
Non-Invasive Prenatal Testing Market Report by Product Type (Consumables, Instruments), Test Type (Materni 21, Harmony, Panaroma, Verifi, NIFTY, and Others), Technology (NGS, WGS, and Others), Method (Ultrasound Detection, Biochemical Screening Tests, Cell-Free DNA in Maternal Plasma Tests, Fetal Cells in Maternal Blood Tests, and Others), Application (Trisomy, Microdeletion Syndrome, and Others), End-User (Hospitals, Diagnostic Laboratories, and Others), and Region 2026-2034
Market Overview:
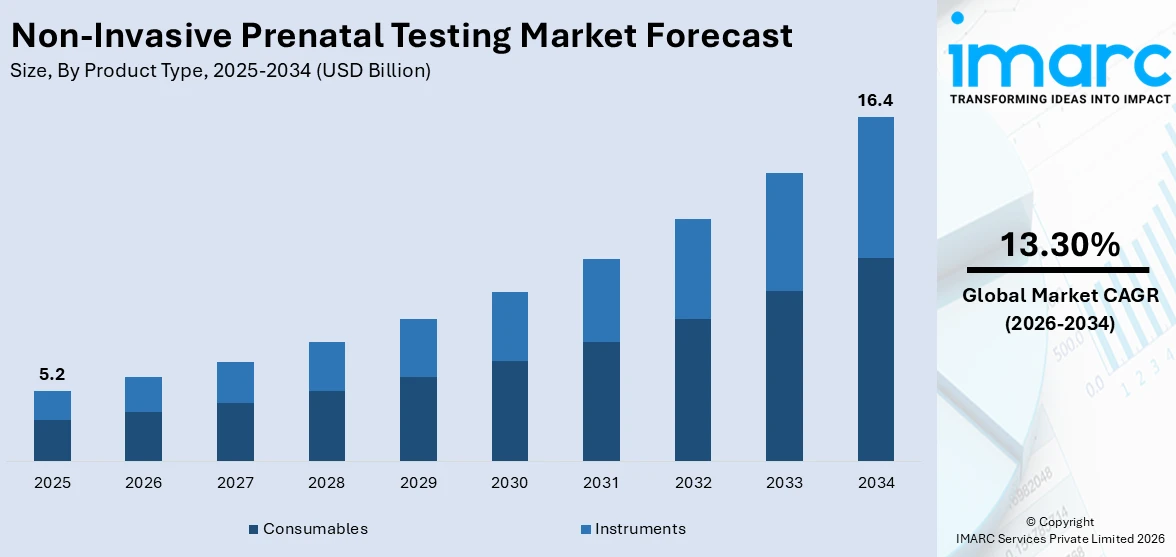
The global non-invasive prenatal testing market size reached USD 5.2 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 16.4 Billion by 2034, exhibiting a growth rate (CAGR) of 13.30% during 2026-2034.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 5.2 Billion |
|
Market Forecast in 2034
|
USD 16.4 Billion |
| Market Growth Rate 2026-2034 | 13.30% |
Non-invasive prenatal testing (NIPT) is a form of prenatal screening conducted on a pregnant woman’s blood sample to detect the presence of fetal abnormalities. The test is primarily used to diagnose genetic disorders such as Down syndrome (or trisomy 21), Edwards syndrome (or trisomy 18), Patau syndrome (or trisomy 13) and Turner syndrome, by analyzing DNA fragments in the mother’s blood. The procedure eliminates the need for various precarious and invasive procedures, such as chorionic villus sampling (CVS) and can be performed after nine weeks of pregnancy.
To get more information on this market Request Sample
The growing prevalence of genetic disorders of the fetus is one of the key factors driving the market growth. Furthermore, technological advancements in the field of life sciences and healthcare industry is also providing a boost to the market. For instance, the development of DNA sequencing technologies, regenerative medicines and breakthroughs in molecular research have created immense possibilities to combat the chances of any serious disease at the primitive stage itself. Additionally, the increasing acceptance rate for prenatal testing, along with rising health consciousness and awareness regarding the benefits and significance of such testing among the masses, is also driving the market growth. Healthcare providers, as well as expecting parents, are adopting alternatives to diagnose other genetic disorders, including spina bifida, cleft palate, and sickle cell anemia, and treat them in the early stages of fetal development. Moreover, the shifting trend toward childbearing at high maternal age (35 years or older) is also positively impacting the market growth. Advancing maternal age can lead to numerous health complications, such as high blood pressure, reduced fertility, risk of miscarriage and increased risk of chromosomal anomalies in the fetus.
Key Market Segmentation:
IMARC Group provides an analysis of the key trends in each sub-segment of the global non-invasive prenatal testing market report, along with forecasts at the global, regional and country level from 2026-2034. Our report has categorized the market based on product type, test type, technology, method, application and end-user.
Breakup by Product Type:
- Consumables
- Instruments
Breakup by Test Type:
- Materni 21
- Harmony
- Panaroma
- Verifi
- NIFTY
- Others
Breakup by Technology:
- NGS
- WGS
- Others
Breakup by Method:
- Ultrasound Detection
- Biochemical Screening Tests
- Cell-Free DNA in Maternal Plasma Tests
- Fetal Cells in Maternal Blood Tests
- Others
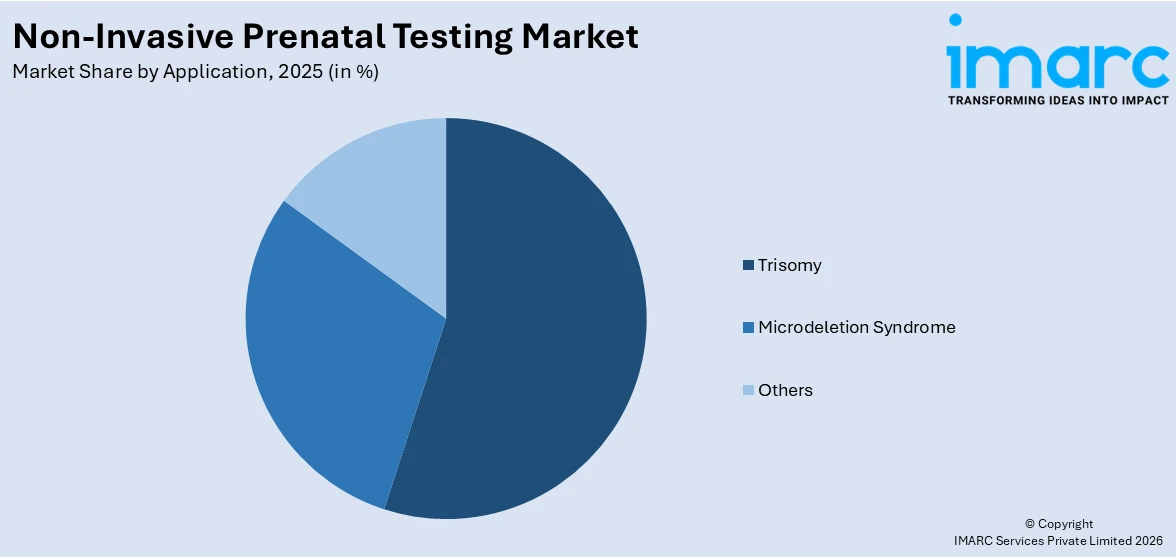
Breakup by Application:
Access the comprehensive market breakdown Request Sample
- Trisomy
- Microdeletion Syndrome
- Others
Breakup by End-User:
- Hospitals
- Diagnostic Laboratories
- Others
Breakup by Region:
- North America
- United States
- Canada
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
Competitive Landscape:
The report provides a comprehensive analysis of the competitive landscape in the global non-invasive prenatal testing market with detailed profiles of all major companies, including:
- Berry Genomics
- BGI
- CENTOGENE GmbH
- Eurofins LifeCodexx GmbH
- F. Hoffmann-La Roche Ltd
- Igenomix
- Illumina, Inc.
- Laboratory Corporation of America Holdings
- MedGenome
- Myriad Genetics, Inc.
- Natera, Inc.
- Quest Diagnostics Incorporated
- Thermo Fisher Scientific Inc.
- Yourgene Health
Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Segment Coverage | Product Type, Test Type, Technology, Method, Application, End-User, Region |
| Region Covered | North America, Asia Pacific, Europe, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, China, Japan, India, South Korea, Australia, Indonesia, Germany, France, United Kingdom, Italy, Spain, Russia, Brazil, Mexico |
| Companies Covered | Berry Genomics, BGI, CENTOGENE GmbH, Eurofins LifeCodexx GmbH, F. Hoffmann-La Roche Ltd, Igenomix, Illumina, Inc., Laboratory Corporation of America Holdings, MedGenome, Myriad Genetics, Inc., Natera, Inc., Quest Diagnostics Incorporated, Thermo Fisher Scientific Inc., Yourgene Health, etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Frequently Asked Questions About the Non-Invasive Prenatal Testing Market Report
The global non-invasive prenatal testing market was valued at USD 5.2 Billion in 2025.
We expect the global non-invasive prenatal testing market to exhibit a CAGR of 13.30% during 2026-2034.
The high prevalence of genetic disorders, coupled with the introduction of numerous awareness programs towards the availability of early diagnosis and treatment facilities, is primarily driving the global non-invasive prenatal testing market.
The sudden outbreak of the COVID-19 pandemic had led to postponement of elective prenatal screening tests to reduce the risk of the coronavirus infection upon hospital visits and interaction with medical equipment.
Based on the product type, the global non-invasive prenatal testing market has been segmented into consumables and instruments, where consumables hold the majority of the total market share.
Based on the test type, the global non-invasive prenatal testing market can be divided into Materni 21, Harmony, Panaroma, Verifi, NIFTY, and others. Currently, Materni 21 test exhibits a clear dominance in the market.
Based on the technology, the global non-invasive prenatal testing market has been bifurcated into NGS, WGS, and others. Among these, NGS technology currently accounts for the majority of the global market share.
Based on the method, the global non-invasive prenatal testing market can be categorized into ultrasound detection, biochemical screening tests, cell-free DNA in maternal plasma tests, fetal cells in maternal blood tests, and others. Currently, cell-free DNA in maternal plasma tests method holds the largest market share.
Based on the application, the global non-invasive prenatal testing market has been divided into trisomy, microdeletions syndrome, and others. Among these, trisomy accounts for the majority of the total market share.
Based on the end-user, the global non-invasive prenatal testing market can be segregated into hospitals, diagnostic laboratories, and others. Currently, diagnostic laboratories exhibit a clear dominance in the market.
On a regional level, the market has been classified into North America, Europe, Asia Pacific, Middle East and Africa, and Latin America, where North America currently dominates the global market.
Some of the major players in the global non-invasive prenatal testing market include Berry Genomics, BGI, CENTOGENE GmbH, Eurofins LifeCodexx GmbH, F. Hoffmann-La Roche Ltd, Igenomix, Illumina, Inc., Laboratory Corporation of America Holdings, MedGenome, Myriad Genetics, Inc., Natera, Inc., Quest Diagnostics Incorporated, Thermo Fisher Scientific Inc., and Yourgene Health.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












