
Recombinant DNA Technology Market Report by Product (Medical, Non-Medical), Component (Expression System, Cloning Vector), Application (Food and Agriculture, Health and Disease, Environment, and Others), End User (Biotechnology and Pharmaceutical Companies, Academic and Government Research Institutes, and Others), and Region 2026-2034
Global Recombinant DNA Technology Market:
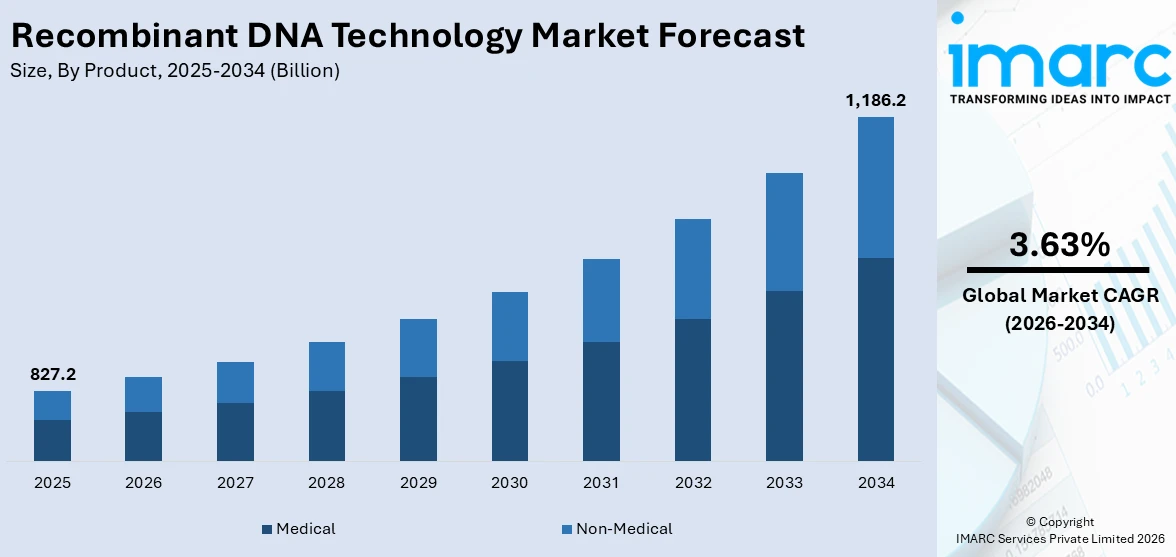
The global recombinant DNA technology market size reached USD 827.2 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 1,186.2 Billion by 2034, exhibiting a growth rate (CAGR) of 3.63% during 2026-2034. The ongoing advancements in genetic engineering and biotechnology, the growing demand for biopharmaceuticals, increasing shift towards personalized medicines, and the rising prevalence of chronic diseases are primarily driving the market's growth.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025 |
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
| Market Size in 2025 | USD 827.2 Billion |
| Market Forecast in 2034 | USD 1,186.2 Billion |
| Market Growth Rate (2026-2034) | 3.63% |
Recombinant DNA Technology Market Analysis:
- Major Market Drivers: The rising demand for improving different health conditions coupled with the improving treatment strategies on account of the developing diagnostics kits, monitoring devices, and new therapeutic approaches, represents one of the key factors impelling the growth of the market.
- Key Market Trends: Rapid urbanization, growing global population, reducing arable land, considerable rising food shortage, and increasing adoption of GMO crops, are escalating the market demand. In addition, the integration of recombinant DNA technology with effective gene therapy for replacing defective genes with normal ones and the production of antigen-specific antibodies for clinical research, studies, and disease diagnosis, are anticipated to drive the market growth in the upcoming years.
- Competitive Landscape: Some of the prominent recombinant DNA technology market companies include Amgen Inc, Cibus, F.Hoffmann-La Roche Ltd, GenScript, GlaxoSmithKline plc., Horizon Discovery Group plc, Merck KGaA, New England Biolabs, Novo Nordisk A/S, Pfizer Inc., Sanofi S.A, and Syngene International Ltd (Biocon Limited), among many others.
- Geographical Trends: According to the recombinant DNA technology market dynamics, North America holds a prominent share in the recombinant DNA technology market, driven by extensive research and development activities, well-established healthcare infrastructure, and significant government funding. Moreover, the growth in Europe is supported by a strong focus on research and development and high adoption of biopharmaceuticals.
- Challenges and Opportunities: Rising regulatory compliances and high competition among key players are hampering the market growth. However, there is an increasing demand for environmentally friendly and fuel-efficient landing gear systems. Manufacturers can capitalize on this trend by developing lightweight materials and energy-efficient designs.
To get more information on this market Request Sample
Recombinant DNA Technology Market Trends:
Rising Demand for Biopharmaceuticals
The rising demand for biopharmaceuticals, including monoclonal antibodies, vaccines, and insulin, is a major driver. For instance, according to IMARC, the global biopharmaceutical market size reached US$ 300.5 Billion in 2023. Looking forward, IMARC Group expects the market to reach US$ 643.9 Billion by 2032, exhibiting a growth rate (CAGR) of 8.6% during 2024-2032. Recombinant DNA technology is pivotal in the production of these products, which are essential for treating various chronic diseases. These factors are expected to propel the recombinant DNA technology market in the coming years.
Increasing Prevalence of Chronic Diseases
The increasing prevalence of chronic diseases, such as cancer, diabetes, and cardiovascular diseases, is one of the significant factors driving the market's growth. For instance, according to the IDF Diabetes Atlas (2021), 10.5% of adults aged 20 to 79 suffer from diabetes. Also, by 2045, one in every eight persons, or roughly 783 million, will have diabetes, representing a 46% rise. Recombinant DNA technology enables the production of essential therapeutic proteins and hormones, such as insulin for diabetes management and erythropoietin for anemia treatment. These factors are further positively influencing the recombinant DNA technology industry market.
Technological Advancements
Continuous technological innovations, such as CRISPR and other gene-editing tools, are enhancing the precision and application scope of recombinant DNA technology, driving the market expansion. For instance, in August 2024, a group of German researchers developed an enhanced CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) technique to solve and improve existing obstacles in CAR-T cell therapy that targets both blood and solid tumors. CRISPR is a unique technology that enables geneticists and medical researchers to edit portions of the genome by removing, inserting, or changing DNA sequences, thereby boosting the recombinant DNA technology systems market revenue.
Global Recombinant DNA Technology Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global recombinant DNA technology market report, along with forecasts at the global, regional, and country levels from 2026-2034. Our report has categorized the market based on product, component, application, and end user.
Breakup by Product:
- Medical
- Therapeutic Agent
- Human Protein
- Vaccines
- Non-Medical
- Biotech Crops
- Specialty Chemicals
- Others
The report has provided a detailed breakup and analysis of the recombinant DNA technology market based on the product. This includes medical (therapeutic agent, human protein, and vaccines), and non-medical (biotech crops, specialty chemicals, and others).
According to the recombinant DNA technology market outlook, recombinant DNA technology enables the development of gene therapies to treat genetic disorders like cystic fibrosis, hemophilia, and muscular dystrophy. This technology has revolutionized the production of human insulin, making it more accessible for diabetic patients. While crops like Bt cotton, Roundup Ready soybeans, and Golden Rice have been developed using rDNA technology to enhance resistance to pests, herbicides, and to improve nutritional content.
Breakup by Component:
- Expression System
- Cloning Vector
The report has provided a detailed breakup and analysis of the recombinant DNA technology market based on the component. This includes expression system and cloning vector.
According to the recombinant DNA technology market overview, an expression system refers to the host organism and the molecular machinery used to produce recombinant proteins. It includes the host cells, the vectors, and the regulatory sequences necessary for gene expression. While a cloning vector is a DNA molecule used to carry foreign genetic material into a host cell, where it can be replicated and/or expressed. It is used in the development of genetically modified crops with desirable traits such as pest resistance and improved nutritional content.
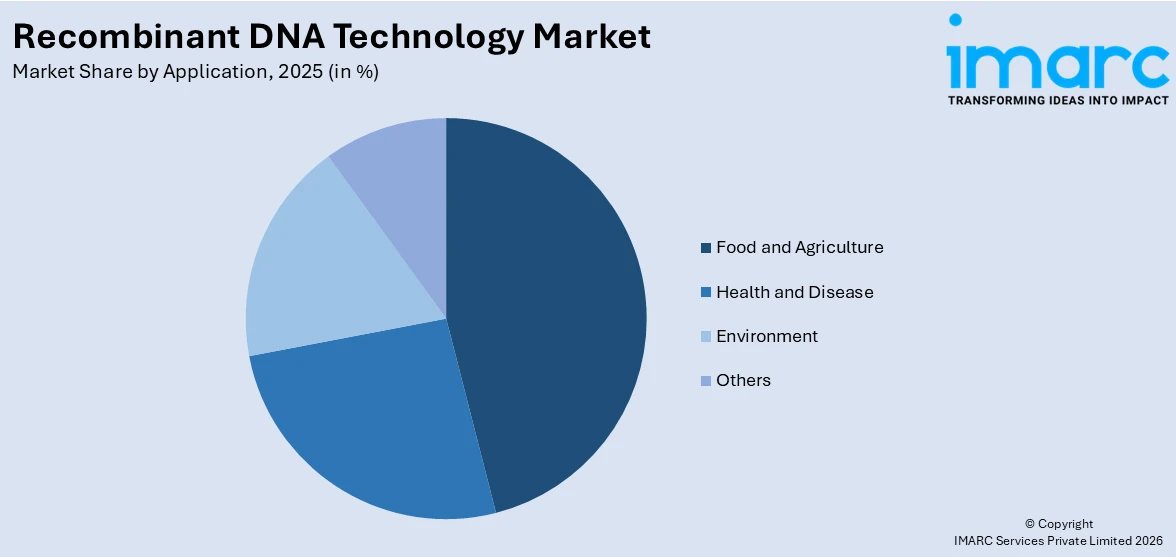
Breakup by Application:
Access the comprehensive market breakdown Request Sample
- Food and Agriculture
- Health and Disease
- Environment
- Others
A detailed breakup and analysis of the recombinant DNA technology market based on application has also been provided in the report. This includes food and agriculture, health and disease, environment, and others.
Recombinant DNA technology is used to develop genetically modified (GM) crops with desirable traits such as increased yield, pest resistance, herbicide tolerance, and improved nutritional content. This is crucial for meeting the food demands of a growing global population. Apart from this, in health and disease, it enables the production of insulin, growth hormones, monoclonal antibodies, and vaccines. The demand for effective and affordable biopharmaceuticals drives this sector. Besides this, it is also used to engineer microorganisms that can break down pollutants and toxins in the environment. This helps in cleaning up oil spills, heavy metals, and other environmental contaminants.
Breakup by End User:
- Biotechnology and Pharmaceutical Companies
- Academic and Government Research Institutes
- Others
A detailed breakup and analysis of the recombinant DNA technology market based on end user has also been provided in the report. This includes biotechnology and pharmaceutical companies, academic and government research institutes, and others.
In biotechnology and pharmaceutical companies, recombinant DNA technology is used to produce human insulin in bacteria, which is a major advancement over previous methods using animal insulin. Companies use rDNA to develop monoclonal antibodies for the treatment of various diseases, including cancer and autoimmune disorders. While government research institutes like the CDC use rDNA technology to monitor and study pathogens, enabling rapid response to outbreaks and epidemics.
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia and others); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa.
According to the recombinant DNA technology market statistics, North America is one of the prominent regions for rDNA technology due to significant investments in biotechnology and pharmaceuticals. Moreover, Europe has a well-established biotechnology sector, particularly in countries like Germany, France, and the UK. The growth of the European region is driven by the strong collaboration between academic institutions, research institutes, and industry.
Competitive Landscape:
The market research report has provided a comprehensive analysis of the competitive landscape. Detailed profiles of all major market companies have also been provided. Some of the key players in the market include:
- Amgen Inc
- Cibus
- F.Hoffmann-La Roche Ltd
- GenScript
- GlaxoSmithKline plc.
- Horizon Discovery Group plc
- Merck KGaA
- New England Biolabs
- Novo Nordisk A/S
- Pfizer Inc.
- Sanofi S.A
- Syngene International Ltd (Biocon Limited)
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
Recombinant DNA Technology Market Recent Developments:
- August 2024: German researchers developed an enhanced CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) technique to solve and improve existing obstacles in CAR-T cell therapy that targets both blood and solid tumors.
- July 2024: Scientists at the CSIR-Institute of Genomics and Integrative Biology in New Delhi developed an upgraded genome-editing device that can modify DNA more precisely and efficiently.
- February 2024: Belgian researchers from the VIB-KU Leuven Center for Microbiology and the VIB-UGent Center for Plant Systems Biology created a new toolbox comprising 16 different short DNA sequences that may initiate controlled and selective recombination processes in any genome.
Recombinant DNA Technology Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Predictive Market Assessment:
|
| Products Covered |
|
| Components Covered | Expression System, Cloning Vector |
| Applications Covered | Food and Agriculture, Health and Disease, Environment, Others |
| End Users Covered | Biotechnology and Pharmaceutical Companies, Academic and Government Research Institutes, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Amgen Inc, Cibus, F.Hoffmann-La Roche Ltd, GenScript, GlaxoSmithKline plc., Horizon Discovery Group plc, Merck KGaA, New England Biolabs, Novo Nordisk A/S, Pfizer Inc., Sanofi S.A, Syngene International Ltd (Biocon Limited), etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report:
- How has the global recombinant DNA technology market performed so far and how will it perform in the coming years?
- What has been the impact of COVID-19 on the global recombinant DNA technology market?
- What are the key regional markets?
- What is the breakup of the market based on the product?
- What is the breakup of the market based on the component?
- What is the breakup of the market based on the application?
- What is the breakup of the market based on the end user?
- What are the various stages in the value chain of the industry?
- What are the key driving factors and challenges in the industry?
- What is the structure of the global recombinant DNA technology market and who are the key players?
- What is the degree of competition in the industry?
Key Benefits for Stakeholders:
- IMARC's report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the recombinant DNA technology market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global recombinant DNA technology market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the recombinant DNA technology industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












