
Generic Oncology Drugs Market: Global Industry Trends, Share, Size, Growth, Opportunity and Forecast 2026-2034
Generic Oncology Drugs Market Size:
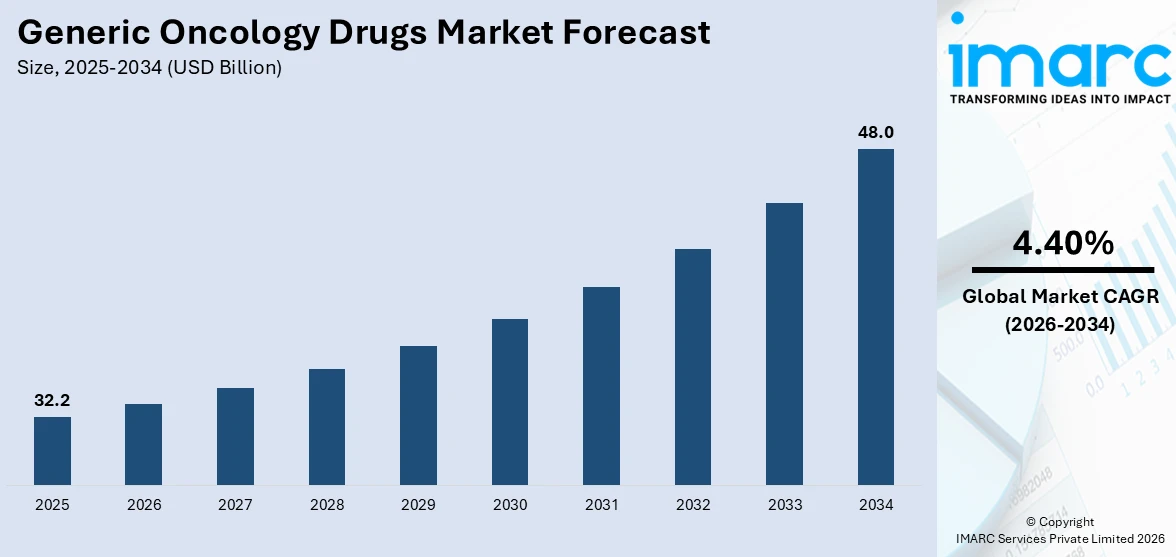
The global generic oncology drugs market size reached USD 32.2 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 48.0 Billion by 2034, exhibiting a growth rate (CAGR) of 4.40% during 2026-2034. The growing prevalence of cancer due to excessive smoking of tobacco products and consumption of alcohol, rising investments in research operations for developing novel drugs, and increasing implementation of stringent regulations for streamlining the drug production process are some of the major factors propelling the growth of the market.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 32.2 Billion |
|
Market Forecast in 2034
|
USD 48.0 Billion |
| Market Growth Rate 2026-2034 | 4.40% |
Generic Oncology Drugs Market Analysis:
- Major Market Drivers: The rising prevalence of cancer worldwide is leading to the increased demand for affordable treatment options which represents the major driver of the market. Generic drugs offer a cost-effective alternative to branded oncology treatments, making them accessible to a broader patient population. The expiration of patents for several blockbuster oncology drugs has opened the market for generics, further fueling the market.
- Key Market Trends: The growing focus on biosimilars is gaining traction as patents for biologic oncology drugs expire which represents a key trend of the market. The development of new generic formulations with improved efficacy and safety profiles is also a significant trend. The market is witnessing a shift toward personalized medicine, where generic drugs are being integrated into tailored treatment regimens.
- Geographical Trends: According to the generic oncology drugs market overview, North America accounted for the largest region in the market. The rising prevalence of cancer, the expiration of patents for major branded oncology drugs, and the increasing demand for affordable treatment options are driving the growth of the market across the region. The supportive government policies and efforts to reduce healthcare costs are encouraging the adoption of generic alternatives, which is further fueling the market growth in the region.
- Competitive Landscape: The report has also provided a comprehensive analysis of the competitive landscape in the market. Competitive analysis such as market structure, key player positioning, top winning strategies, competitive dashboard, and company evaluation quadrant has been covered in the report. Also, detailed profiles of all major companies have been provided.
- Challenges and Opportunities: Some of the key generic oncology drugs market recent opportunities include the increasing demand for cost-effective cancer treatment, especially as more branded drugs lose patent protection. In line with this, the expanding access to cancer care in emerging markets and the growing acceptance of biosimilars also offer growth potential, enabling companies to capture a larger share of the generic oncology drug market. However, the market for generic oncology drugs also faces several challenges including strict regulatory requirements, intense competition, and pricing pressures which can impact profitability and market entry. In addition to this, the complex manufacturing processes for oncology drugs and the need for rigorous clinical trials create barriers for new entrants.
To get more information on this market Request Sample
Generic Oncology Drugs Market Trends:
Increasing Cases of Cancer
The rising incidence of cancer globally is acting as a major growth-inducing factor in the market. As cancer cases increase, so does the demand for effective and affordable treatment options. According to the American Cancer Society, 2,001,140 new cancer cases and 611,720 cancer deaths are expected to occur in the United States in 2024. Generic oncology drugs provide a cost-effective alternative to branded medications, making cancer treatment more accessible to a broader patient population. This is particularly important in low- and middle-income countries, where the financial burden of cancer treatment can be overwhelming. With cancer emerging as a leading cause of morbidity and mortality, the availability of cost-effective and reliable treatment options is paramount, which, in turn, is expected to generate the generic oncology drugs market revenue significantly. For instance, in March 2024, Zydus Lifesciences announced the launch of olaparib, a PARP inhibitor, under the brand name IBYRA in India, making advanced cancer treatment accessible to all.
Growing Government Initiatives and Regulations
Several governments across the world are implementing initiatives and regulations to promote the use of generic oncology drugs as part of broader efforts to reduce healthcare costs. For instance, in April 2024, Gland Pharma received approval from the USFDA to market a generic medication for breast cancer treatment, Eribulin Mesylate Injection. It's likely to be the first generic authorization in the market, having sales of around USD 92 million in the US. According to the generic oncology drugs market forecast, these initiatives often include policies that encourage the substitution of branded drugs with generics, streamlined approval processes for generic drugs, on incentives for manufacturers to produce affordable oncology medications. By supporting the adoption of generics, governments aim to make cancer treatment more accessible and sustainable, especially in countries with limited healthcare resources.
Rising Patent Expirations and Cost-Effective Solutions
The growing expiration of patents for many blockbuster oncology drugs is opening the market to generic versions, which are significantly more affordable than their branded counterparts. As patents expire, generic manufacturers can produce and market these drugs, leading to increased competition and lower prices. The availability of affordable generic oncology drugs ensures that more patients can access treatment, thereby creating a positive generic oncology drugs market outlook across the globe. For instance, according to a study by the Department of Pharmaceuticals, Government of India (GoI), 24 huge blockbuster medications, including the well-known asthma inhaler Symbicort and the internationally recognized arthritis and cancer medications Humira and Keytruda, are expected to lose their patents by 2030. Drugs that had annual sales of more than $250 billion, or Rs 20,000 crore, were confiscated. By 2030, 75 lakh crore are projected to become off-patent, thus providing a huge market for the manufacture of low-cost generics not only in India but across the globe.
Generic Oncology Drugs Market Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the market, along with forecasts at the global and regional levels for 2026-2034.
Regional Insights:
- North America
- Europe
- Asia Pacific
- Middle East and Africa
- Latin America
North America leads the market, accounting for the largest generic oncology drugs market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America, Europe, Asia Pacific, the Middle East and Africa, and Latin America. According to the report, North America represents the largest regional market for generic oncology drugs.
The rising prevalence of cancer which increases the demand for cost-effective treatment options is driving the growth of the market across the region. The expiration of patents for major branded oncology drugs has opened the market for generic alternatives, offering more affordable options for patients and healthcare providers. The growing acceptance and availability of biosimilars, coupled with advancements in generic drug manufacturing are further contributing to the generic oncology drugs market growth in the region. For instance, in June 2024, Teva Pharmaceuticals, Inc., a subsidiary of Teva Pharmaceutical Industries Ltd. based in the United States, announced the inauguration of an approved generic of Victoza1, in the U.S. The first-ever generic GLP-1, Victoza (liraglutide injection 1.8mg), helps meet the growing demand in the US market for this class of medications. Teva is further demonstrating its commitment to its sophisticated generic pharmaceutical portfolio with this launch. Liraglutide injection is recommended to lower the risk of cardiovascular events in individuals with type 2 diabetes mellitus. In addition to this, supportive government policies and initiatives aimed at reducing healthcare costs are encouraging the generic oncology drugs demand.
Competitive Landscape:
- The report has also provided a comprehensive analysis of the competitive landscape in the market. Competitive analysis such as market structure, key player positioning, top winning strategies, competitive dashboard, and company evaluation quadrant has been covered in the report. Also, detailed profiles of all major companies have been provided.
- The market for generic oncology drugs is highly competitive, with key players including Teva Pharmaceuticals, Mylan N.V., Sandoz (a Novartis division), and Pfizer, among others. These generic oncology drugs companies are focused on expanding their portfolios through the development of cost-effective generic versions of blockbuster oncology drugs. For instance, in April 2024, Teva Pharmaceuticals International GmbH, a subsidiary of Teva Pharmaceutical Industries Ltd., and mAbxience, a Fresenius Kabi majority-owned group with partial ownership from Insud Pharma, announced that they had signed a strategic licensing agreement for a biosimilar candidate which is presently in progress for the treatment of numerous oncology indications. Moreover, the market is characterized by intense price competition, with companies striving to gain market share by offering lower-cost alternatives to branded treatments. In addition to this, some of the key generic oncology drugs market recent developments include strategic partnerships, mergers, and acquisitions are common as companies seek to strengthen their positions and expand their reach in the global oncology market.
Generic Oncology Drugs Market News:
- In June 2023, India's Aurobindo Pharma announced that its subsidiary Eugia Pharma Specialities had signed a voluntary sub-licensing agreement with Medicines Patent Pool to develop and market an anti-cancer drug- Nilotinib capsules. Chronic myeloid leukemia is treated with nilotinib pills.
- In May 2024, the global oncology company BeiGene and Glenmark Pharmaceuticals' subsidiary Glenmark Specialty S.A. announced that they have entered into an exclusive marketing and distribution deal, the company stated. Tislelizumab is a new anti-PD-1 monoclonal antibody that has been approved by the FDA, EMA, and NMPA for the treatment of metastatic or advanced esophageal squamous cell carcinoma. It is also being developed broadly for the treatment of other cancer types.
Generic Oncology Drugs Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the generic oncology drugs market from 2020-2034.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the global generic oncology drugs market.
- The study maps the leading, as well as the fastest-growing, regional markets.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the generic oncology drugs industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Frequently Asked Questions About the Generic Oncology Drugs Market Report
The global generic oncology drugs market was valued at USD 32.2 Billion in 2025.
We expect the global generic oncology drugs market to exhibit a CAGR of 4.40% during 2026-2034.
The sudden outbreak of the COVID-19 pandemic has led to the growing adoption of generic oncology drugs over other methods of cancer treatments for reducing the risk of the coronavirus infection upon hospital visits.
The rising awareness towards the benefits of early cancer diagnosis, along with the increasing preference for generic oncology drugs among patients, owing to the high costs of full-length cancer treatment, is primarily driving the global generic oncology drugs market.
On a regional level, the market has been classified into North America, Europe, Asia Pacific, Middle East and Africa, and Latin America, where North America currently dominates the global market.
The growing focus on biosimilars is gaining traction as patents for biologic oncology drugs expire which represents a key trend of the market. The development of new generic formulations with improved efficacy and safety profiles is also a significant trend. The market is witnessing a shift toward personalized medicine, where generic drugs are being integrated into tailored treatment regimens.
Ethical issues in the development of generic oncology drug include concerns about intellectual property, quality and safety, access and affordability, drug shortages, clinical trials, and transparency.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












