
Endoscope Reprocessing Market Report by Product (High-Level Disinfectants and Test Strips, Detergents and Wipes, Automated Endoscope Reprocessors, Endoscope Drying, Storage and Transport Systems, and Others), Process (Manual Cleaning, Automated Cleaning), End User (Hospitals and Clinics, Ambulatory Surgery Centers, and Others), and Region 2026-2034
Endoscope Reprocessing Market Size:
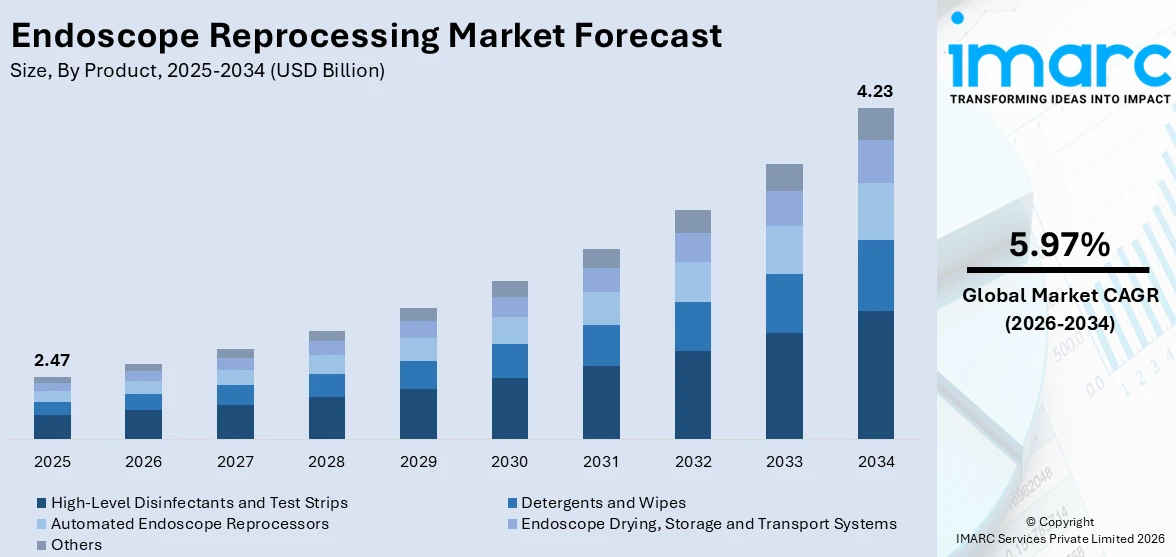
The global endoscope reprocessing market size reached USD 2.47 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 4.23 Billion by 2034, exhibiting a growth rate (CAGR) of 5.97% during 2026-2034. The market is primarily being driven by the rising demand for minimally invasive surgeries, and the implementation of stringent regulations on infection control. Additionally, rapid technological advancements in reprocessing equipment, growing awareness about patient safety, and the surging prevalence of endoscopy procedures are further contributing to the market growth.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 2.47 Billion |
|
Market Forecast in 2034
|
USD 4.23 Billion |
| Market Growth Rate 2026-2034 | 5.97% |
Endoscope Reprocessing Market Analysis:
- Major Market Drivers: The major drivers for the endoscope reprocessing market include a significant rise in the prevalence of endoscopic procedures, driven by the growing incidence of chronic diseases. Technological advancements in endoscopic equipment are enhancing the efficiency and effectiveness of these procedures. Additionally, stringent infection control regulations are fostering the demand for reliable reprocessing solutions. This, along with the growing awareness about patient safety and the risks associated with hospital-acquired infections (HAIs) further significantly boost the endoscope reprocessing demand and need. The expansion of healthcare facilities, coupled with the increasing volume of endoscopic procedures, underscores the demand for efficient and high-capacity reprocessing solutions. These factors collectively contribute to the robust growth and expansion of the endoscope reprocessing market.
- Key Market Trends: Some of the key market trends include the widespread adoption of automated reprocessing systems to enhance efficiency and consistency, increasing use of single-use endoscopes to mitigate cross-contamination risks, and advancements in high-level disinfectants and test strips for better efficacy. There is also a growing emphasis on eco-friendly reprocessing solutions and compliance with international safety standards, driving innovation and improvements in endoscope reprocessing technologies.
- Geographical Trends: As per the endoscope reprocessing market report, North America holds a leading position in the market, largely due to its sophisticated healthcare infrastructure and high frequency of endoscopic procedures. Stringent regulatory standards for infection control in the region emphasize the importance of effective reprocessing solutions, driving market growth. The strong commitment to patient safety and quality of care in North America, combined with substantial investments in healthcare technology, propels the adoption of advanced endoscope reprocessing solutions. Additionally, the presence of major market players and continuous technological innovations contribute significantly to North America's dominance in the market. These factors collectively reinforce the region's leading position and foster continued growth and development in the endoscope reprocessing sector.
- Competitive Landscape: Some of the major market players in the endoscope reprocessing industry include Advanced Sterilization Products (ASP) (Fortive Corporation), ARC Healthcare Solutions Inc., Belimed AG (Metall Zug AG), BES Rehab Ltd, Custom Ultrasonics Inc., Ecolab Inc., ENDO-TECHNIK W. Griesat GmbH (Albyn Medical S.L.), Getinge AB, HOYA Corporation, Metrex Research LLC, Olympus Corporation, Shinva Medical Instrument Co. Ltd., Steelco S.p.A. (Miele) and Steris plc., among many others.
- Challenges and Opportunities: Opportunities in the endoscope reprocessing market include the increasing adoption of automated reprocessing systems, the development of eco-friendly and efficient reprocessing solutions, and the expanding healthcare infrastructure in emerging economies. However, challenges persist, such as the high cost of advanced reprocessing equipment, the need for stringent compliance with varying regulatory standards, and the risk of infection due to improper reprocessing. Balancing cost-effectiveness with adherence to safety standards remains a critical challenge for market growth.
To get more information on this market Request Sample
Endoscope Reprocessing Market Trends:
Increasing Demand for Minimally Invasive Surgeries
The rising preference for minimally invasive surgeries is acting as a major driver for the endoscope reprocessing market growth. These procedures, which use endoscopes, offer benefits such as reduced recovery time, lower risk of infection, and less postoperative pain compared to traditional surgeries. According to the data published on National Center for Biotechnology, approximately 17.7 million gastrointestinal (GI) endoscopic procedures are performed annually in the United States, accounting for 68% of all endoscopic procedures. The composite infection rate following GI endoscopic procedures was 0.2%, with higher rates for specific procedures: 0.8% for ERCP, 0.123% for non-ERCP upper GI endoscopies, and 0.073% for lower GI endoscopies. As a result, the frequency of endoscopic procedures is increasing, leading to a higher demand for effective reprocessing solutions to ensure the safety and longevity of endoscopes.
Implementation of Stringent Infection Control Regulations
As per industry reports, failures in reprocessing flexible endoscopes are associated with hospital-acquired infections (HAIs), which place a significant burden on patients and healthcare systems. HAIs are the most reported adverse events affecting patients, with a prevalence of 6.5% across Europe. Surgical site infections, comprising 18.3% of all HAIs, negatively affect patient quality of life and increase morbidity and mortality rates. They also contribute to a significant financial burden on healthcare systems. In Germany, surgical site infections can lead to an average hospital stay extension of 17.9 days and an additional healthcare cost of €22,905 per patient. As a result, governments and healthcare organizations worldwide are implementing stringent regulations to prevent hospital-acquired infections (HAIs). To comply with these regulations and avoid penalties, healthcare facilities are investing in advanced endoscope reprocessing systems. This ensures that endoscopes are thoroughly cleaned, disinfected, and sterilized, thereby reducing the risk of HAIs and improving patient safety.
Technological Advancements in Reprocessing Equipment
Advancements in technologies are significantly driving the endoscope reprocessing market growth. Innovations, such as automated reprocessing machines, enhanced disinfectants, and sophisticated tracking systems, have improved the efficiency and reliability of the reprocessing process. These technologies ensure that endoscopes are consistently cleaned to high standards, reduce the risk of human error, and streamline the workflow in healthcare facilities, making them a critical factor in the adoption and expansion of endoscope reprocessing solutions. For instance, in January 2023, Agilis Robotics, a leading developer of flexible robotic instruments, completed its second round of live animal testing with its proprietary robot for endoscopic surgery. The results demonstrated promising outcomes in terms of efficacy, accuracy, and safety of the robotic system. Consequently, the increasing integration of robotics in endoscopic surgery is expected to boost the number of procedures performed, thereby driving market growth.
Endoscope Reprocessing Market Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the market, along with forecasts at the global, regional, and country levels for 2026-2034. Our report has categorized the market based on product, process, and end user.
Breakup by Product:
- High-Level Disinfectants and Test Strips
- Detergents and Wipes
- Automated Endoscope Reprocessors
- Endoscope Drying, Storage and Transport Systems
- Others
High-level disinfectants and test strips account for the majority of the market share
The report has provided a detailed breakup and analysis of the market based on the product. This includes high-level disinfectants and test strips, detergents and wipes, automated endoscope reprocessors, endoscope drying, storage and transport systems, and others. According to the report, high-level disinfectants and test strips represented the largest segment.
High-level disinfectants and test strips are enhancing the endoscope reprocessing market value due to their effectiveness and efficiency in ensuring patient safety. These disinfectants are specifically designed to eliminate a broad spectrum of pathogens, including highly resistant organisms such as Mycobacterium tuberculosis and Clostridium difficile. Their fast-acting nature and compatibility with automated reprocessors make them a preferred choice in busy healthcare settings. Test strips play a crucial role in validating the efficacy of the disinfection process, providing immediate confirmation that the minimum recommended concentration of the disinfectant is met, thus ensuring compliance with safety standards and reducing the risk of infections. For instance, RAPICIDE high-level disinfectant is a glutaraldehyde-based, fast-acting, reusable solution for flexible endoscopes in automated reprocessors, requiring no mixing or activation. It reprocesses heat-sensitive, semi-critical medical devices in systems like the DSD-201, inactivating pathogens such as TB, C-diff, and CRE with a 5-minute contact time at 95°F (35°C). The disinfectant enhances efficiency with only two rinses needed. RAPICIDE™ Test Strips confirm the minimum recommended concentration of glutaraldehyde, requiring a 3-second dip and a 75-second read time. This example illustrates how specific products drive the demand for high-level disinfectants and test strips in the market, thereby increasing the endoscope reprocessing market value.
Breakup by Process:
- Manual Cleaning
- Automated Cleaning
Automated cleaning holds the largest share of the industry
A detailed breakup and analysis of the market based on the process have also been provided in the report. This includes manual cleaning and automated cleaning. According to the report, automated cleaning accounted for the largest market share.
Automated cleaning dominates the endoscope reprocessing market due to its ability to ensure consistent and high-level disinfection, reducing the risk of human error associated with manual cleaning. Automated systems, such as automated endoscope reprocessors (AERs), provide standardized, efficient, and effective cleaning and disinfecting processes, which are critical for patient safety. These systems enhance workflow efficiency within healthcare facilities by minimizing both the time and labor involved in reprocessing endoscopes. Additionally, automated cleaning systems are specifically designed to accommodate the intricate designs of modern endoscopes, ensuring thorough decontamination and adherence to stringent regulatory standards. As part of the endoscope reprocessing market forecast, it is expected that the growing adoption of automated cleaning solutions will continue to shape the market, driven by advancements in technology and the increasing demand for reliable and efficient reprocessing methods.
For instance, in March 2023, PENTAX Medical, a division of the HOYA Group, was awarded the CE mark for its innovative automated pre-cleaning solution, the AquaTYPHOON. Developed in collaboration with PlasmaBiotics, this device offers a robust alternative to the traditional manual pre-cleaning step in endoscope reprocessing, effectively addressing the hygiene challenges healthcare providers encounter in daily clinical practice.
Breakup by End User:
Access the comprehensive market breakdown Request Sample
- Hospitals and Clinics
- Ambulatory Surgery Centers
- Others
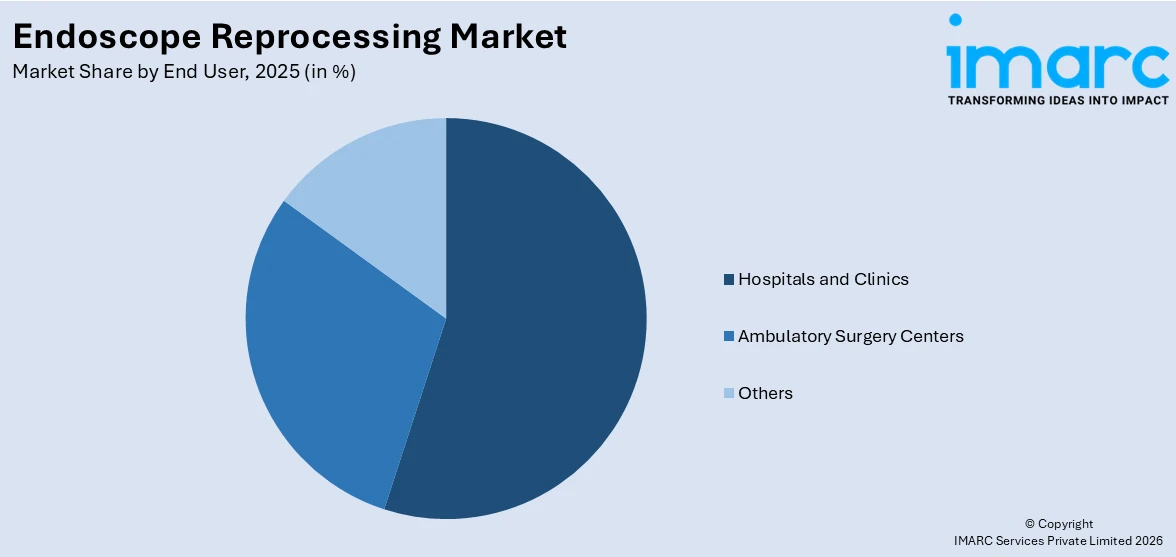
Hospitals and clinics represent the leading market segment
The report has provided a detailed breakup and analysis of the market based on the end user. This includes hospitals and clinics, ambulatory surgery centers, and others. According to the report, hospitals and clinics represented the largest segment.
The endoscope reprocessing market research report highlights that hospitals and clinics are the dominant segment in the market, attributed to the large volume of endoscopic procedures conducted within these facilities. According to industry reports, the number of bariatric surgery procedures in Ireland reached 219 in 2023, significantly surpassing the revised target of 200 set by the Health Service Executive (HSE). This marked a substantial increase from the 109 procedures performed in 2022. These surgeries were conducted at three HSE sites: St Vincent’s University Hospital in Dublin, St Michael’s Hospital in Dún Laoghaire, and University Hospital Galway. Additionally, University Hospital Galway alone performed 49 surgeries in 2023, up from 19 in the previous year.
These facilities require stringent infection control measures to prevent hospital-acquired infections, making effective reprocessing essential. The increasing prevalence of minimally invasive surgeries, which frequently utilize endoscopes, further drives demand. Additionally, regulatory requirements and guidelines mandate rigorous reprocessing protocols to ensure patient safety. The combination of high procedural volume, regulatory compliance, and the critical need for infection prevention establishes hospitals and clinics as the primary market segment for endoscope reprocessing.
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America leads the market, accounting for the largest endoscope reprocessing market share
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America represents the largest regional market for endoscope reprocessing.
North America's dominance in the endoscope reprocessing market can be attributed to its advanced healthcare infrastructure, increasing prevalence of chronic diseases, stringent regulatory standards, and high frequency of endoscopic procedures. For example, a 2024 article by the American Society of Anesthesiologists indicates that approximately 6.1 million upper endoscopies are performed annually in the U.S. Furthermore, the CDC reports that around 129 million Americans suffer from at least one major chronic condition, significantly driving the need for endoscope reprocessing solutions. As endoscopic procedures become more common for both diagnostic and therapeutic purposes, the endoscope reprocessing market outlook reflects a growing demand for effective reprocessing technologies. The region's healthcare systems, equipped with sophisticated technologies and protocols, ensure effective endoscope reprocessing to prevent infections. Additionally, heightened awareness of infection control among healthcare providers and patients, along with continuous technological advancements and a focus on patient safety, further solidify North America's leading position in the market.
Competitive Landscape:
- The market research report has also provided a comprehensive analysis of the competitive landscape in the market. Detailed profiles of all major companies have also been provided. Some of the major market players in the endoscope reprocessing industry include Advanced Sterilization Products (ASP) (Fortive Corporation), ARC Healthcare Solutions Inc., Belimed AG (Metall Zug AG), BES Rehab Ltd, Custom Ultrasonics Inc., Ecolab Inc., ENDO-TECHNIK W. Griesat GmbH (Albyn Medical S.L.), Getinge AB, HOYA Corporation, Metrex Research LLC, Olympus Corporation, Shinva Medical Instrument Co. Ltd., Steelco S.p.A. (Miele) and Steris plc.
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
- According to the endoscope reprocessing market overview, the competitive landscape is marked by intense rivalry among leading players, spurred by rapid technological advancements and the launch of innovative products. Companies are intensely focused on developing highly efficient and effective reprocessing solutions to address the growing demand for superior infection control and patient safety. This includes advancements in automated systems and the increased use of single-use endoscopes designed to reduce contamination risks. The market dynamics are further influenced by stringent regulatory standards that require high-quality reprocessing equipment to ensure compliance. To navigate these challenges and opportunities, firms frequently engage in strategic partnerships, mergers, and acquisitions to expand their product portfolios and geographic reach. Furthermore, ongoing investments in research and development (R&D) are crucial, as they drive innovation and refinement of reprocessing technologies, thereby intensifying competition among endoscope reprocessing companies.
- For instance, in November 2023, HOYA Corporation announced the complete acquisition of WASSENBURG Medical B.V., a global manufacturer known for its comprehensive range of endoscope reprocessing systems. This acquisition is anticipated to help WASSENBURG enhance innovation by combining its engineering capabilities with HOYA's PENTAX Medical division.
- In January 2024, Canon Medical Systems and Olympus Corporation announced a business alliance to collaborate on Endoscopic Ultrasound Systems (EUS). This collaboration aims to combine Canon's advanced Aplio i800 diagnostic ultrasound system with Olympus' Ultrasound Endoscope to deliver high-quality imaging solutions.
Endoscope Reprocessing Market News:
- In June 2023, Olympus announced the launch of the new ETD Endoscope Washer Disinfector, available in Basic and Premium versions, aimed at enhancing efficiency and sustainability in endoscope reprocessing. The device completes a full reprocessing cycle in just 29 minutes and uses less water, chemicals, and electricity, making it eco-friendlier and more cost-effective. It employs Peracetic Acid (PAA) or Glutaraldehyde (GA) for disinfection, achieving a 6-log reduction in microbial contamination. Features such as ergonomic design, removable baskets, touch-free adapter connection, and an intelligent drying program streamline the process and reduce the workload for healthcare professionals.
- In January 2023, IQ Endoscopes, a medical device company, secured a USD 6.6 million investment in a funding round led by BGF. This investment will support the development and launch of IQ Endoscope’s new medical device aimed at early diagnosis of various cancers and gastrointestinal conditions. Consequently, the increasing introduction of advanced endoscopy devices is expected to drive the demand for endoscope reprocessing solutions.
Endoscope Reprocessing Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Billion USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Products Covered | High-Level Disinfectants and Test Strips, Detergents and Wipes, Automated Endoscope Reprocessors, Endoscope Drying, Storage, And Transport Systems, Others |
| Processes Covered | Manual Cleaning, Automated Cleaning |
| End Users Covered | Hospitals and Clinics, Ambulatory Surgery Centers, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Advanced Sterilization Products (ASP) (Fortive Corporation), ARC Healthcare Solutions Inc., Belimed AG (Metall Zug AG), BES Rehab Ltd, Custom Ultrasonics Inc., Ecolab Inc., ENDO-TECHNIK W. Griesat GmbH (Albyn Medical S.L.), Getinge AB, HOYA Corporation, Metrex Research LLC, Olympus Corporation, Shinva Medical Instrument Co. Ltd., Steelco S.p.A. (Miele), Steris plc, etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the endoscope reprocessing market from 2020-2034.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the global endoscope reprocessing market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the endoscope reprocessing industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Frequently Asked Questions About the Endoscope Reprocessing Market Report
The global endoscope reprocessing market was valued at USD 2.47 Billion in 2025.
We expect the global endoscope reprocessing market to exhibit a CAGR of 5.97% during 2026-2034.
The sudden outbreak of the COVID-19 pandemic has led to rising demand for endoscope reprocessing to treat the coronavirus-infected patients with gastrointestinal disease symptoms, such as nausea, vomiting, and diarrhea.
The growing adoption of endoscope reprocessing to prevent the risk of infection and to improve patient safety from unusual symptoms, including dysphagia, diarrhea, weight loss, etc., is primarily driving the global endoscope reprocessing market.
Based on the product, the global endoscope reprocessing market can be categorized into high-level disinfectants and test strips, detergents and wipes, automated endoscope reprocessors, endoscope drying, storage and transport systems, and others. Among these, high-level disinfectants and test strips account for the majority of the total market share.
Based on the process, the global endoscope reprocessing market has been segregated into manual cleaning and automated cleaning, where automated cleaning currently holds the largest market share.
Based on the end user, the global endoscope reprocessing market can be bifurcated into hospitals and clinics, ambulatory surgery centers, and others. Currently, hospitals and clinics exhibit a clear dominance in the market.
On a regional level, the market has been classified into North America, Asia-Pacific, Europe, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global endoscope reprocessing market include Advanced Sterilization Products (ASP) (Fortive Corporation), ARC Healthcare Solutions Inc., Belimed AG (Metall Zug AG), BES Rehab Ltd, Custom Ultrasonics Inc., Ecolab Inc., ENDO-TECHNIK W. Griesat GmbH (Albyn Medical S.L.), Getinge AB, HOYA Corporation, Metrex Research LLC, Olympus Corporation, Shinva Medical Instrument Co. Ltd., Steelco S.p.A. (Miele) and Steris plc.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












