
Interbody Fusion Cage Market Report by Product Type (Lumbar Cage, Cervical Cage, Thoraco-Lumbar Cage, Thoracic Cage), Surgery (Anterior, Posterior, Lateral, Transforaminal), End User (Hospitals and Clinics, Ambulatory Surgical Center, and Others), and Region 2026-2034
Interbody Fusion Cage Market Size:
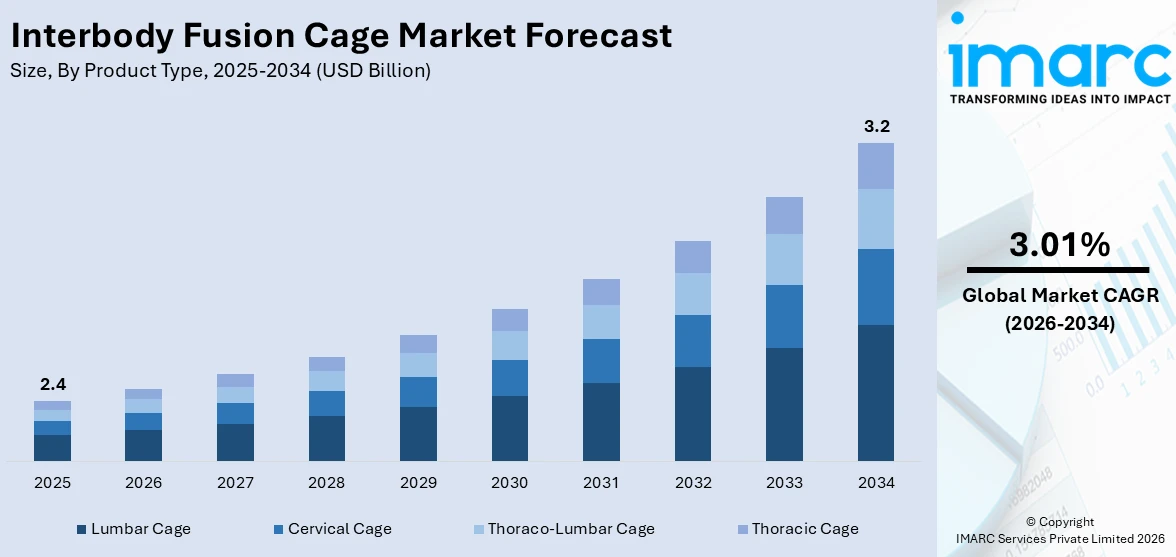
The global interbody fusion cage market size reached USD 2.4 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 3.2 Billion by 2034, exhibiting a growth rate (CAGR) of 3.01% during 2026-2034. The market is experiencing steady growth driven by the rapid technological advancements in the formulation of cage material and design, the increasing geriatric population that is more prone to developing spinal cord injuries, and the rising product demand for spine reconstruction during surgeries.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 2.4 Billion |
|
Market Forecast in 2034
|
USD 3.2 Billion |
| Market Growth Rate 2026-2034 | 3.01% |
Interbody Fusion Cage Market Analysis:
- Market Growth and Size: The global market is experiencing robust growth, driven by various factors contributing to increased demand. The market size has expanded significantly over recent years, and this trend is expected to continue in the foreseeable future.
- Major Market Drivers: Key drivers include the rising prevalence of degenerative spine conditions, an aging population, and an increasing number of spinal surgeries globally. Additionally, a growing awareness of minimally invasive surgical procedures and advancements in healthcare infrastructure contribute to the market growth.
- Technological Advancements: Technological advancements in interbody fusion cage design and materials are playing a crucial role in market growth. Innovations such as 3D-printed cages, bioresorbable materials, and improved surgical techniques are enhancing the efficacy and safety of interbody fusion procedures, driving adoption rates.
- Industry Applications: These cages find applications across a spectrum of spine-related conditions, including degenerative disc disease, spinal stenosis, and spondylolisthesis. The versatility of these devices in addressing various spinal disorders is a significant factor contributing to their widespread use.
- Key Market Trends: Key trends include an increasing shift towards outpatient and minimally invasive procedures, patient-specific implants for personalized treatment, and a focus on biocompatible materials. Additionally, the adoption of navigation technologies in spinal surgeries is gaining traction.
- Geographical Trends: Geographically, the market exhibits varying trends influenced by factors such as healthcare infrastructure development, regulatory landscapes, and the prevalence of spine-related disorders. Emerging economies are witnessing accelerated growth due to an increased focus on healthcare, while developed regions are characterized by a higher adoption rate of advanced technologies.
- Competitive Landscape: The market features a competitive landscape with several key players vying for market share. Companies are actively involved in research and development activities to introduce novel products and gain a competitive edge. Partnerships, mergers, and acquisitions are common strategies to expand market presence.
- Challenges and Opportunities: Challenges include stringent regulatory requirements, reimbursement issues, and the high cost associated with advanced technologies. However, opportunities abound in addressing unmet medical needs, expanding into untapped markets, and collaborating with healthcare providers to enhance patient outcomes.
- Future Outlook: The future of the market appears promising, with sustained growth anticipated. As technological innovations continue and healthcare systems change, the market is likely to witness further expansion. The increasing focus on patient-centric solutions and the integration of data-driven approaches in spine surgery are expected to shape the market growth in the coming years.
To get more information on this market Request Sample
Interbody Fusion Cage Market Trends:
Technological advancements and innovation
The market is strongly influenced by continuous technological advancements and innovations in medical devices. As technology develops, manufacturers are developing these cages with improved materials, designs, and features. These innovations aim to enhance surgical outcomes, reduce recovery times, and address specific patient needs. For instance, the introduction of 3D printing technology has allowed for the creation of patient-specific interbody fusion cages, promoting better anatomical fit and integration. Additionally, the incorporation of advanced biomaterials with properties that mimic natural bone enhances fusion rates and overall implant success. Surgeons and patients alike are increasingly drawn to these advanced technologies, driving the growth of the market.
Rising incidence of spinal disorders and degenerative conditions
The prevalence of spinal disorders and degenerative conditions, such as degenerative disc disease and spondylolisthesis, is a significant factor fueling the demand for interbody fusion cages. As the global population ages, the incidence of these spinal conditions is increasing. Interbody fusion procedures provide a viable solution for stabilizing the spine, relieving pain, and restoring function. The aging population, coupled with lifestyle factors contributing to spinal issues, has led to a growing number of surgical interventions, thereby propelling the market forward.
Increasing minimally invasive surgical procedures
The trend toward minimally invasive surgery (MIS) has gained momentum in recent years, and this shift has positively impacted the market. Patients and surgeons prefer minimally invasive approaches due to advantages such as smaller incisions, reduced blood loss, and faster recovery times. Interbody fusion cages designed for use in minimally invasive procedures have become pivotal in facilitating these surgical techniques. Manufacturers are investing in the development of cage designs specifically tailored for MIS, contributing to the expansion of the market as more surgeons adopt and advocate for minimally invasive approaches in spinal surgeries.
Interbody Fusion Cage Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the market, along with forecasts at the global, regional, and country levels for 2026-2034. Our report has categorized the market based on product type, surgery, and end user.
Breakup by Product Type:
- Lumbar Cage
- Cervical Cage
- Thoraco-Lumbar Cage
- Thoracic Cage
Lumbar cage account for the majority of the market share
The report has provided a detailed breakup and analysis of the market based on the product type. This includes lumbar cage, cervical cage, thoraco-lumbar cage, and thoracic cage. According to the report, lumbar cage represented the largest segment.
The lumbar cage segment holds a predominant position in the market, constituting the majority of the market share. Lumbar cages are specifically designed for the lower back region, addressing prevalent conditions like degenerative disc disease and instability in the lumbar spine. The high prevalence of lumbar spine issues, coupled with the effectiveness of lumbar cages in providing stability and promoting fusion, contributes to their dominant market share. Surgeons frequently opt for lumbar cages in spinal fusion procedures, driving the growth of this segment.
On the other hand, cervical cages, designed for the neck region, represent another significant segment in the market. As cervical spine disorders and conditions gain attention, the demand for cervical cages is on the rise. These cages offer support to the cervical spine during fusion surgeries, addressing issues such as herniated discs and cervical instability.
Additionally, thoraco-lumbar cages bridge the lumbar and thoracic spine segments, catering to patients with conditions that span across these areas. This segment addresses a range of spinal disorders and deformities, providing stability and support for the thoraco-lumbar junction.
Furthermore, the thoracic cage segment focuses on the upper and mid-back region. While comprising a smaller share compared to lumbar cages, thoracic cages are crucial in addressing specific conditions affecting the thoracic spine, such as fractures and deformities. As advancements in spinal surgery continue, each segment plays a vital role in meeting the diverse needs of patients, contributing collectively to the overall growth of the market.
Breakup by Surgery:
- Anterior
- Posterior
- Lateral
- Transforaminal
Anterior holds the largest share of the industry
A detailed breakup and analysis of the market based on the surgery have also been provided in the report. This includes anterior, posterior, lateral, and transforaminal. According to the report, anterior accounted for the largest market share.
The market is segmented based on surgical approaches, with the anterior approach commanding the largest share in the industry. The anterior approach involves accessing the spine from the front of the body, allowing surgeons to reach the intervertebral disc space through the abdomen. This approach is commonly utilized in lumbar and cervical spinal fusion surgeries, offering direct access to the affected area and enabling the placement of interbody fusion cages with precision. The popularity of the anterior approach is attributed to its effectiveness in providing stability, promoting fusion, and minimizing disruption to the posterior elements of the spine.
On the other hand, the posterior approach, involving access from the back, is another significant segment. It is employed in various spinal surgeries, particularly for conditions requiring stabilization and fusion in the thoracic and lumbar regions. The posterior approach allows for decompression and fusion procedures, contributing to its substantial market presence.
Moreover, lateral and transforaminal approaches, while comprising smaller shares, play crucial roles in specific cases. The lateral approach involves accessing the spine from the side, facilitating minimally invasive procedures, while the transforaminal approach targets nerve roots through the intervertebral foramen. These approaches cater to different patient needs and surgical complexities, contributing to the overall diversity and growth of the interbody fusion cage market.
Breakup by End User:
Access the comprehensive market breakdown Request Sample
- Hospitals and Clinics
- Ambulatory Surgical Center
- Others
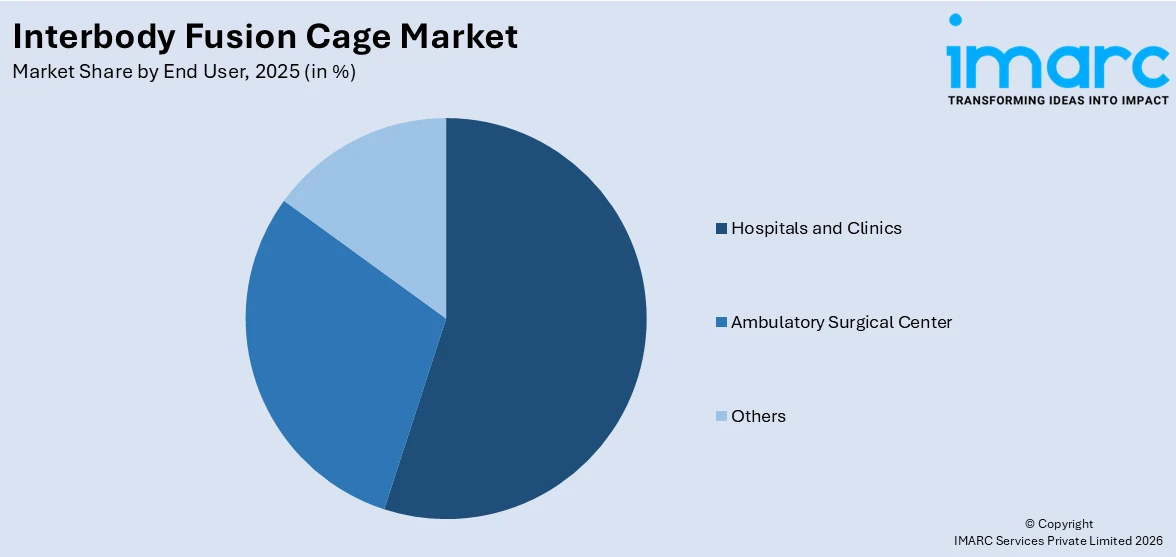
Hospitals and clinics represent the leading market segment
The report has provided a detailed breakup and analysis of the market based on the end user. This includes hospitals and clinics, ambulatory surgical center, and others. According to the report, hospitals and clinics represented the largest segment.
Hospitals and clinics represent a substantial portion of the market share. These healthcare facilities are key contributors to the demand for interbody fusion cages, given their role as primary settings for spinal surgeries and related procedures. The extensive infrastructure specialized surgical departments, and a higher volume of patients seeking spinal interventions contribute to the significant market presence of hospitals and clinics. The market dynamics across the segment reflect the diverse healthcare settings in which interbody fusion procedures are performed, contributing to the overall growth and accessibility of these interventions.
On the other hand, ambulatory surgical centers (ASCs) constitute another essential segment in the market. ASCs have gained popularity due to their ability to provide specialized surgical care in an outpatient setting. The streamlined and efficient nature of ASCs, coupled with the increasing trend toward outpatient procedures, makes them a favorable choice for certain spinal surgeries. As healthcare delivery models improve, ASCs play a vital role in shaping the landscape of the market, offering patients alternative and convenient options for surgical interventions.
Breakup by Region:
- North America
- United States
- Canada
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America leads the market, accounting for the largest interbody fusion cage market share
The market research report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America accounted for the largest market share.
North America holds a prominent position in the market, leading in market share. The region benefits from advanced healthcare infrastructure, a high prevalence of spinal disorders, and a significant focus on technological innovations. The presence of key market players, coupled with a well-established reimbursement framework, contributes to the dominance of North America in driving the growth of the market.
Europe represents a substantial market for these cages, characterized by a growing incidence of spinal conditions and an aging population. The region demonstrates a strong emphasis on research and development, leading to the introduction of innovative products. Factors such as increased awareness about spinal health and sophisticated healthcare systems contribute to the steady growth of the market in Europe.
The Asia-Pacific region is experiencing notable growth in the market, propelled by rising healthcare investments, a growing patient population, and increasing awareness of advanced spinal treatment options. Countries like China and India are witnessing a rise in demand for interbody fusion procedures due to lifestyle changes and an expanding middle-class population. The Asia-Pacific market is becoming a key focus for both established and emerging players in the industry.
Latin America is contributing to the global market, driven by improving healthcare infrastructure and a rising prevalence of spinal disorders. Economic development and increasing access to medical services in countries like Brazil and Mexico are influencing the market positively. While the market share may be comparatively smaller than other regions, Latin America presents opportunities for growth in the market.
The Middle East and Africa region show a growing interest in interbody fusion procedures, supported by advancements in healthcare facilities and a rising awareness of spinal health. The market in this region is changing, with governments investing in healthcare infrastructure and the growing availability of specialized surgical services contributing to the expansion of the market.
Leading Key Players in the Interbody Fusion Cage Industry:
The key players in the market are driving growth through strategic initiatives focused on research and development, mergers and acquisitions, and global market expansion. These companies invest significantly in R&D to innovate and introduce advanced cage designs, materials, and technologies. Mergers and acquisitions enable them to broaden their product portfolios, enhance market presence, and access new technologies swiftly. Moreover, these key players actively engage in partnerships with healthcare institutions and surgeons to gain valuable insights and ensure the alignment of their products with changing clinical needs. Expanding their global footprint is another strategy, as they tap into emerging markets with increasing healthcare infrastructure development. By combining these approaches, key players play a crucial role in shaping the market landscape, fostering growth, and addressing the changing demands of surgeons and patients worldwide.
The market research report has provided a comprehensive analysis of the competitive landscape. Detailed profiles of all major companies have also been provided. Some of the key players in the market include:
- Alphatec Spine, Inc.
- Aurora Spine Inc.
- B. Braun Melsungen AG
- Johnson & Johnson
- Medtronic Plc.
- NuVasive Inc
- Orthofix US LLC
- Precision Spine Inc.
- SpineArt SA
- Ulrich Medicals USA
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
Latest News:
- October 31, 2023: Alphatec Holdings, Inc. announced that the successful close of a $150 Million secondary offering on October 27, 2023, positions the Company to accelerate investment in the revenue-generating assets needed to capitalize on industry disruption.
- September 13, 2023: Aurora Spine Inc. announced the full release of its proprietary SiLO TFX MIS Sacroiliac Joint Fixation Device system.
- March 29, 2022: B. Braun Melsungen AG announced to divests Colombian provider business.
Interbody Fusion Cage Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Product Types Covered | Lumbar Cage, Cervical Cage, Thoraco-Lumbar Cage, Thoracic Cage |
| Surgeries Covered | Anterior, Posterior, Lateral, Transforaminal |
| End Users Covered | Hospitals and Clinics, Ambulatory Surgical Center, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Alphatec Spine, Inc., Aurora Spine Inc., B. Braun Melsungen AG, Johnson & Johnson, Medtronic Plc., NuVasive Inc, Orthofix US LLC, Precision Spine Inc., SpineArt SA, Ulrich Medicals USA, etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the interbody fusion cage market from 2020-2034.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the global interbody fusion cage market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the interbody fusion cage industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Key Questions Answered in This Report
The global interbody fusion cage market was valued at USD 2.4 Billion in 2025.
We expect the global interbody fusion cage market to exhibit a CAGR of 3.01% during 2026-2034.
The rising popularity of minimally invasive surgeries, along with the growing preference towards interbody fusion cage, as it aids in providing immediate structural support to the spine and restoring stability and alignment over traditional fusion methods, is primarily driving the global interbody fusion cage market.
The sudden outbreak of the COVID-19 pandemic had led to the postponement of elective spinal fusion surgical procedures to reduce the risk of the coronavirus infection upon hospital visits and interaction with medical equipment, thereby negatively impacting the global market for interbody fusion cage.
Based on the product type, the global interbody fusion cage market can be segmented into lumbar cage, cervical cage, thoraco-lumbar cage, and thoracic cage. Currently, lumbar cage holds the majority of the total market share.
Based on the surgery, the global interbody fusion cage market has been divided into anterior, posterior, lateral, and transforaminal. Among these, anterior currently accounts for the largest market share.
Based on the end user, the global interbody fusion cage market can be bifurcated into hospitals and clinics, ambulatory surgical center, and others. Currently, hospitals and clinics exhibit a clear dominance in the market.
On a regional level, the market has been classified into North America, Europe, Asia Pacific, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global interbody fusion cage market include Alphatec Spine, Inc., Aurora Spine Inc., B. Braun Melsungen AG, Johnson & Johnson, Medtronic Plc., NuVasive Inc, Orthofix US LLC, Precision Spine Inc., SpineArt SA, Ulrich Medicals USA, etc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












