
Virtual Clinical Trials Market Report by Study Design (Interventional, Observational, Expanded Access), Indication (Oncology, Cardiovascular, and Others), and Region 2026-2034
Virtual Clinical Trials Market Size:
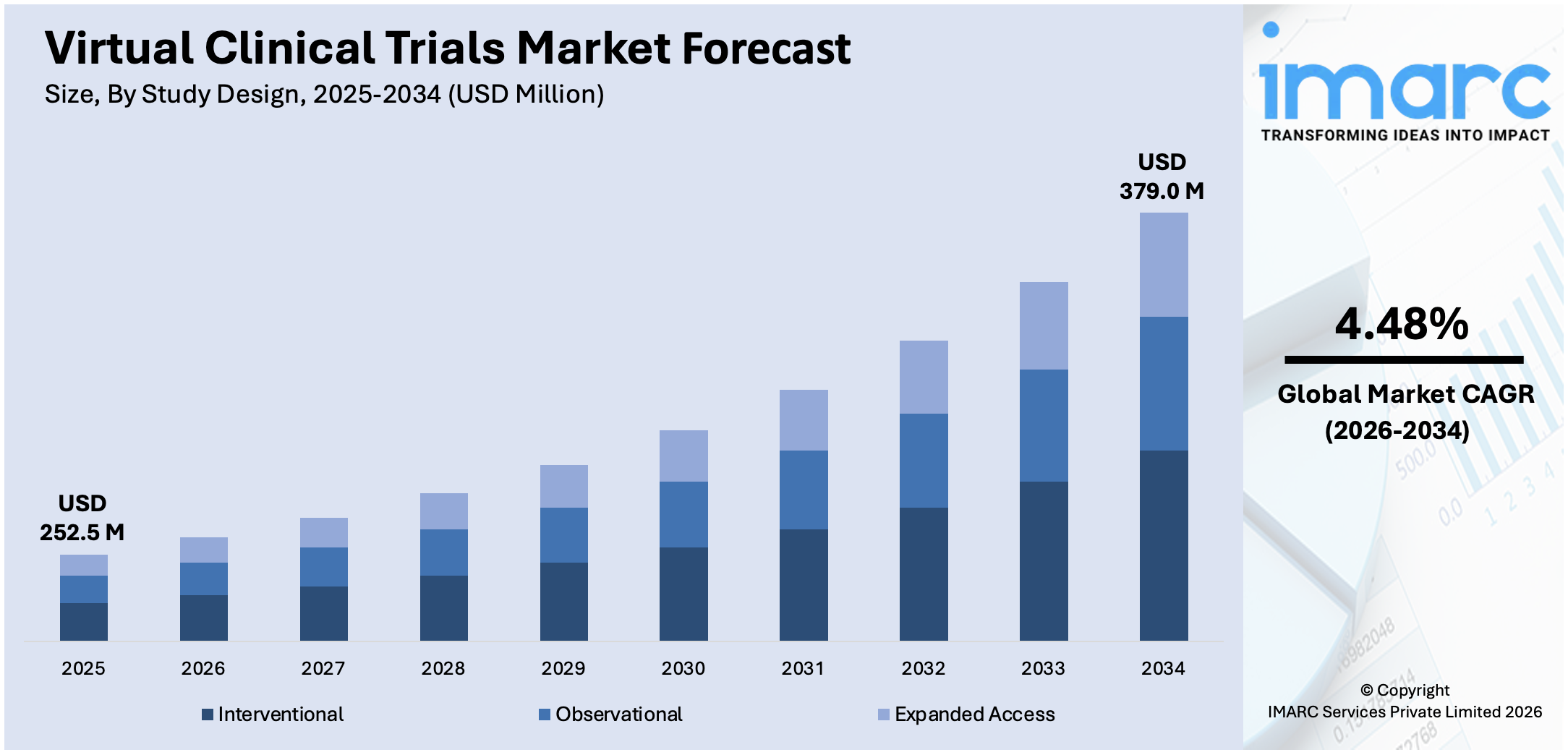
The global virtual clinical trials market size reached USD 252.5 Million in 2025. Looking forward, IMARC Group expects the market to reach USD 379.0 Million by 2034, exhibiting a growth rate (CAGR) of 4.48% during 2026-2034. Increasing demand for patient-centric approaches, advancements in technology like telemedicine and wearables, the impact of the COVID-19 pandemic, the emphasis on real-world evidence, and collaboration among stakeholders and efforts to address disparities in trial participation are some of the key factors contributing to the market growth.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 252.5 Million |
|
Market Forecast in 2034
|
USD 379.0 Million |
| Market Growth Rate 2026-2034 | 4.48% |
Virtual Clinical Trials Market Analysis:
- Major Market Drivers: The primary factors driving the virtual clinical trials market share is the growing emphasis on patient-centric trials, the need for reduced drug development cycles, and the increasing use of digital health technology are all aiding the market. The improvement of telemedicine, wearables, and mobile health apps has made remote monitoring of patients more achievable, reducing the need for physical site visits, which is further supporting the virtual clinical trials market growth. Increased acceptance of virtual trials by regulatory agencies, as well as a focus on cost savings in drug development, are other factors stimulating the virtual clinical trials market demand. The COVID-19 epidemic, which has illustrated how virtual trials can keep study performance continually even under limitations, has accelerated virtual clinical trials market outlook.
- Key Market Trends: As per the virtual clinical trials market trends, the market is becoming more focused on integrated platforms that provide an end-to-end solution for conducting of virtual clinical trials, starting from patient recruitment and ending with data analytics. Real-time data collection and usage of artificial intelligence (AI) and machine learning (ML) are on the rise, and while they do not dominate the market yet, they are being used increasingly to help make trials more efficient and informed, which is further driving the virtual clinical trials market recent price. As per the virtual clinical trials market report, the trend of globalization, facilitated by the use of mentioned technologies that provide access to a larger and more diverse patient population, is supporting the market growth. The virtual clinical trials market analysis also sees a growing number of partnerships between pharmaceutical companies and technology providers in their pursuit of innovation.
- Geographical Trends: As per the virtual clinical trials market statistics, North America is the largest market for virtual clinical trials. The key reasons for its dominance include developed healthcare IT infrastructure, high adoption of digital health and telemedicine, and favorable regulatory guidelines. The US alone has a significant number of technology companies and pharma giants working on deploying virtual trial platforms. Moreover, the region’s strong focus on clinical research and development alongside the presence of a broad patient pool make it the ideal place for piloting remote trials, which is further supporting the virtual clinical trials market forecast. As per the virtual clinical trials market overview, Europe and Asia-Pacific are progressively investing more in the development of virtual trial technology while becoming more aware of its benefits.
- Competitive Landscape: As per the virtual clinical trials market recent developments, some of the key virtual clinical trials market companies include Clinical Ink Inc., Covance Inc., ICON Plc, IQVIA Inc., LEO Innovation Lab, Medable Inc., Medidata Solutions Inc. (Dassault Systèmes SE), Medpace Holdings Inc., Oracle Corporation, Parexel International Corporation (Pamplona Capital Management), PRA Health Sciences, Signant Health (Genstar Capital), etc.
- Challenges and Opportunities: The virtual clinical trials market is not without challenges, including data privacy and security, the need for standardization and validation of digital tools, and inertia in pharma to move from traditional trial modalities, among others. As per the virtual clinical trials market recent opportunities, continuous evolution of strong cybersecurity, establishment of consistent global standards for virtual trials, and education to involve stakeholders in direct-to-patient and hybrid trials should solve most of the worries are some of the factors supporting the market growth. The market has a high growth potential, enabling efficiencies in clinical trials, cost savings, and broader patient access and diversity to clinical research, with the overarching benefits of bringing new therapies to patients faster, which is further accelerating the virtual clinical trials market revenue.
To get more information on this market Request Sample
Virtual Clinical Trials Market Trends:
Surging focus on patient-centric approaches
One of the key drivers that define the current state of the global virtual clinical trials market, and its evolution is the increasingly patient-centric and patient-focused approach. Traditional models are often complicated and inefficient in terms of patient recruitment, retention, and engagement. Virtual trials are designed with an orientation on patient comfort, convenience, and accessibility. As a result, decentralized designs make it possible for patients to be examined and treated without leaving the house, which means they do not need to attend multiple site visits and struggle with time and money spent on transportation. When patients are placed in the center of research, both practically and philosophically, they feel accepted and large-impactful, which creates a sufficiently inclusive ecosystem.
Advancements in Technology
Technology, especially telemedicine, wearables, and remote monitoring, is also essential in promoting the use of VCTs across the globe. Telemedicine facilitates remote consultations between the patients and the physicians, ensuring constant communication and support for the required period. Wearables are also a crucial form of technology that makes it possible to monitor individual health conditions 24/7 by providing real-time vitals and other data to the researchers. Furthermore, remote monitoring is also essential as it promotes data collection and storage and minimizes the errors that may come from timely manual documentation.
Impact of the COVID-19 Pandemic
COVID-19 also played a major role in the formation of the worldwide virtual clinical trials market. Thus, due to the shutdown and social distancing actions imposed in the course of the pandemic, several standard clinical trials were enforced to shut down. Therefore, the pandemic occurred to be the formation for recognition; the industry of virtual clinical extreme vulnerability to centralized trials. Virtual trials, on the other hand, promote a high degree of continuity and save researchers who are bound to continue their research through trial participants who are to remain healthy and safe. The pandemic therefore reignited innovation as stakeholders moved from standard design to various types of trials and used available technology to participate in remote data collection, monitoring, and patient recruitment.
Virtual Clinical Trials Market Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the market, along with forecasts at the global, regional, and country levels for 2026-2034. Our report has categorized the market based on study design and indication.
Breakup by Study Design:
- Interventional
- Observational
- Expanded Access
Interventional represents the leading market segment
The report has provided a detailed breakup and analysis of the market based on the study design. This includes interventional, observational, and expanded access. According to the report, interventional represented the largest segment.
The interventional segment in the global virtual clinical trials market is driven by several key factors, such as the increasing demand for innovative therapies and treatments fuels the adoption of virtual interventional trials, which offer a more agile and efficient approach to drug development. By leveraging virtual technologies, researchers can streamline the process of recruiting and enrolling participants, expediting the evaluation of investigational treatments and accelerating time-to-market for new drugs. Additionally, virtual interventional trials enable researchers to overcome geographical barriers and access a more diverse pool of participants, enhancing the generalizability and external validity of study findings. Moreover, the growing emphasis on patient-centered care and personalized medicine drives the need for tailored interventions and precision therapies, further driving the uptake of virtual interventional trials. These trials allow for the customization of treatment protocols and the delivery of interventions based on individual patient characteristics and preferences, leading to improved patient outcomes and satisfaction.
Breakup by Indication:
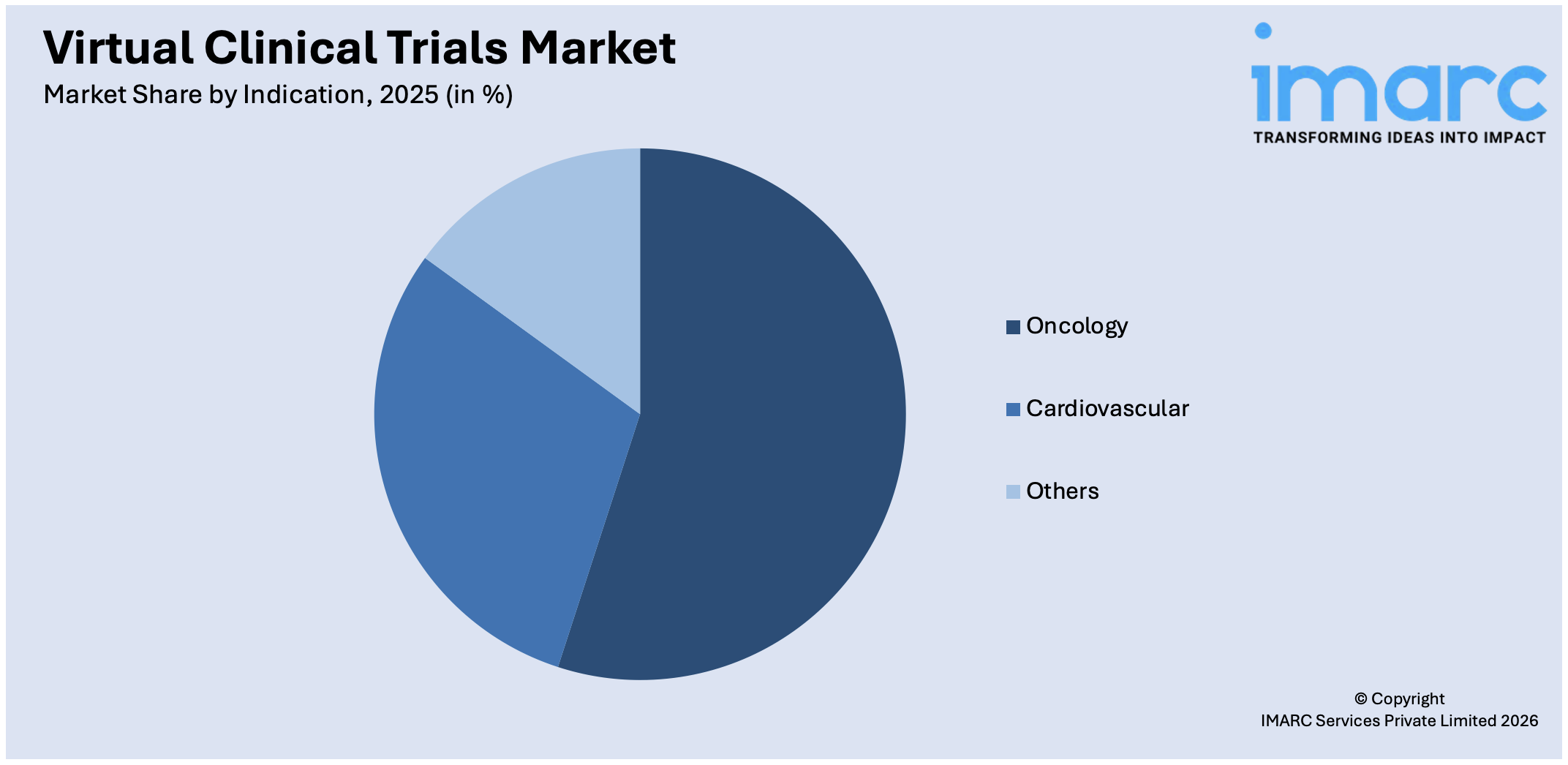
Access the comprehensive market breakdown Request Sample
- Oncology
- Cardiovascular
- Others
Oncology represents the leading market segment
The report has provided a detailed breakup and analysis of the market based on the indication. This includes oncology, cardiovascular, and others. According to the report, oncology represented the largest segment.
The oncology segment is driven by the increasing prevalence of cancer worldwide, spurred by factors such as aging populations, lifestyle changes, and environmental exposures. Additionally, advancements in oncology research and treatment modalities, including targeted therapies, immunotherapies, and precision medicine, are revolutionizing cancer care and driving demand for innovative clinical trial designs. Moreover, the growing understanding of the molecular and genetic mechanisms underlying various cancer types is facilitating the development of personalized treatment approaches tailored to individual patient profiles, fueling the need for specialized oncology trials. Furthermore, the rising incidence of rare and genetically-defined cancers is driving the demand for niche oncology trials aimed at developing targeted therapies for these patient populations, necessitating the use of innovative trial methodologies and collaboration among stakeholders to overcome challenges related to patient recruitment, trial logistics, and data collection in these specialized settings.
Breakup by Region:
- North America
- United States
- Canada
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America leads the market, accounting for the largest virtual clinical trials market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America represents the largest regional market for virtual clinical trials.
The North America region is driven by the increasing demand for patient-centric approaches and decentralized trial designs, where virtual clinical trials offer solutions to enhance patient engagement and retention rates. Advancements in technology, particularly in telemedicine, wearables, and remote monitoring tools, play a crucial role in propelling the adoption of virtual trial methodologies, enabling seamless data collection and analysis while reducing the burden on both patients and researchers. Moreover, the impact of the COVID-19 pandemic has accelerated the shift towards virtual trials in North America, as traditional trial methods faced disruptions, highlighting the need for flexible and resilient approaches to clinical research. Regulatory support and a favorable policy environment further bolster the growth of the virtual clinical trials market in North America, facilitating streamlined approval processes and fostering innovation in trial design and execution. Additionally, the region benefits from the presence of leading pharmaceutical companies and research institutions, driving investment in virtual trial infrastructure and capabilities to expedite drug development timelines and bring therapies to market more swiftly.
Competitive Landscape:
- The market research report has also provided a comprehensive analysis of the competitive landscape in the market. Detailed profiles of all major companies have also been provided. Some of the major market players in the virtual clinical trials industry include Clinical Ink Inc., Covance Inc., ICON Plc, IQVIA Inc., LEO Innovation Lab, Medable Inc., Medidata Solutions Inc. (Dassault Systèmes SE), Medpace Holdings Inc., Oracle Corporation, Parexel International Corporation (Pamplona Capital Management), PRA Health Sciences, Signant Health (Genstar Capital), etc.
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
- In the competitive landscape of the virtual clinical trials market, key players are actively engaged in various strategic initiatives to maintain their market position and drive growth. These initiatives include investments in research and development (R&D) to enhance virtual trial technologies and platforms, with a focus on improving data collection, patient engagement, and trial efficiency. Additionally, key players are forming strategic partnerships and collaborations with technology companies, research organizations, and regulatory agencies to leverage complementary expertise and resources, expand their geographical presence, and strengthen their capabilities in virtual trial design and execution. Furthermore, key players are investing in marketing and promotional activities to increase awareness and adoption of virtual trial solutions among pharmaceutical companies, contract research organizations, and healthcare providers. Moreover, mergers and acquisitions are prevalent strategies among key players, enabling them to broaden their service offerings, acquire innovative technologies, and consolidate their market position through vertical integration or diversification.
Virtual Clinical Trials Market News:
- In June 2022: Medidata announced ten highly innovative organizations have joined its groundbreaking Sensor Cloud Network. AliveCor, Aural Analytics, Biobeat, Blue Spark Technologies, Glooko, Indie Health, University of Arizona, Carnegie Mellon University, University of Rochester, and University of Vermont are now part of the first cross-sector collaboration focused on solving the challenges related to sensor integrations, standardization of sensor data, and the development of novel digital biomarkers and algorithms.
- In June 2021: Veradigm®, a leading provider of healthcare data and technology solutions and a business unit of Allscripts Healthcare Solutions (NASDAQ: MDRX), and PRA Health Sciences, Inc. announced the creation of the industry’s leading electronic healthcare records-based clinical research network that reaches more than 25,000 physicians and 40 million patients across the United States.
Virtual Clinical Trials Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Study Designs Covered | Interventional, Observational, Expanded Access |
| Indications Covered | Oncology, Cardiovascular, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Clinical Ink Inc., Covance Inc., ICON Plc, IQVIA Inc., LEO Innovation Lab, Medable Inc., Medidata Solutions Inc. (Dassault Systèmes SE), Medpace Holdings Inc., Oracle Corporation, Parexel International Corporation (Pamplona Capital Management), PRA Health Sciences, Signant Health (Genstar Capital), etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Questions Answered in This Report:
- How has the global virtual clinical trials market performed so far, and how will it perform in the coming years?
- What are the drivers, restraints, and opportunities in the global virtual clinical trials market?
- What is the impact of each driver, restraint, and opportunity on the global virtual clinical trials market?
- What are the key regional markets?
- Which countries represent the most attractive virtual clinical trials market?
- What is the breakup of the market based on the study design?
- Which is the most attractive study design in the virtual clinical trials market?
- What is the breakup of the market based on the indication?
- Which is the most attractive indication in the virtual clinical trials market?
- What is the competitive structure of the market?
- Who are the key players/companies in the global virtual clinical trials market?
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the virtual clinical trials market from 2020-2034.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the global virtual clinical trials market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the virtual clinical trials industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












