
India Biopsy Devices Market Size, Share, Trends and Forecast by Procedure Type, Product, Application, Guidance Technique, End User, and Region, 2026-2034
India Biopsy Devices Market Summary:
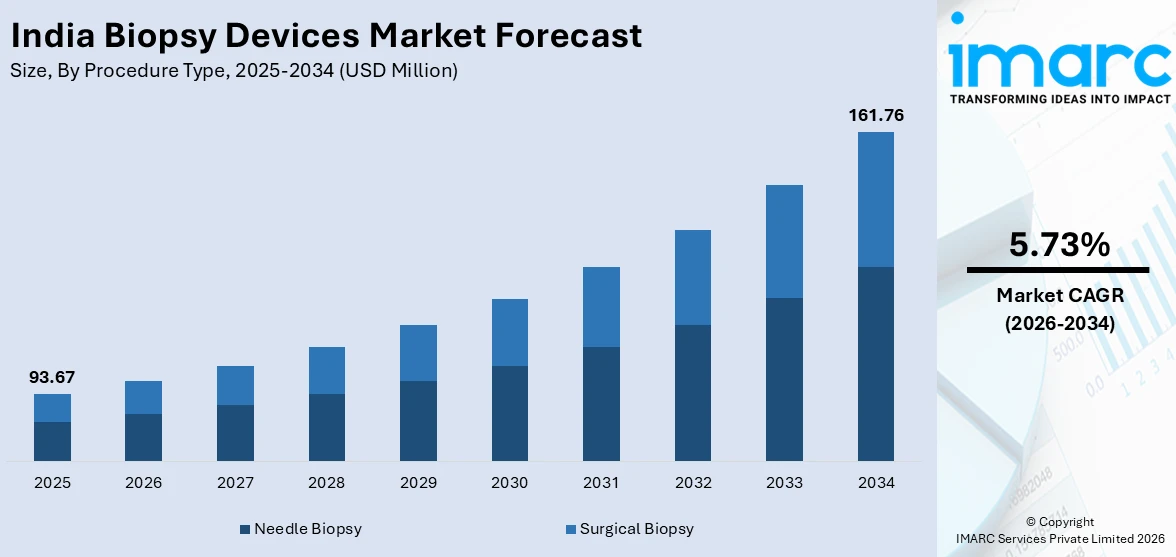
The India biopsy devices market size was valued at USD 93.67 Million in 2025 and is projected to reach USD 161.76 Million by 2034, growing at a compound annual growth rate of 5.73% during 2026-2034.
The India biopsy devices market is experiencing sustained growth driven by escalating cancer prevalence, expanding healthcare infrastructure, and a higher preference for minimally invasive diagnostic procedures. Advances in imaging-guided biopsy techniques, coupled with increased awareness about early cancer detection, are reshaping diagnostic workflows across hospitals, clinics, and research institutes. Government support for oncology programs and healthcare modernization is further strengthening the India biopsy devices market share.
Key Takeaways and Insights:
- By Procedure Type: Needle biopsy dominates the market with a share of 62.3% in 2025, driven by its minimally invasive nature, widespread clinical acceptance, and effectiveness across multiple tissue types for early cancer diagnosis.
- By Product: Biopsy needles lead the market with a share of 32.4% in 2025, owing to their versatility, precision, and critical role in enabling accurate tissue sampling across a wide range of oncological applications.
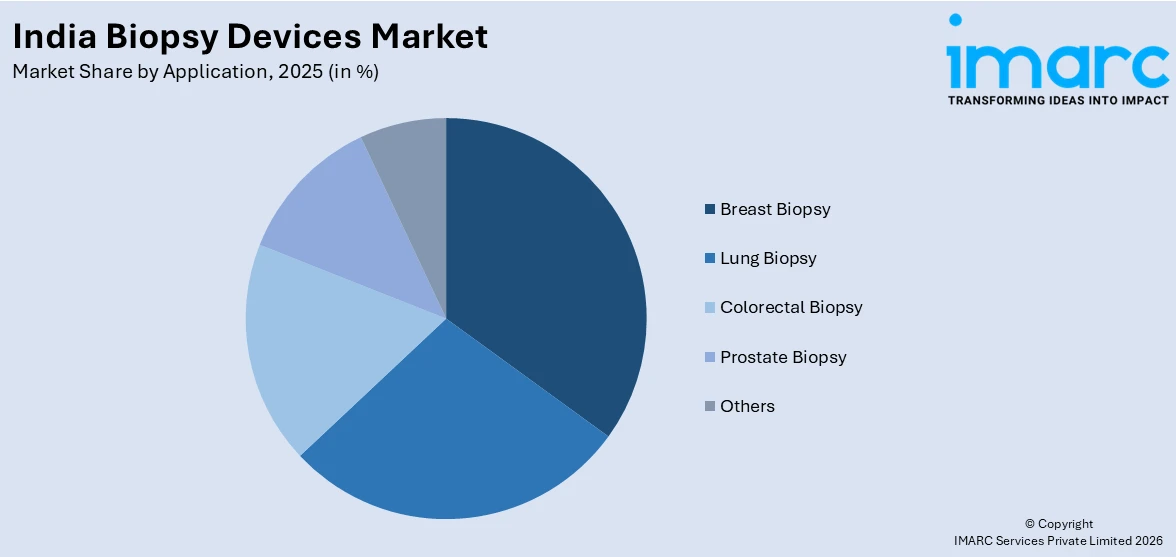
- By Application: Breast biopsy represents the largest segment with a market share of 28.5% in 2025, reflecting the high incidence of breast cancer in India and the growing clinical emphasis on early tissue-based diagnosis for improved treatment outcomes.
- By Guidance Technique: Ultrasound-guided biopsy dominates the market with a share of 42.6% in 2025, as real-time visualization capabilities enable precise needle placement, improving procedural accuracy and patient safety across oncology settings.
- By End User: Hospitals and clinics lead the market with a share of 55.2% in 2025, supported by their advanced diagnostic infrastructure, trained oncology specialists, and high patient footfall for cancer-related procedures.
- By Region: West and Central India represents the largest segment with a market share of 31.5% in 2025, due to the concentration of tertiary care facilities, established oncology centers, and superior healthcare infrastructure across Maharashtra and Gujarat.
- Key Players: The India biopsy devices market features a semi-consolidated competitive landscape, with global medical device manufacturers and domestic players competing through product innovation, precision technologies, and expanded hospital partnerships.
To get more information on this market Request Sample
The India biopsy devices market is being driven by a combination of demographic shifts, technological advancements, and evolving individual preferences. Increasing awareness about health, wellness, and lifestyle products is prompting higher demand across multiple sectors, while urbanization and rising disposable incomes are fueling usage of premium goods and services. According to government projections, India’s senior citizen population is expected to reach approximately 230 million by 2036, accounting for nearly 15% of the total population. This significant demographic shift is expected to create substantial opportunities in healthcare, retirement planning, insurance, and leisure industries. Moreover, innovations in digital platforms and e-commerce are enabling greater accessibility and convenience, reshaping purchasing patterns and market dynamics. Businesses are also leveraging data analytics and personalized marketing strategies to better target users and anticipate emerging trends. Collectively, these factors are fostering market growth and encouraging investment across sectors, signaling a vibrant and evolving economic landscape.
India Biopsy Devices Market Trends:
Automation and Workflow Efficiency Driving Device Adoption
The India biopsy devices market is increasingly influenced by the need for automation and streamlined clinical workflows, enabling faster, safer, and more precise tissue sampling. Automated systems reduce procedural complexity, minimize operator errors, and enhance patient comfort, which is critical in high-volume diagnostic centers. For instance, in 2024, Mammotome launched the AutoCore Single Insertion Core Biopsy System, the first automated spring-loaded device for breast biopsies. Its single-insertion, one-handed operation and touchless tissue transfer reduce procedural steps while improving accuracy. This device illustrates the market trend of integrating automation to improve clinical efficiency and patient experience, encouraging wider adoption of minimally invasive biopsy technologies across hospitals and specialty diagnostic centers in India.
Portability and Accessibility Expanding Minimally Invasive Care
The growing emphasis on portable and compact biopsy devices reflects the need to expand diagnostic access beyond major urban hospitals. Lightweight, all-in-one systems allow clinicians to provide high-quality, minimally invasive procedures in smaller clinics and tier II/III cities, bridging care gaps. In 2025, Univlabs Technologies introduced India’s first portable GynoStart Suite for diagnostic hysteroscopy, supporting endometrial biopsies, infertility assessments, and IUD localization. The device’s compact 4K imaging and integrated workflow make advanced gynecological diagnostics accessible in clinics that previously lacked such technology. This example highlights how portability and integrated design are key market drivers, enabling broader adoption of minimally invasive biopsy procedures nationwide.
AI and Multi-Modal Imaging Enhancing Diagnostic Precision
Increasing demand for precision oncology and early cancer detection is driving the adoption of AI-enabled, multi-modal biopsy systems. These devices improve accuracy, reduce complications, and allow clinicians to detect malignancies earlier, aligning with international standards. In 2024, Sir H N Reliance Foundation Hospital in Mumbai introduced Maharashtra’s first MR Fusion Prostate Biopsy machine, combining MRI, ultrasound, and AI guidance. This minimally invasive device increased detection rates to 90% while avoiding rectal entry and lowering infection risks. Its launch demonstrates the market trend toward integrating AI and advanced imaging into biopsy devices, improving patient outcomes and positioning India as a leader in precision cancer diagnostics.
Market Outlook 2026-2034:
The India biopsy devices market is witnessing growth during the forecast period due to the rising prevalence of cancer and other chronic diseases, increasing awareness regarding early diagnosis, and growing adoption of minimally invasive procedures. The market generated a revenue of USD 93.67 Million in 2025 and is projected to reach a revenue of USD 161.76 Million by 2034, growing at a compound annual growth rate of 5.73% from 2026-2034. Advancements in imaging technologies, expansion of diagnostic infrastructure, and supportive government healthcare initiatives are further contributing to increased demand for biopsy devices across the country.
India Biopsy Devices Market Report Segmentation:
| Segment Category | Leading Segment | Market Share |
|---|---|---|
|
Procedure Type |
Needle Biopsy |
62.3% |
|
Product |
Biopsy Needles |
32.4% |
|
Application |
Breast Biopsy |
28.5% |
|
Guidance Technique |
Ultrasound-guided Biopsy |
42.6% |
|
End User |
Hospitals and Clinics |
55.2% |
|
Region |
West and Central India |
31.5% |
Procedure Type Insights:
- Surgical Biopsy
- Needle Biopsy
Needle biopsy exhibits a clear dominance with a 62.3% share of the total India biopsy devices market in 2025.
Needle biopsy dominated the market due to its minimally invasive nature, high accuracy, and cost effectiveness compared to surgical alternatives. It enables efficient tissue sampling with reduced patient discomfort, shorter recovery time, and lower risk of complications. The procedure is widely used for diagnosing various cancers and chronic conditions, making it a preferred choice among healthcare providers. Increasing awareness regarding early disease detection and the growing adoption of outpatient procedures further support its usage. Additionally, the availability of skilled professionals and compatible imaging technologies enhances its accessibility across healthcare facilities in India.
The segment’s dominance is further driven by advancements in imaging guided techniques that improve precision and procedural outcomes. Needle biopsy procedures are increasingly integrated with ultrasound, CT, and MRI guidance, enabling accurate targeting of affected tissues. Rising demand for faster diagnosis and reduced hospital stays is encouraging the shift toward minimally invasive procedures. Expansion of diagnostic centers and oncology services across urban and semi urban regions also contributes to higher adoption. Moreover, lower procedural costs and improved patient acceptance strengthen its preference, ensuring that needle biopsy remains the leading procedure type in the market.
Product Insights:
- Biopsy Guidance Systems
- Needle Based Biopsy Guns
- Biopsy Needles
- Biopsy Forceps
- Others
Biopsy needles dominate with a market share of 32.4% of the total India biopsy devices market in 2025.
Biopsy needles lead the market due to their essential role in performing minimally invasive diagnostic procedures. These devices are widely used across various biopsy types, including breast, lung, and liver, due to their precision and efficiency in tissue sampling. Their cost effectiveness and ease of use make them highly accessible across hospitals and diagnostic centers. Additionally, increasing demand for early and accurate diagnosis of cancer and other chronic diseases is driving their widespread adoption. The availability of different needle types designed for specific procedures further supports their dominance in the market.
The segment’s leadership is also supported by continuous advancements in needle design, improving accuracy, safety, and patient comfort. Innovations such as automated and image guided biopsy needles enhance procedural efficiency and reduce complications. The growing preference for minimally invasive techniques further increases their utilization across healthcare settings. Additionally, expansion of diagnostic infrastructure and rising awareness about early disease detection contribute to higher procedure volumes. Strong demand from both public and private healthcare facilities, along with consistent supply and affordability, ensures that biopsy needles remain the leading product segment in the India biopsy devices market.
Application Insights:
Access the comprehensive market breakdown Request Sample
- Breast Biopsy
- Lung Biopsy
- Colorectal Biopsy
- Prostate Biopsy
- Others
Breast biopsy leads with a market share of 28.5% of the total India biopsy devices market in 2025.
Breast biopsy dominates the market attributed to the rising prevalence of breast cancer and increasing awareness regarding early diagnosis. The growing screening programs and initiatives promoting regular health checkups are significantly increasing the number of breast cancer detections, thereby driving the demand for biopsy procedures. Healthcare providers are increasingly focusing on accurate and timely diagnosis to improve treatment outcomes. Additionally, advancements in imaging technologies and minimally invasive biopsy techniques are making breast biopsies safer and more efficient. The availability of specialized diagnostic centers further supports higher procedure volumes across both urban and semi urban regions in India.
The dominance of breast biopsy is also driven by improving healthcare infrastructure and increasing access to advanced diagnostic tools. Hospitals and diagnostic centers are adopting modern biopsy technologies to enhance precision and patient comfort. Rising awareness among women regarding preventive healthcare and self-examination practices is contributing to early detection rates. Government and private sector initiatives supporting cancer awareness campaigns further boost demand for diagnostic procedures. Moreover, the growing number of trained healthcare professionals and expansion of oncology services strengthen the adoption of breast biopsy techniques, ensuring its leading position in the market.
Guidance Technique Insights:
- Ultrasound-guided Biopsy
- Stereotactic-guided Biopsy
- MRI-guided Biopsy
- Others
Ultrasound-guided biopsy exhibits a clear dominance with a 42.6% share of the total India biopsy devices market in 2025.
Ultrasound guided biopsy is the leading segment in the market owing to its real time imaging capability, cost effectiveness, and widespread availability across healthcare settings. This technique allows precise needle placement while minimizing complications, making it highly preferred by clinicians. It is widely used for diagnosing conditions in organs, such as the liver, breast, and thyroid. Additionally, ultrasound system is more accessible compared to other imaging modalities, especially in mid-sized hospitals and diagnostic centers. The non-invasive nature and lack of radiation exposure further enhance its adoption across both urban and semi urban healthcare facilities in India.
The dominance of ultrasound guided biopsy is also driven by its ability to deliver faster procedures and improved patient comfort. It enables clinicians to perform minimally invasive biopsies with greater accuracy, reducing the need for repeat procedures. The growing awareness about early disease detection and increasing screening programs are further driving its usage. Technological advancements in ultrasound imaging, including improved resolution and portability, enhance procedural efficiency. Moreover, lower operational costs and ease of use make it suitable for a wide range of healthcare providers, reinforcing its leading position in the market.
End User Insights:
- Hospitals and Clinics
- Academic and Research Institutes
- Others
Hospitals and clinics dominate with a market share of 55.2% of the total India biopsy devices market in 2025.
Hospitals and clinics hold the biggest market share driven by their central role in providing comprehensive diagnostic and treatment services. These facilities handle a high volume of patients requiring biopsy procedures for accurate disease detection, particularly for cancer and other chronic conditions. Availability of advanced medical equipment, skilled healthcare professionals, and specialized departments enables efficient use of biopsy devices. Additionally, hospitals and clinics are often the first point of care, where initial diagnosis and follow up procedures are conducted. Increasing patient preference for institutional healthcare further supports their dominance in the market.
The segment’s leadership is also influenced by continuous expansion of healthcare infrastructure and rising investments in both public and private hospitals. These facilities are increasingly adopting advanced biopsy technologies to improve diagnostic accuracy and patient outcomes. Integration of modern imaging and minimally invasive procedures enhances efficiency and reduces recovery time. Government initiatives promoting early disease detection and screening programs further increase procedure volumes in hospitals and clinics. Moreover, strong referral networks and access to specialized care contribute to higher utilization rates, reinforcing their position as the leading end users in the market.
Regional Insights:
- North India
- West and Central India
- South India
- East India
West and Central India lead with a market share of 31.5% of the total India biopsy devices market in 2025.
West and Central India represent the largest segment because of the strong presence of advanced healthcare infrastructure and leading hospitals across major cities. Regions, such as Mumbai, Pune, Ahmedabad, and Nagpur, host well established diagnostic centers and multi-specialty hospitals equipped with modern medical technologies. Higher patient awareness and access to quality healthcare services contribute to increased diagnostic procedures, including biopsies. Additionally, the presence of skilled healthcare professionals and specialists supports accurate diagnosis and treatment planning. The growing investments in healthcare facilities and expansion of private hospitals further strengthen the region’s leading position.
The region also benefits from the rising prevalence of cancer and other chronic diseases, increasing demand for early and precise diagnostic procedures. West and Central India benefit from strong pharmaceutical and medical device ecosystems, encouraging adoption of advanced biopsy technologies. Improved healthcare accessibility and expansion of diagnostic networks across urban and semi urban areas support higher procedure volumes. Moreover, government initiatives to enhance cancer screening and early detection programs contribute to increased usage of biopsy devices. Strong distribution networks and collaborations between healthcare providers and device manufacturers further reinforce the region’s leadership in the India biopsy devices market.
Market Dynamics:
Growth Drivers:
Why is the India Biopsy Devices Market Growing?
High-Quality Core Instruments Supporting Tissue Integrity
The increasing demand for instruments capable of obtaining larger, intact tissue samples for accurate diagnosis is impelling the market growth. Such tools enhance reliability, reduce repeat procedures, and improve patient outcomes across oncology, pathology, and surgical diagnostics. In 2023, Terumo India launched the BIOPINCE™ Ultra Full Core Biopsy Instrument, engineered for precise, high-quality core collection. Its user-friendly design supports a wide range of tissue sampling procedures. This example underscores the broader trend of adopting specialized, high-quality core biopsy devices to meet the growing need for diagnostic accuracy and standardized clinical protocols in Indian healthcare facilities.
Expansion of Healthcare Infrastructure
Rising investments in hospitals, specialty clinics, diagnostic centers, and ambulatory care facilities are fueling sustained demand for advanced biopsy devices across India. Expanding public and private healthcare expenditure, particularly in tier II and tier III cities, is enhancing access to minimally invasive diagnostic procedures and improving early disease detection. Reflecting this commitment, in 2024, the Prime Minister of India inaugurated health sector projects worth over INR 12,850 Crore, including new AIIMS facilities, medical colleges, and critical care blocks, alongside expanded Ayushman Bharat coverage and support for domestic medical device manufacturing under the PLI scheme. These initiatives are strengthening infrastructure and enabling broader adoption of sterile, high-precision biopsy tools, supporting long-term market growth.
Rising Prevalence of Cancer and Chronic Diseases
A key factor driving the India biopsy devices market is the rising prevalence of cancer and other chronic diseases, which increases the demand for precise diagnostic interventions. Early and accurate detection is becoming essential for effective treatment planning, prompting healthcare providers to invest in advanced biopsy technologies. Heightened awareness among physicians and patients regarding timely diagnosis further reinforces this trend. In 2024, the Indian Council of Medical Research (ICMR) reported in The Journal of the American Medical Association (JAMA) that India recorded an estimated 1.56 million new cancer cases, highlighting the growing patient population requiring tissue sampling for histopathological analysis. Consequently, reliable biopsy devices are increasingly indispensable in modern clinical settings, supporting improved outcomes and strengthening market growth across the country.
Market Restraints:
What Challenges the India Biopsy Devices Market is Facing?
High Procurement Costs of Advanced Biopsy Systems
Advanced biopsy platforms, including MRI-guided and robotic-assisted systems, involve significant capital expenditure that remains prohibitive for many secondary and tier-2 hospitals in India. Import dependency on high-end biopsy equipment, combined with applicable customs duties and distribution costs, elevates overall device pricing. This cost barrier restricts adoption primarily to large metropolitan hospitals and specialized cancer centers, limiting broader market penetration in smaller clinical settings across the country.
Shortage of Trained Oncologists and Radiologists in Smaller Cities
The effective use of image-guided biopsy devices requires specialized clinical training in radiology, interventional oncology, and pathology. India continues to face a significant shortage of trained oncologists and interventional radiologists, particularly outside major metropolitan centers. This skills gap limits the deployment of advanced biopsy technologies in tier-2 and tier-3 cities, constraining overall market expansion despite available device supply and growing patient demand in these regions.
Import Dependency and Evolving Regulatory Compliance Requirements
India's medical device market relies heavily on imported biopsy products from global manufacturers, creating vulnerabilities related to foreign exchange fluctuations, supply chain disruptions, and import duty variations. Regulatory requirements under the Central Drugs Standard Control Organization add compliance complexity for manufacturers and distributors alike. Evolving device registration norms and post-market surveillance obligations increase operational burdens, particularly for smaller domestic distributors serving tier-2 and rural healthcare markets.
Competitive Landscape:
The India biopsy devices market exhibits a semi-consolidated competitive structure, characterized by the presence of established global medical device manufacturers alongside a growing cohort of domestic distributors and equipment providers. Leading international companies leverage expansive product portfolios encompassing biopsy needles, guidance systems, needle-based biopsy guns, forceps, and accessories to maintain strong institutional relationships with major hospitals and diagnostic chains. Competition is increasingly centered on technological differentiation, with firms investing in advanced ultrasound-compatible products, minimally invasive device designs, and digital pathology integration. Domestic players compete primarily on cost efficiency, after-sales service quality, and last-mile distribution capabilities in tier-2 and tier-3 markets. Strategic alliances with hospital networks, participation in government procurement programs, and the growing importance of product certifications under India's medical device regulatory framework are shaping competitive positioning. Innovation in single-use, disposable biopsy devices is emerging as a key differentiator in the current competitive landscape.
Recent Developments:
- January 2026: Apollo Athenaa Women’s Cancer Centre in Delhi has introduced MRI-guided Vacuum-Assisted Breast Biopsy (VABB) to enhance early breast cancer detection, particularly in younger women with dense breast tissue. This advanced diagnostic tool enables precise, minimally invasive sampling, allowing cancers that often go undetected in mammography to be identified at Stage 0. By integrating high-resolution MRI with AI-supported protocols, the hospital has reduced scan times while improving accuracy and clinical confidence in biopsy results.
- August 2025: Gurugram-based Univlabs Technologies launched GynoStart, India’s first portable gynecology endoscopy system, designed as an all-in-one diagnostic tool for smaller clinics and hospitals. The system integrated a 4K camera, LED light source, display, and recording unit into a compact, transportable case, enabling rapid evaluation of uterine health, infertility workups, and guided endometrial biopsies.
India Biopsy Devices Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Million USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Predictive Market Assessment:
|
| Procedure Types Covered | Surgical Biopsy, Needle Biopsy |
| Products Covered | Biopsy Guidance Systems, Needle Based Biopsy Guns, Biopsy Needles, Biopsy Forceps, Others |
| Applications Covered | Breast Biopsy, Lung Biopsy, Colorectal Biopsy, Prostate Biopsy, Others |
| Guidance Techniques Covered | Ultrasound-guided Biopsy, Stereotactic-guided Biopsy, MRI-guided Biopsy, Others |
| End Users Covered | Hospitals and Clinics, Academic and Research Institutes, Others |
| Regions Covered | North India, West and Central India, South India, East India |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Frequently Asked Questions About the India Biopsy Devices Market Report
The India biopsy devices market size was valued at USD 93.67 Million in 2025.
The India biopsy devices market is expected to grow at a compound annual growth rate of 5.73% during 2026-2034 to reach USD 161.76 Million by 2034.
Needle biopsy holds the largest share at 62.3% in 2025, owing to its minimally invasive nature, high diagnostic accuracy, and widespread clinical adoption across oncology and radiology departments throughout India.
Key factors driving the India biopsy devices include the demand for portable, compact systems that expand access to minimally invasive procedures, exemplified by Univlabs Technologies’ 2025 GynoStart Suite, a 4K diagnostic hysteroscopy device enhancing care in smaller clinics and tier II/III cities.
Major challenges include high procurement costs of advanced biopsy systems, shortage of trained oncologists and radiologists in smaller cities, import dependency on specialized equipment, evolving regulatory compliance requirements, and limited affordability of premium devices in rural and semi-urban healthcare settings.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












