
Pharmaceutical Analytical Testing Outsourcing Market by Type (Active Pharmaceutical Ingredients (API), Additives, Finished Products), Service (Bioanalytical Testing, Method Development and Validation, Stability Testing, and Others), End User (Pharmaceutical Companies, Biopharmaceutical Companies, Contract Research Organizations), and Region 2026-2034
Pharmaceutical Analytical Testing Outsourcing Market Overview:
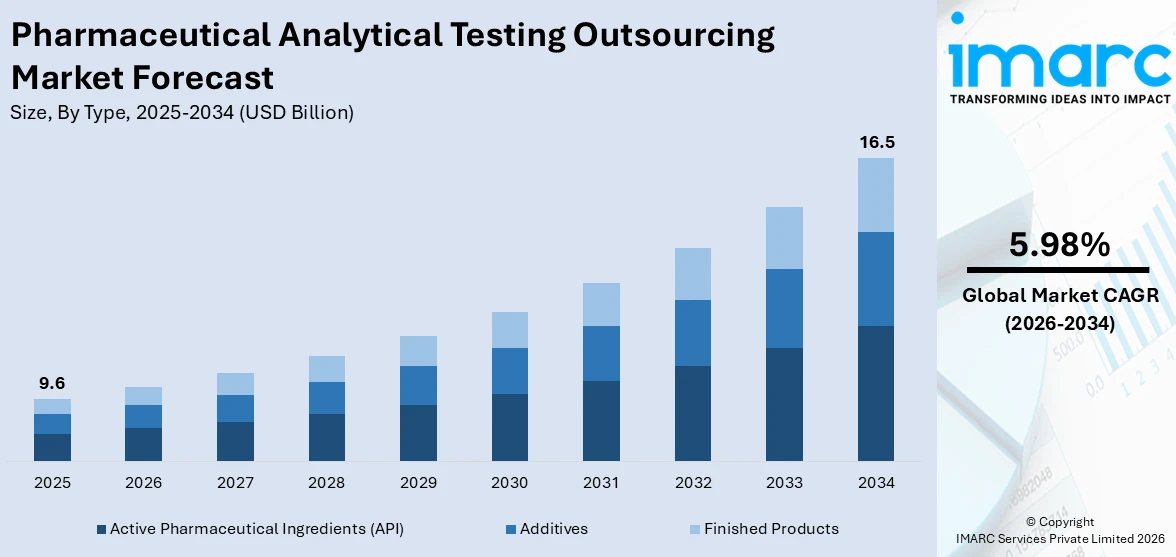
The global pharmaceutical analytical testing outsourcing market size reached USD 9.6 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 16.5 Billion by 2034, exhibiting a growth rate (CAGR) of 5.98% during 2026-2034. The market is driven by the growing focus on new drug delivery systems, such as liposomes, nanoparticles, and microspheres, increasing prevalence of chronic diseases like cancer, diabetes, and cardiovascular disease, and rising emphasis on drug development.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
| Market Size in 2025 | USD 9.6 Billion |
| Market Forecast in 2034 | USD 16.5 Billion |
| Market Growth Rate (2026-2034) | 5.98% |
Pharmaceutical Analytical Testing Outsourcing Market Analysis:
- Major Market Drivers: The increasing emphasis on targeted medication delivery systems, especially in oncology and chronic illness management, is supporting the market growth.
- Key Market Trends: The growing need to determine the pharmacokinetics, pharmacodynamics, and safety profiles of novel medications is offering a favorable market outlook.
- Geographical Trends: North America enjoys the leading position in the market because of its well-established pharmaceutical sector.
- Competitive Landscape: Some of the major market players in the pharmaceutical analytical testing outsourcing industry include Ajinomoto Bio-Pharma Services (Ajinomoto Co. Inc.), Alcami Corporation Inc., Boston Analytical Inc., Catalent Inc., Charles River Laboratories International Inc., Eurofins Scientific SE, Intertek Group plc, Laboratory Corporation of America Holdings, Pace Analytical Services LLC, SGS S.A., Thermo Fisher Scientific Inc., West Pharmaceutical Services Inc., and WuXi AppTec Inc., among many others.
- Challenges and Opportunities: Challenges in the market include stringent regulatory requirements and data security concerns, while opportunities lie in the rising demand for biologics and biosimilars.
To get more information on this market Request Sample
Pharmaceutical Analytical Testing Outsourcing Market Trends:
Increasing focus on drug delivery
According to the IMARC Group’s report, the global pharmaceutical drug delivery market reached US$ 1,397.6 Billion in 2023. New drug delivery systems, such as liposomes, nanoparticles, and microspheres, necessitate advanced analytical testing techniques. As pharmaceutical businesses are creating more complicated formulations, the need for specialized analytical testing services is growing. Drug delivery methods frequently seek to improve medications' bioavailability. Analytical testing is required for conducting bioavailability and bioequivalence studies, which is driving outsourcing demand as businesses are seeking expertise in this field. There is an increasing emphasis on targeted medication delivery systems, notably in oncology and chronic illness management. Analytical testing is critical for defining these systems, including their release profiles and interactions with biological systems, necessitating additional outsourcing. As medication delivery technology is advancing, so do regulatory regulations. Pharmaceutical companies frequently outsource analytical testing to maintain compliance with increasing rules around new drugs.
Rising demand for bioanalytical testing
The increasing prevalence of chronic diseases like cancer, diabetes, and cardiovascular disease is catalyzing the demand for effective treatments. Bioanalytical testing is critical in determining the pharmacokinetics, pharmacodynamics, and safety profiles of novel medications, which is leading to greater outsourcing of these testing services. The pharmaceutical sector is experiencing tremendous increase in biologics and biosimilars, which necessitate specialist bioanalytical testing to assess their complicated structures and interactions. Outsourcing these testing services provides firms with access to the essential skills and technology. Regulatory organizations, such as the Food and Drug Administration (FDA) and European Medicines Agency (EMA), demand extensive bioanalytical testing as part of the drug licensing process. Pharmaceutical companies frequently outsource these testing to ensure compliance with changing regulatory standards and to benefit from the specialist knowledge of contract research organizations (CROs). The IMARC Group’s report shows that the global bioanalytical testing services market is expected to reach 9% during 2024-2032.
Growing emphasis on drug development
As per the PDF shared by the United Nations Office on Drugs and Crime (UNODC) on World Drug Report 2023, around 86% of people worldwide, especially in low- and middle-income countries, have little access to drugs like pharmaceutical opioids, used for pain relief. As a result, there is a rise in the demand for drug development across the globe. Pharmaceutical companies are making significant investments in research and development (R&D) activities to discover new medications and treatments. This increase in investment creates a greater demand for analytical testing services to assist development activities. As medication development is advancing, formulations are becoming increasingly complex, particularly with the introduction of biologics, biosimilars, and innovative drug delivery methods. Analytical testing is required to characterize these complicated mixtures, prompting outsourcing to specialized laboratories. The competitive nature of the pharmaceutical sector needs speedier drug development procedures. Companies outsource analytical testing to streamline processes, shorten deadlines, and increase efficiency. The pharmaceutical sector must meet severe regulatory criteria. Outsourcing analytical testing ensures compliance with Good Laboratory Practice (GLP) and Good Manufacturing Practice (GMP) requirements, which is crucial for regulatory submissions.
Pharmaceutical Analytical Testing Outsourcing Market Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the market, along with pharmaceutical analytical testing outsourcing market forecasts at the global, regional, and country levels for 2026-2034. Our report has categorized the market based on type, service, and end user.
Breakup by Type:
- Active Pharmaceutical Ingredients (API)
- Additives
- Finished Products
The report has provided a detailed breakup and analysis of the market based on the type. This includes active pharmaceutical ingredients (API), additives, and finished products.
The active pharmaceutical ingredients (API) segment includes analytical testing services for raw materials and compounds that have therapeutic effects in pharmaceuticals. This segment is critical since APIs must undergo rigorous testing for purity, potency, and stability to assure their safety and efficacy in therapeutic formulations. Outsourcing API testing gives pharmaceutical companies access to specialist laboratories with modern analytical tools and knowledge, allowing them to perform complex tests and method validations. The growing emphasis on drug development, along with the increasing number of biologics and biosimilars, is driving the demand for API testing services, making it a critical part of the market.
The additives area includes the analytical testing of excipients and other auxiliary ingredients found in medicine formulations, such as stabilizers, preservatives, and fillers. These additives serve an important role in improving the performance, stability, and distribution of active substances in pharmaceutical products. As formulations are becoming more complex, rigorous testing of these additives is required to assure compatibility, safety, and efficacy. Outsourcing additive testing allows pharmaceutical businesses to benefit from the expertise of contract research organizations (CROs) that specialize in assessing the physicochemical properties and interactions of diverse excipients. This tendency is fueled further by the increased need for innovative drug delivery technologies, which require thorough additive testing to enhance formulation performance.
The finished products section includes analytical testing services for pharmaceutical items that enter the market. This testing is necessary to ensure that the products fulfill the requirements and regulatory criteria for safety, quality, and efficacy. Comprehensive testing of final products includes several assessments, such as stability studies, potency testing, and impurity analysis. As regulatory authorities highlight the need of quality assurance, the demand for completed product testing is growing. Pharmaceutical businesses benefit from outsourcing this area because they gain access to specialist laboratories capable of performing a variety of analytical techniques while adhering to Good Manufacturing Practices (GMP), which is catalyzing the pharmaceutical analytical testing outsourcing market demand. This category is especially important in a competitive market, where the timely introduction of high-quality medications is critical to success.
Breakup by Service:
- Bioanalytical Testing
- Clinical
- Non-Clinical
- Method Development and Validation
- Extractable and Leachable
- Impurity Method
- Technical Consulting
- Others
- Stability Testing
- Drug Substance
- Stability Indicating Method Validation
- Accelerated Stability Testing
- Photostability Testing
- Others
- Others
A detailed breakup and analysis of the market based on the service have also been provided in the report. This includes bioanalytical testing (clinical and non-clinical), method development and validation (extractable and leachable, impurity method, technical consulting, and others), stability testing (drug substance, stability indicating method validation, accelerated stability testing, photostability testing, and others), and others.
The bioanalytical testing section provides a variety of services for assessing the pharmacokinetics, pharmacodynamics, and safety profiles of pharmaceutical substances in biological matrices. This testing is critical for medication research since it determines the absorption, distribution, metabolism, and elimination (ADME) of active pharmaceutical ingredients (APIs) in the body. Bioanalytical testing frequently employs complicated assays, such as immunoassays, mass spectrometry, and chromatography, to reliably assess drug quantities in biological samples. The growing need for biologics, biosimilars, and customized medicine is propelling the growth of this market, as pharmaceutical companies outsource specialist skills and innovative technology to assure precise and trustworthy testing findings.
The method development and validation segment focuses on creating and verifying analytical methods to ensure their accuracy, reliability, and reproducibility in measuring pharmaceutical compounds. This service is essential for pharmaceutical companies to establish robust methodologies for quantifying APIs and assessing their quality throughout the drug development process. Outsourcing method development and validation allows companies to leverage the expertise of specialized laboratories with access to cutting-edge technologies and regulatory knowledge, thereby impelling the pharmaceutical analytical testing outsourcing market growth. As the pharmaceutical landscape becomes increasingly complex, with the rise of novel drug formulations and biologics, the demand for these services is rising. Method validation is also critical for regulatory submissions, making this segment integral to the overall analytical testing process.
The stability testing segment involves evaluating the stability of pharmaceutical products over time under various environmental conditions, such as temperature, humidity, and light exposure. This testing is vital for determining the shelf life, storage conditions, and expiration dates of finished products. Stability testing ensures that pharmaceutical products maintain their quality, potency, and safety throughout their intended shelf life. Outsourcing stability testing enables pharmaceutical companies to comply with regulatory requirements while accessing specialized expertise in designing stability studies and analyzing data. As regulatory agencies continue to emphasize the importance of stability data in the approval process, the demand for outsourcing this service is expected to increase, particularly with the growing focus on biologics and complex formulations that may have unique stability challenges.
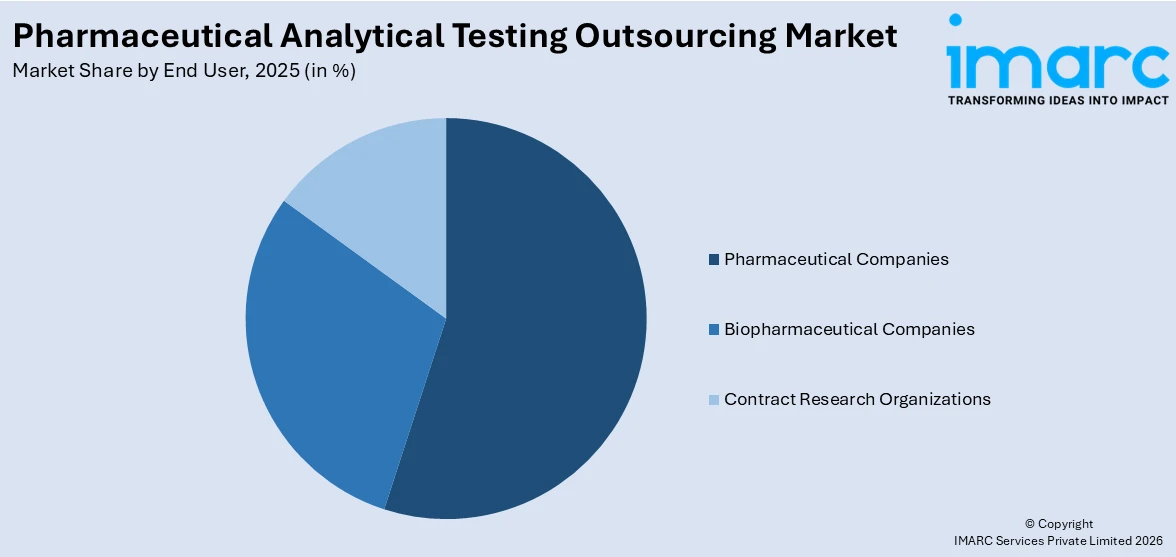
Breakup by End User:
Access the comprehensive market breakdown Request Sample
- Pharmaceutical Companies
- Biopharmaceutical Companies
- Contract Research Organizations
Pharmaceutical companies represent the leading market segment
The report has provided a detailed breakup and analysis of the market based on the end user. This includes pharmaceutical companies, biopharmaceutical companies, and contract research organizations. According to the report, pharmaceutical companies represent the largest segment.
Developing new medications is an expensive and time-consuming process. Pharmaceutical businesses can save money on operations by outsourcing analytical testing, allowing them to better manage resources. This is especially crucial as businesses are striving to optimize profits while managing significant R&D costs. The field of pharmaceutical testing is ever-changing, with new analytical techniques and technologies emerging. Outsourcing allows pharmaceutical businesses to gain access to cutting-edge technology and procedures without investing heavily in in-house facilities. Pharmaceutical businesses rely heavily on the ability to quickly bring innovative medications to market. Outsourcing analytical testing can simplify processes and shorten schedules, allowing for faster product development and regulatory submissions.
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America leads the market, accounting for the largest pharmaceutical analytical testing outsourcing market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America represents the largest regional market for pharmaceutical analytical testing outsourcing.
North America, particularly the United States, is home to some of the world’s largest and most influential pharmaceutical companies. The region has a well-established pharmaceutical sector that invests heavily in research and development (R&D) activities, resulting in a high demand for analytical testing services to support drug discovery and development. The presence of stringent regulatory bodies, such as the Food and Drug Administration (FDA) in the US and Health Canada, ensures that pharmaceutical products meet rigorous safety and efficacy standards. This regulatory environment is creating the need for comprehensive analytical testing to comply with Good Laboratory Practices (GLP) and Good Manufacturing Practices (GMP), making outsourcing a common strategy for pharmaceutical companies in the region, especially in the US. The IMARC Group’s report shows that the United States pharmaceutical analytical testing outsourcing market size is projected to exhibit a growth rate (CAGR) of 8.20% during 2024-2032.
Competitive Landscape:
- The pharmaceutical analytical testing outsourcing market research report has also provided a comprehensive analysis of the competitive landscape in the market. Detailed profiles of all major companies have also been provided. Some of the major market players in the pharmaceutical analytical testing outsourcing industry include Ajinomoto Bio-Pharma Services (Ajinomoto Co. Inc.), Alcami Corporation Inc., Boston Analytical Inc., Catalent Inc., Charles River Laboratories International Inc., Eurofins Scientific SE, Intertek Group plc, Laboratory Corporation of America Holdings, Pace Analytical Services LLC, SGS S.A., Thermo Fisher Scientific Inc., West Pharmaceutical Services Inc., and WuXi AppTec Inc.
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
- Key players in the market are focusing on several strategic initiatives to enhance their market presence and service offerings. They are investing in advanced analytical technologies, such as high-performance liquid chromatography (HPLC) and mass spectrometry, to provide accurate and reliable testing services. These companies are also expanding their laboratory infrastructure and geographic reach through mergers and acquisitions (M&A), collaborations, and partnerships with pharmaceutical firms to cater to the growing demand for specialized testing, particularly in biologics and biosimilars. For instance, in September 2024, Pace Life Sciences, LLC, a contract development, and manufacturing organization (CDMO) and a division of Pace, a Science and Technology Company, acquired Catalent Inc.’s Center-of-Excellence for small molecule analytical services located in Research Triangle Park (RTP), NC. This acquisition will support numerous biopharma and pharma clients with analytical services to bring life-changing medicines to market. Additionally, key players are enhancing their capabilities in regulatory compliance, offering end-to-end services that support drug development from early research to commercialization. To maintain competitive advantage, they are emphasizing quality assurance, data integrity, and faster turnaround times, as well as exploring innovative testing solutions like automation and artificial intelligence (AI) to optimize processes.
Pharmaceutical Analytical Testing Outsourcing Market News:
- March 2024: LGM Pharma, a leading provider of tailored API and CDMO services for the entire drug product lifecycle, announced a significant expansion of its Analytical Testing Services (ATS) offering, as well as the addition of new suppository manufacturing capabilities to its contract development and manufacturing (CDMO) portfolio.
- September 2023: Global contract development and manufacturing organization (CDMO) Recipharm opened a new analytical lab in Bengaluru, India to enhance its global testing capabilities for nitrosamines, extractables and leachables, and elemental impurity testing. This will also expand Recipharm’s existing analytical capabilities and provide support for biotech and pharma customers in their research and development (R&D) activities of oral solids, drug delivery, inhalation, vaccines and injectables.
Pharmaceutical Analytical Testing Outsourcing Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Pharmaceutical Analytical Testing Outsourcing Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Types Covered | Active Pharmaceutical Ingredients (API), Additives, Finished Products |
| Services Covered |
|
| End Users Covered | Pharmaceutical Companies, Biopharmaceutical Companies, Contract Research Organizations |
| Region Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Ajinomoto Bio-Pharma Services (Ajinomoto Co. Inc.), Alcami Corporation Inc., Boston Analytical Inc., Catalent Inc., Charles River Laboratories International Inc., Eurofins Scientific SE, Intertek Group plc, Laboratory Corporation of America Holdings, Pace Analytical Services LLC, SGS S.A., Thermo Fisher Scientific Inc., West Pharmaceutical Services Inc., WuXi AppTec Inc., etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report:
- How has the global pharmaceutical analytical testing outsourcing market performed so far, and how will it perform in the coming years?
- What are the drivers, restraints, and opportunities in the global market?
- What is the impact of each driver, restraint, and opportunity on the global market?
- What are the key regional markets?
- Which countries represent the most attractive market?
- What is the breakup of the market based on the type?
- Which is the most attractive type in the market?
- What is the breakup of the market based on the service?
- Which is the most attractive service in the market?
- What is the breakup of the market based on the end user?
- Which is the most attractive end user in the market?
- What is the competitive structure of the market?
- Who are the key players/companies in the global pharmaceutical analytical testing outsourcing market?
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the pharmaceutical analytical testing outsourcing market from 2020-2034.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the global pharmaceutical analytical testing outsourcing market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the pharmaceutical analytical testing outsourcing industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












