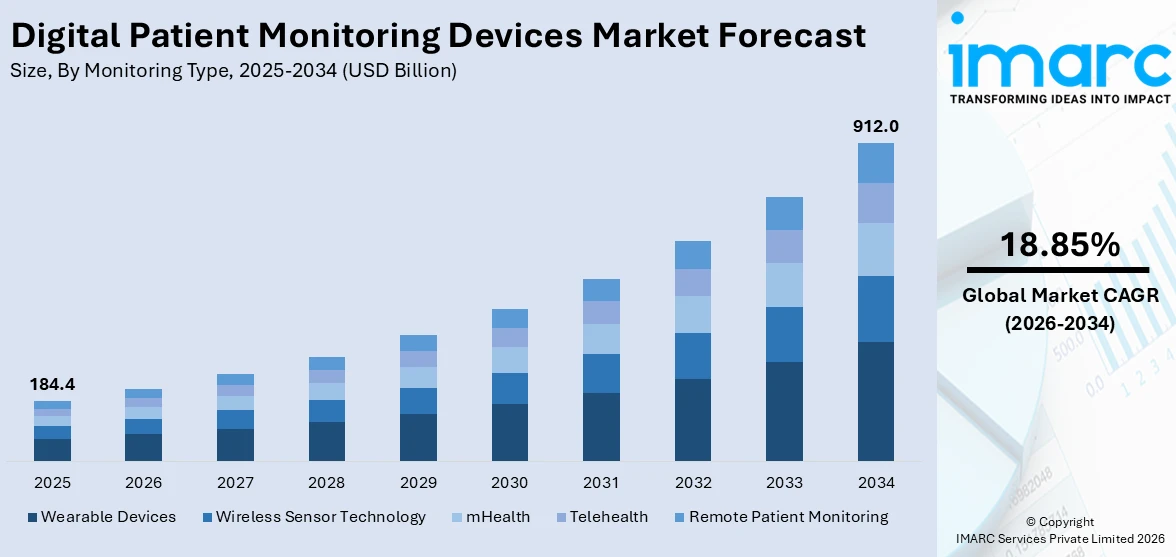
The global digital patient monitoring devices market size reached USD 184.4 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 912.0 Billion by 2034, exhibiting a growth rate (CAGR) of 18.85% during 2026-2034. The rising prevalence of chronic diseases, shift towards remote patient monitoring, and technological advancements are primarily driving the digital patient monitoring market growth.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 184.4 Billion |
|
Market Forecast in 2034
|
USD 912.0 Billion |
| Market Growth Rate 2026-2034 | 18.85% |
Digital Patient Monitoring Devices Market Analysis:
- Major Market Drivers: Significant growth in the healthcare industry, the rising demand for continuous monitoring devices, and the increasing prevalence of chronic medical ailments, such as hypertension, asthma, blood pressure, and diabetes, represent key factors majorly driving the digital patient monitoring devices market growth.
- Key Market Trends: Various advancements in technology, such as the introduction of smart digital patient monitoring devices that assist in the quick analysis of common physiological parameters, are providing an impetus to the market growth. Additionally, the increasing demand for various sensor-based wearable monitoring devices, such as continuous glucose monitors that help in tracking the patient's blood sugar levels on a real-time basis, is favoring the digital patient monitoring devices market demand.
- Competitive Landscape: Some of the prominent digital patient monitoring devices companies include Abbott, Baxter International Inc., GE HealthCare, Masimo Corporation, Medtronic plc, Nihon Kohden Corporation, OMRON Healthcare Co., Ltd., and VitalConnect, among many others.
- Geographical Trends: According to the digital patient monitoring devices market dynamics, North America is dominating the overall market owing to the increasing prevalence of chronic diseases, rising healthcare costs, and advancements in technology such as IoT and AI.
- Challenges and Opportunities: Regulatory hurdles, integration with healthcare systems, and data privacy security concerns are some of the key challenges that the market is facing. However, the increasing prevalence of chronic diseases, such as diabetes, cardiovascular ailments, respiratory disorders, etc., creates a growing demand for continuous patient monitoring solutions.
To get more information on this market Request Sample
Digital Patient Monitoring Devices Market Trends:
Rising Prevalence of Chronic Diseases
The rising prevalence of chronic ailments has created a demand for continuous monitoring solutions. For instance, according to an article published by the Mayo Clinic, in 2020, an estimated 523 million people globally reported some type of CVD, with nearly 19 million fatalities attributed to it. This accounts for around 32% of all global deaths, an 18.7% rise from 2010. Digital patient monitoring devices enable healthcare providers to remotely monitor patients' health parameters and intervene early, leading to better management of chronic conditions and reduced healthcare costs. These factors are further contributing to the digital patient monitoring devices market share.
Increasing Geriatric Population
The increasing geriatric population is one of the key factors driving the digital patient monitoring devices market. For instance, according to an article published by the World Health Organization, by 2030, one in every six persons in the world will be 60 or older. At this time, the proportion of the population aged 60 and up will rise from 1 billion in 2020 to 1.4 billion. By 2050, the global population of persons aged 60 and up will double (2.1 billion). The number of people aged 80 and older is anticipated to triple between 2020 and 2050, reaching 426 million. Many elderly individuals prefer to age in place, staying in their own homes rather than moving to assisted living facilities or nursing homes. Digital monitoring devices support aging in place by providing healthcare providers and caregivers with real-time data on the patient's health status remotely. These factors are further bolstering the digital patient monitoring market revenue.
Technological Advancements
Technological advancements are significantly driving the growth of the digital patient monitoring devices market. Wearable devices, such as smartwatches, fitness trackers, and patches, allow for seamless integration into daily life, enabling patients to monitor their health in real-time and healthcare providers to gather data remotely. For instance, in July 2024, KORE, a Georgia-based IoT startup, and mCare Digital launched mCareWatch 241, a wristwatch designed for virtual patient monitoring. The watch has a geo-fence alert, a pedometer, a heart rate monitor, speed dialing, fall detection, a call feature that allows users to request emergency assistance, a web dashboard and mobile app, and GPS tracking and reminders. These factors are positively influencing the digital patient monitoring devices market forecast.
Global Digital Patient Monitoring Devices Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global digital patient monitoring devices market report, along with forecasts at the global, regional, and country levels from 2026-2034. Our report has categorized the market based on monitoring type, product, and end user.
Breakup by Monitoring Type:
- Wireless Sensor Technology
- mHealth
- Telehealth
- Wearable Devices
- Remote Patient Monitoring
Currently, wearable devices hold the majority of the total market share
The report has provided a detailed breakup and analysis of the digital patient monitoring devices market based on the monitoring type. This includes wireless sensor technology, mHealth, telehealth, wearable devices, and remote patient monitoring. According to the report, wearable devices hold the majority of the total market share.
According to the digital patient monitoring devices market outlook, wearable devices such as smartwatches and fitness trackers have gained widespread popularity among consumers for tracking fitness metrics, sleep patterns, and overall wellness. Consumers are increasingly using these devices to monitor health indicators like heart rate variability, stress levels, and even blood oxygen saturation. For instance, in June 2023, STAT Health, a digital health company based in the U.S., launched a wearable general wellness device that can be worn 24/7 that analyzes blood flow to the brain to help with recognizing issues in the body that arise when standing.
Breakup by Product:
- Diagnostic Monitoring Devices
- Vital Sign Monitors
- Sleep Monitors
- Fetal Monitors
- Neuromonitors
- Others
- Therapeutic Monitoring Devices
- Insulin Monitors
- Respiratory Monitors
- Others
Diagnostic monitoring devices currently exhibit a clear dominance in the market
A detailed breakup and analysis of the digital patient monitoring devices market based on the product has also been provided in the report. This includes diagnostic monitoring devices (vital sign monitors, sleep monitors, fetal monitors, neuromonitors, and others) and therapeutic monitoring devices (insulin monitors, respiratory monitors, and others). According to the digital patient monitoring market report, diagnostic monitoring devices currently exhibit a clear dominance in the market.
As per the digital patient monitoring devices market outlook, diagnostic monitoring devices are indispensable for accurate diagnosis, disease monitoring, and treatment planning across various medical specialties. Healthcare providers rely on these devices to detect and monitor diseases early, optimize treatment strategies, and improve patient outcomes. Moreover, continuous improvements in sensor technology, connectivity (IoT), artificial intelligence (AI), and data analytics enhance the capabilities and accuracy of diagnostic monitoring devices. For instance, in September 2023, Beacon Biosignals announced the FDA 510(k) Clearance for the Dreem 3S, an advanced wearable headband with integrated machine learning algorithms to capture electroencephalogram (EEG) data from the brain to monitor sleep architecture and help in the diagnosis of disturbed sleep.
Breakup by End User:
Access the comprehensive market breakdown Request Sample
- Hospitals
- Home Settings
- Ambulatory Surgical Centers (ASC)
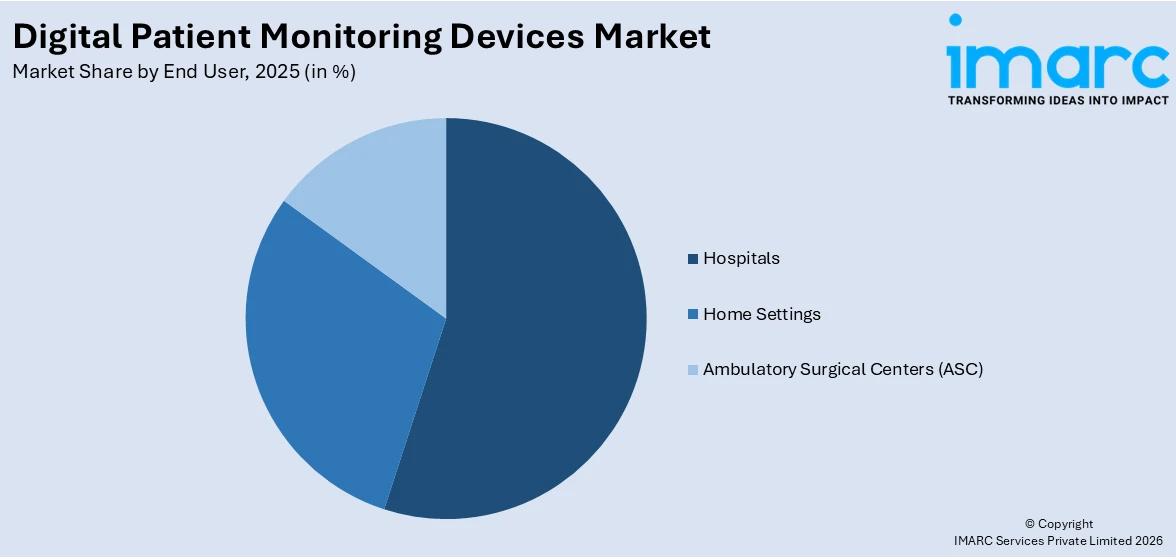
Hospitals account for the largest market share
The report has provided a detailed breakup and analysis of the digital patient monitoring devices market based on the end user. This includes hospitals, home settings, and ambulatory surgical centers (ASC). According to the report, hospitals account for the largest market share.
Digital patient monitoring devices enable hospitals to continuously monitor patients' vital signs, physiological parameters, and health metrics in real time. These devices provide accurate and timely data, facilitating early detection of clinical deterioration, prompt interventions, and improved patient safety.
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America currently dominates the global market
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America dominates the global market.
According to the digital patient monitoring devices market statistics, North America faces a rising prevalence of chronic diseases, such as diabetes, cardiovascular ailments, and respiratory disorders. For instance, as per the November 2022 study released by the Canadian Chronic Disease Surveillance System, around 6.3 million adults 65 years of age and above are anticipated to be living with chronic diseases in the near future. Digital patient monitoring devices enable continuous monitoring of vital signs, glucose levels, blood pressure, and other health metrics, empowering patients and healthcare providers to manage chronic conditions more effectively.
Competitive Landscape:
The report has also provided a comprehensive analysis of the competitive landscape in the global digital patient monitoring devices market. Competitive analysis such as market structure, market share by key players, player positioning, top winning strategies, competitive dashboard, and company evaluation quadrant has been covered in the report. Also, detailed profiles of all major companies have been provided. Some of the companies covered are:
- Abbott
- Baxter International Inc.
- GE HealthCare
- Masimo Corporation
- Medtronic plc
- Nihon Kohden Corporation
- OMRON Healthcare Co., Ltd.
- VitalConnect
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
Digital Patient Monitoring Devices Market Recent Developments:
- July 2024: KORE, a Georgia-based IoT startup, and mCare Digital launched mCareWatch 241, a wristwatch designed for virtual patient monitoring.
- April 2024: Royal Philips, a health technology provider, partnered with Philips to integrate smartQare’s advanced solution, viQtor. This collaboration aims to enable the next generation of continuous patient monitoring both in and out of the hospital, in Europe.
- April 2024: BlueBrix announced new micro frontends for its Patient Support Program and Remote Patient Monitoring Modules, allowing for seamless integration into digital health applications while boosting care standards.
Digital Patient Monitoring Devices Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Predictive Market Assessment:
|
| Monitoring Types Covered | Wireless Sensor Technology, mHealth, Telehealth, Wearable Devices, Remote Patient Monitoring |
| Products Covered |
|
| End Users Covered | Hospitals, Home Settings, Ambulatory Surgical Centers (ASC) |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Abbott, Baxter International Inc., GE HealthCare, Masimo Corporation, Medtronic plc, Nihon Kohden Corporation, OMRON Healthcare Co., Ltd., VitalConnect, etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the digital patient monitoring devices market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global digital patient monitoring devices market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assist stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the digital patient monitoring devices industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Frequently Asked Questions About the Digital Patient Monitoring Devices Market Report
The global digital patient monitoring devices market was valued at USD 184.4 Billion in 2025.
We expect the global digital patient monitoring devices market to exhibit a CAGR of 18.85% during 2026-2034.
The sudden outbreak of the COVID-19 pandemic has led to the rising adoption of digital patient monitoring devices to remotely monitor the vital parameters of the coronavirus-infected patients, such as blood pressure, heart rate, temperature, etc.
The advent of smart digital and sensor-based wearable monitoring devices, which assist in the quick analysis of common physiological parameters and allows the blood sugar levels of the patients to be tracked, is primarily driving the global digital patient monitoring devices market.
Based on the monitoring type, the global digital patient monitoring devices market can be segmented into wireless sensor technology, mHealth, telehealth, wearable devices, and remote patient monitoring Currently, wearable devices hold the majority of the total market share.
Based on the product, the global digital patient monitoring devices market has been divided into diagnostic monitoring devices and therapeutic monitoring devices, where diagnostic monitoring devices currently exhibit a clear dominance in the market.
Based on the end user, the global digital patient monitoring devices market can be categorized into hospitals, home settings, and Ambulatory Surgical Centers (ASC). Among these, hospitals account for the largest market share.
On a regional level, the market has been classified into North America, Asia-Pacific, Europe, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global digital patient monitoring devices market include Abbott, Baxter International Inc., GE HealthCare, Masimo Corporation, Medtronic plc, Nihon Kohden Corporation, OMRON Healthcare Co., Ltd., and VitalConnect.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












