
Genetic Testing Market Size, Share, Trends and Forecast by Type, Technology, Application, and Region, 2026-2034
Genetic Testing Market Size and Share:
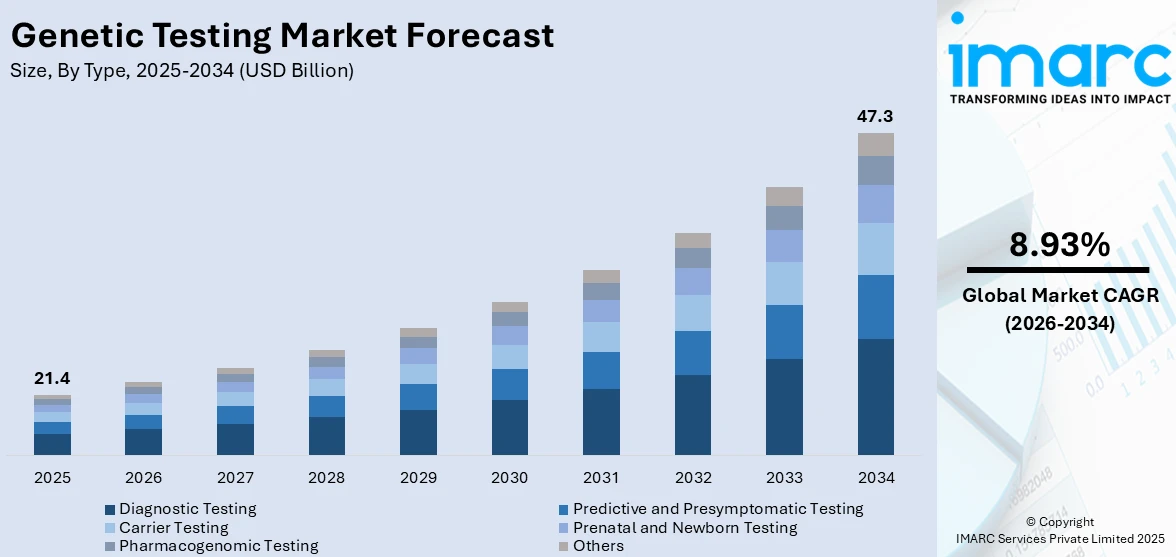
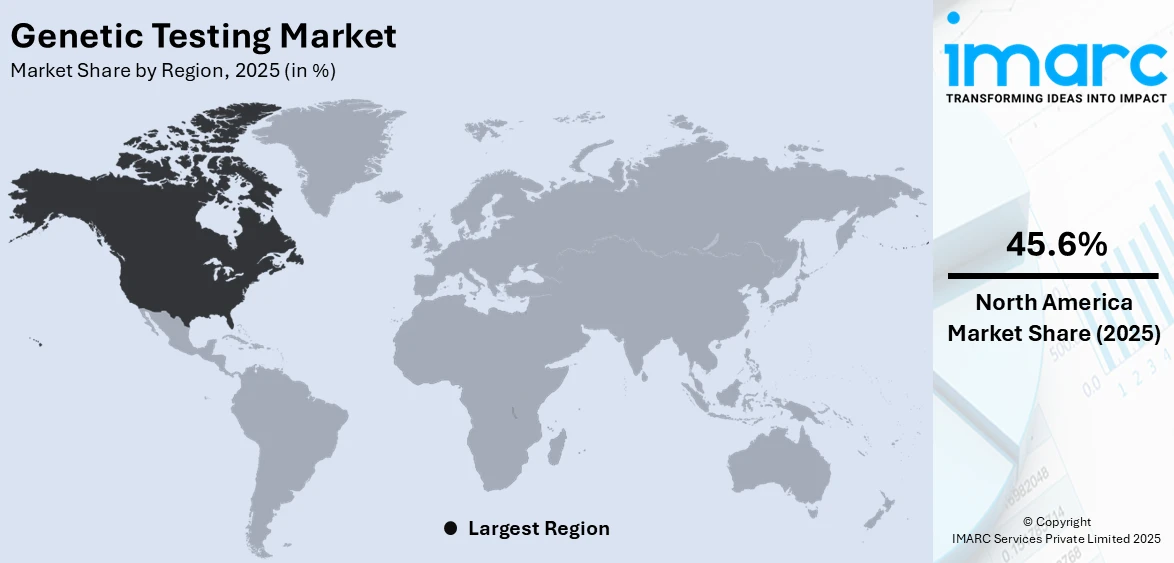
The global genetic testing market size was valued at USD 21.4 Billion in 2025. Looking forward, IMARC Group estimates the market to reach USD 47.3 Billion by 2034, exhibiting a CAGR of 8.93% from 2026-2034. North America currently dominates the market, holding a market share of over 45.6% in 2025. The North American market is primarily driven by technological advancements, escalating demand for personalized medicine, rising prevalence of genetic disorders, introduction of supportive government initiatives, and increasing implication of ethical, legal, and social considerations.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025 |
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
| Market Size in 2025 | USD 21.4 Billion |
| Market Forecast in 2034 | USD 47.3 Billion |
| Market Growth Rate (2026-2034) | 8.93% |
At present, there is a rise in the occurrence of various genetic disorders among the masses. Conditions such as cystic fibrosis, sickle cell anemia, and Huntington’s disease, which have strong genetic components, are receiving more attention in both research and clinical applications. Additionally, the increasing incidence of chronic conditions like cancer and cardiovascular diseases, often linked to genetic predisposition, has further underscored the need for early diagnosis through genetic testing. Early identification of such conditions can significantly improve patient outcomes, making genetic testing an essential tool in preventive healthcare. Advances in genomics are enabling tailored treatment options based on an individual's genetic profile, leading to better therapies with fewer side effects. Physicians and healthcare providers are increasingly adopting genetic testing to guide decisions for cancer treatment, rare disease management, and even chronic condition interventions.
To get more information on this market Request Sample
The United States has emerged as a significant region in the genetic testing market owing to the advances in precision medicine. Healthcare providers are increasingly relying on genetic tests to guide medical decision-making, especially for oncology, cardiology, and rare diseases. By identifying genetic mutations or variations, these tests help clinicians predict disease risks, determine disease progression, and tailor treatments to the individual. Genetic testing has become a critical component of reproductive health in the United States. Carrier screening and non-invasive prenatal testing (NIPT) are increasingly common among expectant parents who want to analyze the risk of passing genetic issues to children. NIPT, in particular, has gained widespread adoption due to its ability to detect chromosomal abnormalities like Down syndrome, via a blood test. This technology has reduced the need for invasive procedures like amniocentesis, which carry higher risks. The growing use of genetic testing in fertility clinics and obstetric practices reflects a broader trend of integrating genomics into family planning and prenatal care. As per the IMARC Group’s predictions, the US genetic testing market is expected to reach USD 14.4 billion by 2033.
Genetic Testing Market Trends:
Advancements in genetic testing technologies
Advancements in genetic testing technologies are instrumental in driving the genetic testing market. These technological innovations have led to more accurate, efficient, and cost-effective genetic analysis methods, expanding the scope of testing into areas such as oncology, pharmacogenomics, and predictive testing, further supporting the market growth. Enhanced sequencing techniques, such as next-generation sequencing (NGS), provide detailed insights into genetic disorders, enabling early detection and personalized treatment plans. For instance, a study on 1914 women revealed cfDNA testing significantly reduces false positives for trisomies 21 and 18 compared to standard screening, with a 100% success rate in detecting aneuploidies and higher predictive values. Furthermore, simplified processes have increased the adoption of genetic testing across various healthcare settings, driving the genetic testing market demand.
Increasing awareness and demand for personalized medicine
Increasing awareness and demand for personalized medicine are playing a significant role in shaping the genetic testing market outlook. Personalized medicine, which relies on genetic information to tailor treatments, is driving the demand for genetic tests. This demand is particularly evident in sectors such as oncology and reproductive health, where genetic testing guides therapy decisions and assesses genetic disorder risks in offspring. For example, according to a study, a survey with a 36% response rate showed that 83% of the 312 respondents had minimal inquiries about direct-to-consumer genetic testing, 14% dealt with result interpretation, and over 50% supported the role of genetic counsellors and clinical settings for genetic testing, with 70% open to considering it for patients with privacy, anonymity, or access concerns. Additionally, the growing interest in understanding genetic predispositions to health conditions further bolsters the market growth.
Rise in genetic disorders and chronic diseases
The rise in genetic disorders and chronic diseases globally is further escalating the genetic testing market statistics. Genetic testing plays a vital role in the early detection, diagnosis, and management of various conditions like sickle cell anemia, cystic fibrosis, and Huntington’s disease. Moreover, with the increasing incidence of chronic diseases, including cancer, cardiovascular diseases, and diabetes, there is a heightened need for testing to identify genetic mutations predisposing individuals to these conditions. The World Health Organization (WHO) reports that non-communicable diseases (NCDs), such as cancer, cardiovascular diseases, and diabetes, are responsible for 73% of global deaths annually. Genetic testing can help identify individuals at risk for these conditions. Early detection through testing improves patient outcomes, further propels the market. The genetic testing market is substantial, with a considerable growth in the number of genetic testing per year in the US and other developed countries. Genetic testing in pregnancy is also becoming increasingly prevalent, providing valuable insights into fetal health and genetic conditions.
Genetic Testing Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global genetic testing market, along with forecast at the global, regional, and country levels from 2026-2034. The market has been categorized based on type, technology, and application.
Analysis by Type:
- Predictive and Presymptomatic Testing
- Carrier Testing
- Prenatal and Newborn Testing
- Diagnostic Testing
- Pharmacogenomic Testing
- Others
Diagnostic testing is employed to reveal or confirm specific inherited disease in symptomatic people. This sector is on the rise due to its contribution to a precise diagnosis, which is necessary for appropriate treatment and control of a plethora of genetic conditions. The fact of its broad fields of using from rare genetical diseases till common conditions brings attention to this issue in medical sector. Accurate diagnosis through diagnostic testing is critical for determining the most effective treatment strategies. Many conditions require specific therapies that depend on precise identification of the underlying issue. For instance, in infectious diseases, diagnostic tests such as cultures or polymerase chain reaction (PCR) tests can identify the causative pathogen, enabling the prescription of targeted antibiotics or antivirals. An investigation that appeared in the Journal of Molecular Diagnostics revealed that the percentage of medical professionals who had integrated genetic diagnostic testing for hereditary cancers definitely had grown from 19.8% in 2014 to 38.4% in 2018.
Analysis by Technology:
- Cytogenetic Testing and Chromosome Analysis
- Biochemical Testing
- Molecular Testing
- DNA Sequencing
- Others
Molecular testing (DNA sequencing and others) leads the market with 45.2% of market share in 2025. Molecular testing portion cover PCR, NGS, and other filtered DNA related analysis terms. It is very practical as it can be applied for many sections, from detecting genetic changes that give rise to diseases, to exploring cancer genetics for the reason of targeted treatments, and to unearthing genetic predispositions for different conditions. The unique capability of molecular testing associated with the precision it offers has established the field of genetic diagnostics, individualized medicine, and research as a key area in molecular science, giving it a leading position in this field. A 2021 study evaluated various COVID-19 molecular tests, revealing a high sensitivity rate of 95.9% for minimizing false negatives, and a specificity rate of 97.2% to reduce false positives.
Analysis by Application:
Access the comprehensive market breakdown Request Sample
- Cancer Diagnosis
- Genetic Disease Diagnosis
- Cardiovascular Disease Diagnosis
- Others
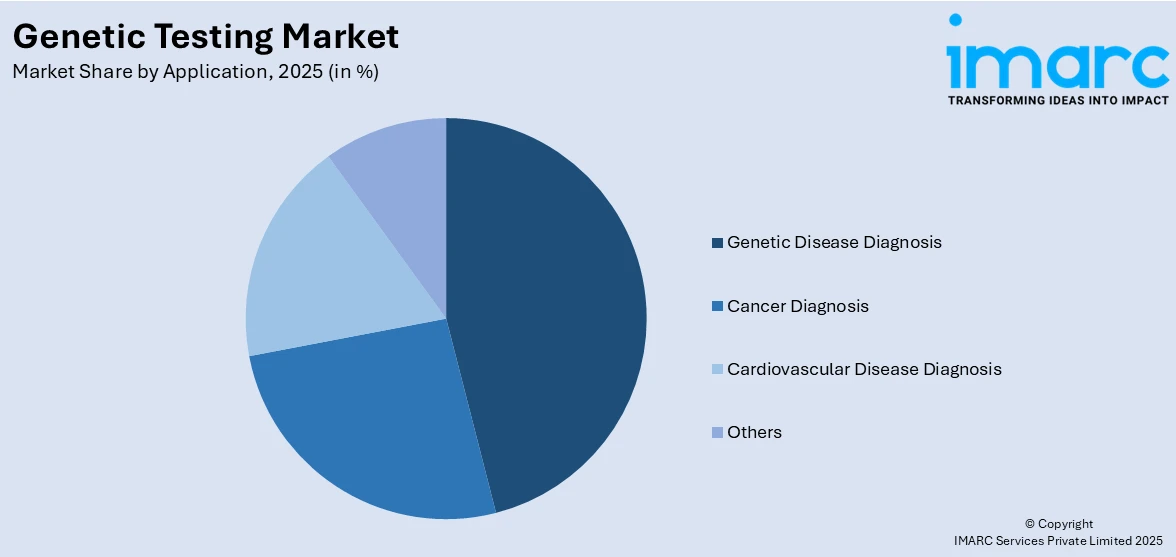
Genetic disease diagnosis leads the market with 40.5% of market share in 2025. Genetic testing is currently utilized to diagnose a large number of diseases that are affected by both single-gene and multiple-gene factors. Genetic disease diagnosis is essential for the early detection of diseases and prompt intervention that finally aid in the condition's management. This is driven by its rear-rooted importance in the field of medicine which is represented in the accurate diagnoses of the genetic diseases not only for treatment but also for family planning. Between 2012 and 2022, the US led in the expansion of genetic tests, primarily for clinical diagnosis, highlighting the need for mandatory reporting and international collaboration for a comprehensive understanding of global genetic testing. Using genetic analysis for cancer diagnosis implies finding out genetic mutations or alterations which cause cancer to develop. This portion is very vital for early tumor detection, diagnosis and controlling different categories of cancer. Moreover, the genetic diagnosis of cardiovascular diseases involves locating these genetic variants to establish an increased risk for such heart-related defects as congenital heart defects, various cardiomyopathies, and arrhythmias.
Regional Analysis:
To get more information on the regional analysis of this market Request Sample
- North America
- United States
- Canada
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
In 2025, North America accounted for the largest market share of 45.6%. North America is propelled by the top-notch healthcare infrastructure, high amount of money being spent on the healthcare sector and also the presence of top companies and research institutions. The region is driven by strong private investments, in the field of personalized medicine, large investments by governments on gene research and favorable policy decisions from the national government. Besides, genetic disorders as well as chronic diseases, with an increasing number of patients and an informed population willing to access testing services, enhance propel this company's leading position in the market. Moreover, companies are making genetic testing highly accessible to the general public, allowing people to purchase test kits online and conduct the process at home. The insights offered by these tests ranging from ancestry information to health risk assessments are generating widespread interest. This segment has particularly benefited from effective marketing strategies, increased awareness, and the growing demand for self-empowerment in health-related decisions. In 2024, Avalon GloboCare Corp., a leading organization in cell-based technology, precision diagnostics, and cellular therapy announced that Laboratory Services MSO, LLC (“LSM”) introduced a direct to consumer (D2C) and non-invasive DNA testing kit for potential predisposition to opioid addiction. These kits are available in the US for effortless at home sample collection.
Genetic Testing Regional Takeaways:
United States Genetic Testing Market Analysis
The United States accounts for 82.00% of the market share in North America. The US genetic testing market is driven by a combination of several factors. One of the key drivers is the rising prevalence of infectious diseases such as tuberculosis, HIV, and influenza, which increases the demand for effective detection and treatment solutions. The well-established healthcare infrastructure and government funding for research further support this demand, creating a favorable environment for market expansion. Additionally, the decision by UnitedHealthcare, the largest insurer in the U.S., to cover genetic tests for over 27 Million individuals since October 2019 is expected to prompt other insurers to follow suit, enhancing market accessibility. These favorable reimbursement trends help reduce the time spent finding optimal treatments, lower the costs of ineffective drugs, and contribute to the overall growth of the genetic testing market. Moreover, the economic burden of chronic diseases, such as cardiovascular conditions, which costs the U.S. healthcare system an estimated USD 320 Billion annually, according to the CDC, is driving a stronger emphasis on early diagnosis and preventive care, further accelerating the market's expansion.
Asia Pacific Genetic Testing Market Analysis
The Asia-Pacific genetic testing market is growing on account of a combination of factors including increasing awareness of the role of genetic testing in addressing public health challenges coupled with advancements in preventive care, as genetic testing helps identify risks for chronic diseases and hereditary disorders. With rapid urbanization and improved healthcare infrastructure, access to affordable genetic testing is expanding. Also, according to research by Bain, Asia-Pacific healthcare spending is expected to exceed 20% of global expenditure by 2030, with a notable 58% of Gen Z expressing willingness to invest in better health outcomes, underscoring the rising demand for personalized care. Public-private partnerships and culturally tailored awareness campaigns are also playing a key role in accelerating innovation, reducing stigma, and driving the acceptance of genetic health services in the region.
Europe Genetic Testing Market Analysis
The adoption of genetic testing in Europe is being driven by the advancements in patient care, particularly through its incorporation with personalized medicine. Genetic insights are also driving the development of targeted therapies for conditions such as neurodegenerative and autoimmune diseases, improving precision treatment. In oncology, genetic testing is identifying mutations that facilitate personalized cancer treatments, which in turn leads to better prognoses. According to the European Union, approximately 5.9 Million cancer patients were discharged from EU hospitals in 2021, with Austria leading at 2,584 discharges per 100,000 inhabitants. Apart from this, the developments in genetic testing are also enhancing cancer care by tailoring interventions to individual genomic profiles, ensuring improved treatment and better outcomes. Additionally, the increasing accessibility of genetic counseling services is also supporting individuals with hereditary health concerns. These collaborative efforts among healthcare providers, governments, and research institutions are thus fostering standardized protocols, ensuring the quality and accuracy of genetic tests. These factors are collectively driving growth in the European genetic testing market.
Latin America Genetic Testing Market Analysis
Increased access to genetic testing in Latin America is significantly driving the genetic testing market in the region primarily by addressing disparities in healthcare delivery and outcomes. Also, the health expenditure in the region grew at an annual rate of 4.9% from 2010-2019, yet remaining below the OECD average, such data presents a lucrative market for genetic testing which has emerged as a cost-effective solution to enhance public health programs. By enabling early detection of genetic diseases, genetic testing also allows for proactive management, leading to improved survival rates and better health outcomes. Additionally, expanding genetic services in newborn screening programs is preventing lifelong complications from treatable conditions, which in turn is fostering greater health equity in the region. Thus, the growing awareness and improved affordability are encouraging wider adoption of genetic testing, empowering individuals to manage genetic health and enhancing overall health literacy.
Middle East and Africa Genetic Testing Market Analysis
The expansion of the genetic testing market in the MENA region is relative to significantly improving the healthcare sector by adopting earlier diagnosis and treatment of inherited conditions. This technology plays a crucial role in population-specific research, identifying genetic variants that are more common in particular communities. Moreover, the high consanguinity rates ranging from 25-60%, and genetic disorders such as autosomal recessive syndromes, hemoglobinopathies, and Down syndrome being more prevalent in the region, the market is further propelling in the region. Also, prenatal testing, supported by genetic testing, is enhancing maternal and fetal health outcomes by reducing the occurrence of these genetic conditions.
Competitive Landscape:
Market’s key players adopt different strategies to achieve fundamental objective of strengthening their market share. This include is investing heavily in research and development to innovation and evolution of genetic testing technologies, especially in the direction of next-generation sequencing (NGS) and CRISPR. They also engage in the establishment of partnerships and collaboration with other biotech companies, academic institutions, as well as healthcare providers to help them grow in their services portfolio and geographical reach. Besides this, manufacturers are making efforts to get the new devices approved by the regulator agencies to be in compliance with regulations and guarantee the patient safety. Additionally, direct-to-consumer (D2C) services have been developed with the aim of leveraging people’s growing demand for personalized healthcare and genetic wellness. In 2024, LetsGetChecked announced the launch of myGeneticScreen, which is designed to provide patients with important and actionable insights into their predisposition for few hereditary conditions with end-to-end support, including personalized or customized health reports and access to genetic counselling.
The report provides a comprehensive analysis of the competitive landscape in the genetic testing market with detailed profiles of all major companies, including:
- 23andme Inc.
- Ambry Genetics Corporation (Konica Minolta Healthcare Americas Inc.)
- Bio-RAD Laboratories Inc.
- Cepheid (Danaher Corporation)
- Eurofins Scientific
- Illumina Inc.
- Invitae Corporation
- Luminex Corporation (DiaSorin)
- Myriad Genetics Inc.
- QIAGEN
- Quest Diagnostics
- Thermo Fisher Scientific
Latest News and Developments:
- March 2024: Nucleus Genomics introduced a whole-genome sequencing test to public. This test analyzes an individual's entire genetic makeup, providing detailed insights for tailored healthcare solutions.
- February 2024: The joint venture between Sysmex Corporation and Hitachi High-Tech Corporation to develop advanced genetic testing systems using capillary electrophoresis sequencers is set to enhance accessibility to genetic testing in untapped economies, addressing growing healthcare needs.
Genetic Testing Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Types Covered | Predictive and Presymptomatic Testing, Carrier Testing, Prenatal and Newborn Testing, Diagnostic Testing, Pharmacogenomic Testing, Others |
| Technologies Covered |
|
| Applications Covered | Cancer Diagnosis, Genetic Disease Diagnosis, Cardiovascular Disease Diagnosis, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | 23andme Inc., Ambry Genetics Corporation (Konica Minolta Healthcare Americas Inc.), Bio-RAD Laboratories Inc., Cepheid (Danaher Corporation), Eurofins Scientific, Illumina Inc., Invitae Corporation, Luminex Corporation (DiaSorin), Myriad Genetics Inc., QIAGEN, Quest Diagnostics, Thermo Fisher Scientific, etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the genetic testing market from 2020-2034.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the global genetic testing market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the genetic testing industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Key Questions Answered in This Report
Genetic testing is a medical procedure that analyzes an individual's DNA to identify genetic variations or mutations. These tests help diagnose genetic disorders, predict disease risks, guide treatment decisions, and provide valuable insights into an individual's genetic predispositions, enabling personalized medicine and preventive care.
The genetic testing market was valued at USD 21.4 Billion in 2025.
IMARC estimates the global genetic testing market to exhibit a CAGR of 8.93% during 2026-2034.
Key factors driving the global genetic testing market include advancements in genetic testing technologies, the increasing prevalence of genetic disorders and chronic diseases, growing demand for personalized medicine, rising awareness about reproductive health, and supportive government initiatives promoting genomics research and diagnostics.
In 2025, prenatal and newborn testing represented the largest segment by type, driven by its growing adoption in assessing chromosomal abnormalities and genetic conditions in early development stages.
Molecular testing leads the market by technology owing to its high precision, applicability in detecting genetic mutations, and extensive use in oncology and personalized medicine.
The genetic disease diagnosis segment is the leading application segment, driven by its critical role in identifying hereditary conditions and improving early intervention and treatment outcomes.
On a regional level, the market has been classified into North America, Asia Pacific, Europe, Latin America, and Middle East and Africa, wherein North America currently dominates the global market.
Some of the major players in the global genetic testing market include 23andme Inc., Ambry Genetics Corporation (Konica Minolta Healthcare Americas Inc.), Bio-RAD Laboratories Inc., Cepheid (Danaher Corporation), Eurofins Scientific, Illumina Inc., Invitae Corporation, Luminex Corporation (DiaSorin), Myriad Genetics Inc., QIAGEN, Quest Diagnostics, Thermo Fisher Scientific, etc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












